Product Characteristics
ANNEX I
SUMMARY OF PRODUCT CHARACTERISTICS
1. NAME OF THE MEDICINAL PRODUCT
Apidra 100 Units/ml solution for injection in a vial
QUALITATIVE AND QUANTITATIVE COMPOSITION
Each ml contains 100 Units insulin glulisine (equivalent to 3.49 mg).
Each vial contains 10 ml of solution for injection, equivalent to 1000 Units.
Insulin glulisine is produced by recombinant DNA technology in
Escherichia coli
.
For a full list of excipients, see section 6.1.
Solution for injection in a vial.
Clear, colourless, aqueous solution.
4.1 Therapeutic indications
Treatment of adults, adolescents and children, 6 years or older with diabetes mellitus, where treatment
with insulin is required.
4.2 Posology and method of administration
The potency of this preparation is stated in units. These units are exclusive to Apidra and are not the
same as IU or the units used to express the potency of other insulin analogues (see section 5.1).
Apidra should be used in regimens that include an intermediate or long acting insulin or basal insulin
analogue and can be used with oral hypoglycaemic agents.
The dose of Apidra should be individually adjusted.
Renal impairment
The pharmacokinetic properties of insulin glulisine are generally maintained in patients with renal
impairment. However, insulin requirements may be reduced in the presence of renal impairment (see
section 5.2).
Hepatic impairment
The pharmacokinetic properties of insulin glulisine have not been investigated in patients with
decreased liver function. In patients with hepatic impairment, insulin requirements may be diminished
due to reduced capacity for gluconeogenesis and reduced insulin metabolism.
Elderly
Limited pharmacokinetic data are available in elderly patients with diabetes mellitus. Deterioration of
renal function may lead to a decrease in insulin requirements.
There is insufficient clinical information on the use of Apidra in children younger than the age of
6 years.
Intravenous use
Apidra can be administered intravenously. This should be carried out by health care professionals.
Apidra must not be mixed with glucose or Ringer’s solution or with any other insulin.
Subcutaneous use
Apidra should be given by subcutaneous injection shortly (0-15 min) before or soon after meals or by
continuous subcutaneous pump infusion.
Apidra should be administered subcutaneously in the abdominal wall, thigh or deltoid or by
continuous infusion in the abdominal wall. Injection sites and infusion sites within an injection area
(abdomen, thigh or deltoid) should be rotated from one injection to the next. The rate of absorption,
and consequently the onset and duration of action, may be affected by the injection site, exercise and
other variables. Subcutaneous injection in the abdominal wall ensures a slightly faster absorption than
other injection sites (see section 5.2).
Care should be taken to ensure that a blood vessel has not been entered. After injection, the site of
injection should not be massaged. Patients must be educated to use proper injection techniques.
When used with a subcutaneous insulin infusion pump, Apidra must not be mixed with diluents or any
other insulin.
Mixing with insulins
When administered as a subcutaneous injection, Apidra must not be mixed with other medicinal
products except NPH human insulin.
For further details on handling, see section 6.6.
Hypersensitivity to the active substance or to any of the excipients.
4.4 Special warnings and precautions for use
Transferring a patient to another type or brand of insulin should be done under strict medical
supervision. Changes in strength, brand (manufacturer), type (regular, neutral protamine Hagedorn
[NPH], lente, long-acting, etc.), origin (animal, human, human insulin analogue) and/or method of
manufacture may result in the need for a change in dose. Concomitant oral antidiabetic treatment may
need to be adjusted.
The use of inadequate doses or discontinuation of treatment, especially in insulin-dependent diabetic,
may lead to hyperglycaemia and diabetic ketoacidosis; conditions which are potentially lethal.
The time of occurrence of hypoglycaemia depends on the action profile of the insulins used and may,
therefore, change when the treatment regimen is changed.
Conditions which may make the early warning symptoms of hypoglycaemia different or less
pronounced include long duration of diabetes, intensified insulin therapy, diabetic nerve disease,
medicinal products such as beta blockers or after transfer from animal-source insulin to human insulin.
Adjustment of dose may be also necessary if patients undertake increased physical activity or change
their usual meal plan. Exercise taken immediately after a meal may increase the risk of
hypoglycaemia.
When compared with soluble human insulin, if hypoglycaemia occurs after an injection with rapid
acting analogues, it may occur earlier.
Uncorrected hypoglycaemic or hyperglycaemic reactions can cause loss of consciousness, coma, or
death.
Insulin requirements may be altered during illness or emotional disturbances.
Medication errors have been reported in which other insulins, particularly long-acting insulins, have
been accidentally administered instead of insulin glulisine. Insulin label must always be checked
before each injection to avoid medication errors between insulin glulisine and other insulins.
This medicinal product contains less than 1 mmol (23 mg) sodium per dose, i.e. it is essentially
‘sodium-free’.
Apidra contains metacresol, which may cause allergic reactions.
Combination of Apidra with pioglitazone
Cases of cardiac failure have been reported when pioglitazone was used in combination with insulin,
especially in patients with risk factors for development of cardiac heart failure. This should be kept in
mind if treatment with the combination of pioglitazone and Apidra is considered. If the combination is
used, patients should be observed for signs and symptoms of heart failure, weight gain and oedema.
Pioglitazone should be discontinued if any deterioration in cardiac symptoms occurs.
4.5 Interaction with other medicinal products and other forms of interaction
Studies on pharmacokinetic interactions have not been performed. Based on empirical knowledge
from similar medicinal products, clinically relevant pharmacokinetic interactions are unlikely to occur.
A number of substances affect glucose metabolism and may require dose adjustment of insulin
glulisine and particularly close monitoring.
Substances that may enhance the blood-glucose-lowering activity and increase susceptibility to
hypoglycaemia include oral antidiabetic agents, angiotensin converting enzyme (ACE) inhibitors,
disopyramide, fibrates, fluoxetine, monoamine oxidase inhibitors (MAOIs), pentoxifylline,
propoxyphene, salicylates and sulfonamide antibiotics.
Substances that may reduce the blood-glucose-lowering activity include corticosteroids, danazol,
diazoxide, diuretics, glucagon, isoniazid, phenothiazine derivatives, somatropin, sympathomimetic
agents (e.g. epinephrine [adrenaline], salbutamol, terbutaline), thyroid hormones, estrogens, progestins
(e.g. in oral contraceptives), protease inhibitors and atypical antipsychotic medicinal products (e.g.
olanzapine and clozapine).
Beta-blockers, clonidine, lithium salts or alcohol may either potentiate or weaken the
blood-glucose-lowering activity of insulin. Pentamidine may cause hypoglycaemia, which may
sometimes be followed by hyperglycaemia.
In addition, under the influence of sympatholytic medicinal products such as beta-blockers, clonidine,
guanethidine and reserpine, the signs of adrenergic counter-regulation may be reduced or absent.
4.6 Pregnancy and lactation
There are no adequate data on the use of insulin glulisine in pregnant women.
Animal reproduction studies have not revealed any differences between insulin glulisine and human
insulin regarding pregnancy, embryonal/foetal development, parturition or postnatal development (see
section 5.3).
Caution should be exercised when prescribing to pregnant women. Careful monitoring of glucose
control is essential.
It is essential for patients with pre-existing or gestational diabetes to maintain good metabolic control
throughout pregnancy. Insulin requirements may decrease during the first trimester and generally
increase during the second and third trimesters. Immediately after delivery, insulin requirements
decline rapidly.
It is unknown whether insulin glulisine is excreted in human milk, but in general insulin does not pass
into breast milk and is not absorbed after oral administration.
Breast-feeding mothers may require adjustments in insulin dose and diet.
4.7 Effects on ability to drive and use machines
The patient's ability to concentrate and react may be impaired as a result of hypoglycaemia or
hyperglycaemia or, for example, as a result of visual impairment. This may constitute a risk in
situations where these abilities are of special importance (e.g. driving a car or operating machines).
Patients should be advised to take precautions to avoid hypoglycaemia whilst driving. This is
particularly important in those who have reduced or absent awareness of the warning symptoms of
hypoglycaemia or have frequent episodes of hypoglycaemia. The advisability of driving should be
considered in these circumstances.
Hypoglycaemia, the most frequent undesirable effect of insulin therapy, may occur if the insulin dose
is too high in relation to the insulin requirement.
The following related adverse reactions from clinical studies were listed below by system organ class
and in order of decreasing incidence (very common: 1/10; common: 1/100 to <1/10; uncommon:
1/1,000 to <1/100; rare: 1/10,000 to <1/1,000; very rare: <1/10,000), not known (cannot be
estimated from the available data).
Within each frequency grouping, undesirable effects are presented in order of decreasing seriousness.
MedDRA
Organ system
classes
Metabolism
and nutrition
disorders
Skin and
subcutaneous
tissue
disorders
Injection site
reactions
Local
hypersensitivity
reactions
General
disorders and
administration
site conditions
Systemic
hypersensitivity
reactions
Metabolism and nutrition disorders
Symptoms of hypoglycaemia usually occur suddenly. They may include cold sweats, cool pale skin,
fatigue, nervousness or tremor, anxiousness, unusual tiredness or weakness, confusion, difficulty in
concentration, drowsiness, excessive hunger, vision changes, headache, nausea and palpitation.
Hypoglycaemia can become severe and may lead to unconsciousness and/or convulsions and may
result in temporary or permanent impairment of brain function or even death.
Skin and subcutaneous tissue disorders
Local hypersensitivity reactions (redness, swelling and itching at the injection site) may occur during
treatment with insulin. These reactions are usually transitory and normally they disappear during
continued treatment.
Lipodystrophy may occur at the injection site as a consequence of failure to rotate injection sites
within an area.
General disorders and administration site conditions
Systemic hypersensitivity reactions may include urticaria, chest tightness, dyspnea, allergic dermatitis
and pruritus. Severe cases of generalized allergy, including anaphylactic reaction, may be
life-threatening.
Hypoglycaemia may occur as a result of an excess of insulin activity relative to food intake and energy
expenditure.
There are no specific data available concerning overdoses with insulin glulisine. However,
hypoglycaemia may develop over sequential stages:
Mild hypoglycaemic episodes can be treated by oral administration of glucose or sugary products. It is
therefore recommended that the diabetic patient constantly carries some sugar lumps, sweets, biscuits
or sugary fruit juice.
Severe hypoglycaemic episodes, where the patient has become unconscious, can be treated by
glucagon (0.5 mg to 1 mg) given intramuscularly or subcutaneously by a person who has received
appropriate instruction, or by glucose given intravenously by a healthcare professional. Glucose must
also be given intravenously, if the patient does not respond to glucagon within 10 to 15 minutes.











Upon regaining consciousness, administration of oral carbohydrate is recommended for the patient in
order to prevent relapse.
After an injection of glucagon, the patient should be monitored in a hospital in order to find the reason
for this severe hypoglycaemia and prevent other similar episodes.
5. PHARMACOLOGICAL PROPERTIES
5.1 Pharmacodynamic properties
Pharmacotherapeutic group: Insulins and analogues for injection, fast-acting. ATC code: A10AB06
Insulin glulisine is a recombinant human insulin analogue that is equipotent to regular human insulin.
Insulin glulisine has a more rapid onset of action and a shorter duration of action than regular human
insulin.
The primary activity of insulins and insulin analogues, including insulin glulisine, is regulation of
glucose metabolism. Insulins lower blood glucose levels by stimulating peripheral glucose uptake,
especially by skeletal muscle and fat, and by inhibiting hepatic glucose production. Insulin inhibits
lipolysis in the adipocyte, inhibits proteolysis and enhances protein synthesis.
Studies in healthy volunteers and patients with diabetes demonstrated that insulin glulisine is more
rapid in onset of action and of shorter duration of action than regular human insulin when given
subcutaneously. When insulin glulisine is injected subcutaneously, the glucose lowering activity will
begin within 10 – 20 minutes. After intravenous administration, a faster onset and shorter duration of
action, as well as a greater peak response were observed as compared with subcutaneous
administration. The glucose-lowering activities of insulin glulisine and regular human insulin are
equipotent when administered by intravenous route. One unit of insulin glulisine has the same
glucose - lowering activity as one unit of regular human insulin.
In a study with 18 male subjects with diabetes mellitus type 1 aged 21 to 50 years, insulin glulisine
displayed dose-proportional glucose lowering effect in the therapeutic relevant dose range 0.075 to
0.15 Units/kg,
and less than proportional increase in glucose lowering effect with 0.3 Units/kg or
higher, like human insulin.
Insulin glulisine takes effect about twice as fast as regular human insulin and completes the glucose
lowering effect about 2 hours earlier than regular human insulin.
A phase I study carried out with insulin glulisine, lispro and regular human insulin in an obese
population has demonstrated that insulin glulisine maintains its rapid-acting properties. In this study,
the time to 20 % of total AUC and the AUC (0-2h) representing the early glucose lowering activity
were respectively of 114 minutes and 427 mg/kg
for insulin glulisine, 121 minutes and 354 mg/kg for
lispro, 150 minutes and 197 mg/kg for regular human insulin (see figure 2).
Figure 2: Glucose infusion rates (GIR) after subcutaneous injection of 0.3 Units/kg of insulin glulisine
(GLULISINE) or insulin lispro (LISPRO) or regular human insulin (REGULAR) in an obese
population.
Another phase I study with insulin glulisine and insulin lispro in a non-diabetic population in
80 subjects with a wide range of body mass indices (18-46 kg/m²) has demonstrated that rapid action
is generally maintained across a wide range of body mass indices (BMI), while total glucose lowering
effect decreases with increasing obesity.
The average total GIR AUC between 0–1 hour was 102±75 mg/kg and 158±100 mg/kg with 0.2 and
0.4 Units/kg
insulin glulisine,
respectively, and was 83.1±72.8 mg/kg and 112.3±70.8 mg/kg with
0.2 and 0.4 Units/kg
insulin
lispro respectively.
A phase I study in 18 obese patients with type 2 diabetes mellitus (BMI between 35 and 40 kg/m
2
)
with insulin glulisine and insulin lispro [90% CI:0.81, 0.95 (p=<0.01)] has shown that insulin glulisine
effectively controls diurnal post-prandial blood glucose excursions.
Type 1 diabetes mellitus-Adults
In a 26-week phase III clinical study comparing insulin glulisine with insulin lispro both injected
subcutaneously shortly (0-15 minutes) before a meal in patients with type 1 diabetes mellitus using
insulin glargine as basal insulin, insulin glulisine was comparable to insulin lispro for glycemic control
as reflected by changes in glycated haemoglobin (expressed as HbA
1c
equivalent) from baseline to
endpoint. Comparable self-monitored blood glucose values were observed. No increase in the basal
insulin dose was needed with insulin glulisine, in contrast to insulin lispro.
A 12-week phase III clinical study performed in patients with type 1 diabetes mellitus receiving
insulin glargine as basal therapy indicate that the immediate postmeal administration of insulin
glulisine provides efficacy that was comparable to immediate premeal insulin glulisine (0-15 minutes)
or regular insulin (30-45 minutes).



































































































In the per protocol population there was a significantly larger observed reduction in GHb in the
premeal glulisine group compared with the regular insulin group.
Type 1 diabetes mellitus-Paediatric
A 26-week phase III clinical study compared insulin glulisine with insulin lispro both injected
subcutaneously shortly (0-15 minutes) before a meal in children (4-5 years: n=9; 6-7 years: n=32 and
8-11 years: n=149) and adolescents (12-17 years: n=382) with type 1 diabetes mellitus using insulin
glargine or NPH as basal insulin. Insulin glulisine was comparable to insulin lispro for glycaemic
control as reflected by changes in glycated haemoglobin (GHb expressed as HbA
1c
equivalent) from
baseline to endpoint and by self-monitored blood glucose values.
There is insufficient clinical information on the use of Apidra in children younger than the age of
6 years.
Type 2 diabetes mellitus-Adults
A 26-week phase III clinical study followed by a 26-week extension safety study was conducted to
compare insulin glulisine (0-15 minutes before a meal) with regular human insulin (30-45 minutes
before a meal) injected subcutaneously in patients with type 2 diabetes mellitus also using NPH
insulin as basal insulin. The average body mass index (BMI) of patients was 34.55 kg/m
2
. Insulin
glulisine was shown to be comparable to regular human insulin with regard to glycated haemoglobin
(expressed as HbA
1c
equivalent) changes from baseline to the 6-month endpoint (-0.46% for insulin
glulisine and -0.30% for regular human insulin, p=0.0029) and from baseline to the 12-month endpoint
(-0.23% for insulin glulisine and -0.13% for regular human insulin, difference not significant). In this
study, the majority of patients (79%) mixed their short acting insulin with NPH insulin immediately
prior to injection and 58% of subjects used oral hypoglycemic agents at randomization and were
instructed to continue to use them at the same dose.
Race and gender
In controlled clinical trials in adults, insulin glulisine did not show differences in safety and efficacy in
subgroup analyses based on race and gender.
5.2 Pharmacokinetic properties
In insulin glulisine the replacement of the human insulin amino acid asparagine in position B3 by
lysine and the lysine in position B29 by glutamic acid favors more rapid absorption.
In a study with 18 male subjects with diabetes mellitus type 1 aged 21 to 50 years, insulin glulisine
displays dose-proportionality for early, maximum and total exposure in the dose range 0.075
to 0.4 Units/kg
.
Absorption and bioavailability
Pharmacokinetic profiles in healthy volunteers and diabetes patients (type 1 or 2) demonstrated that
absorption of insulin glulisine was about twice as fast with a peak concentration approximately twice
as high as compared to regular human insulin.
In a study in patients with type 1 diabetes mellitus after subcutaneous administration of 0.15 Units/kg,
for insulin glulisine the T
max
was 55 minutes and C
max
was 82 ± 1.3 µUnits/ml compared to a T
max
of
82 minutes and a C
max
of 46 ± 1.3 µUnits/ml for regular human insulin. The mean residence time of
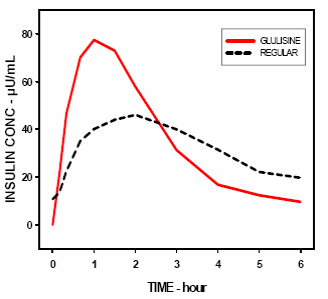
insulin glulisine was shorter (98 min) than for regular human insulin (161 min) (see figure 3).
Figure 3: Pharmacokinetic profile of insulin glulisine and regular human insulin in type 1 diabetes
mellitus patients after a dose of 0.15 Units/kg.
In a study in patients with type 2 diabetes mellitus after subcutaneous administration of 0.2 Units/kg
insulin glulisine, the C
max
was 91 µUnits/ml with the interquartile range from 78 to 104 µUnits/ml.
When insulin glulisine was injected subcutaneously into abdomen, deltoid and thigh, the
concentration-time profiles were similar with a slightly faster absorption when administered in the
abdomen compared to the thigh. Absorption from deltoid sites was in-between (see section 4.2). The
absolute bioavailability (70%) of insulin glulisine was similar between injection sites and of low
intra-subject variability (11%CV). Intravenous bolus administration of insulin glulisine resulted in a
higher systemic exposure when compared to subcutaneous injection, with a C
max
approximately
40-fold higher.
Obesity
Another phase I study with insulin glulisine and insulin lispro in a non-diabetic population in
80 subjects with a wide range of body mass indices (18-46 kg/m²) has demonstrated that rapid
absorption and total exposure is generally maintained across a wide range of body mass indices.
The time to 10% of total INS exposure was reached earlier by approximately 5–6 min with insulin
glulisine.
Distribution and elimination
The distribution and elimination of insulin glulisine and regular human insulin after intravenous
administration is similar with volumes of distribution of 13 l and 22 l and half-lives of 13 and
18 minutes, respectively.
After subcutaneous administration, insulin glulisine is eliminated more rapidly than regular human
insulin with an apparent half-life of 42 minutes compared to 86 minutes. In an across study analysis of
insulin glulisine in either healthy subjects or subjects with type 1 or type 2 diabetes mellitus the
apparent half-life ranged from 37 to 75 minutes (interquartile range).
Insulin glulisine shows low plasma protein binding, similar to human insulin.
Renal impairment
In a clinical study performed in non-diabetic subjects covering a wide range of renal function
(CrCl > 80 ml/min, 30-50 ml/min, < 30 ml/min), the rapid-acting properties of insulin glulisine were
generally maintained. However, insulin requirements may be reduced in the presence of renal
impairment.
Hepatic impairment
The pharmacokinetic properties have not been investigated in patients with impaired liver function.
Elderly
Very limited pharmacokinetic data are available for elderly patients with diabetes mellitus.
Children and adolescents
The pharmacokinetic and pharmacodynamic properties of insulin glulisine were investigated in
children (7-11 years) and adolescents (12-16 years) with type 1 diabetes mellitus. Insulin glulisine was
rapidly absorbed in both age groups, with similar T
max
and C
max
as in adults (see section 4.2).
Administered immediately before a test meal, insulin glulisine provided better postprandial control
than regular human insulin, as in adults (see section 5.1). The glucose excursion (AUC
0-6h
) was
641 mg.h.dl
-1
for insulin glulisine and 801 mg.h.dl
-1
for regular human insulin.
5.3 Preclinical safety data
Non-clinical data did not reveal toxicity findings others than those linked to the blood glucose
lowering pharmacodynamic activity (hypoglycemia), different from regular human insulin or of
clinical relevance for humans.
PHARMACEUTICAL PARTICULARS
Metacresol
Sodium chloride
Trometamol
Polysorbate 20
Hydrochloric acid, concentrated
Sodium hydroxide
Water for injections
In the absence of compatibility studies, this medicinal product must not be mixed with other medicinal
products except NPH human insulin.
When used with an insulin infusion pump, Apidra must not be mixed with other medicinal products.
Apidra was found to be incompatible with Glucose 5 % solution and Ringer’s solution and, therefore,
must not be used with these solution fluids. The use of other solutions has not been studied.
Shelf life after first use of the vial
The product may be stored for a maximum of 4 weeks below 25°C away from direct heat or direct
light. Keep the vial in the outer carton in order to protect from light.
It is recommended that the date of the first use from the vial be noted on the label.
Shelf life for intravenous use
Insulin glulisine for intravenous use at a concentration of 1 Unit/ml is stable between 15 C and 25 ºC
for 48 hours (see section 6.6).
6.4 Special precautions for storage
Store in a refrigerator (2°C - 8°C).
Do not freeze.
Do not put Apidra next to the freezer compartment or a freezer pack.
Keep the vial in the outer carton in order to protect from light.
For storage conditions, see section 6.3.
6.5 Nature and contents of container
10 ml solution in a vial (type I colourless glass) with a stopper (flanged aluminium overseal,
elastomeric chlorobutyl
rubber) and a polypropylene tear-off cap. Packs of 1, 2, 4 and 5 vials are
available.
Not all pack sizes may be marketed.
6.6 Special precautions for disposal and other handling
Subcutaneous use
Apidra vials are for use with insulin syringes with the corresponding unit scale and for use with an
insulin pump system (see section 4.2).
Inspect the vial before use. It must only be used if the solution is clear, colourless, with no solid
particles visible. Since Apidra is a solution, it does not require resuspension before use.
Insulin label must always be checked before each injection to avoid medication errors between insulin
glulisine and other insulins (see section 4.4).
Mixing with insulins
When mixed with NPH human insulin, Apidra should be drawn into the syringe first. Injection should
be given immediately after mixing as no data are available regarding the mixtures made up a
significant time before injection.
Continuous subcutaneous infusion pump
Apidra may be used for Continuous Subcutaneous Insulin Infusion (CSII) in pump systems suitable
for insulin infusion with the appropriate catheters and reservoirs.
Patients using CSII should be comprehensively instructed on the use of the pump system. The infusion
set and reservoir should be changed every 48 hours using aseptic technique.
Patients administering Apidra by CSII must have alternative insulin available in case of pump system
failure.
Apidra should be used at a concentration of 1 Unit/ml insulin glulisine in infusion systems with
sodium chloride 9 mg/ml (0.9%) solution for infusion with or without 40 mmol/l potassium chloride
using coextruded polyolefin/polyamide plastic infusion bags with a dedicated infusion line. Insulin
glulisine for intravenous use at a concentration of 1 Unit/ml is stable at room temperature for 48 hours.
After dilution for intravenous use, the solution should be inspected visually for particulate matter prior
to administration. It must only be used if the solution is clear and colourless, not when cloudy or with
visible particles.
Apidra was found to be incompatible with Glucose 5% solution and Ringer’s solution and, therefore,
must not be used with these solution fluids. The use of other solutions has not been studied.
MARKETING AUTHORISATION HOLDER
Sanofi-Aventis Deutschland GmbH
D-65926 Frankfurt am Main
Germany.
MARKETING AUTHORISATION NUMBER(S)
DATE OF FIRST AUTHORISATION/RENEWAL OF THE AUTHORISATION
Date of first authorisation: 27 September 2004
Date of latest renewal: 20 August 2009
10. DATE OF REVISION OF THE TEXT
Detailed information on this product is available on the website of the European Medicines Agency
http://www.ema.europa.eu/
NAME OF THE MEDICINAL PRODUCT
Apidra 100 Units/ml solution for injection in a cartridge
QUALITATIVE AND QUANTITATIVE COMPOSITION
Each ml contains 100 Units insulin glulisine (equivalent to 3.49 mg).
Each cartridge contains 3 ml of solution for injection, equivalent to 300 Units.
Insulin glulisine is produced by recombinant DNA technology in
Escherichia coli
.
For a full list of excipients, see section 6.1.
Solution for injection in a cartridge.
Clear, colourless, aqueous solution.
4.1 Therapeutic indications
Treatment of adults, adolescents and children, 6 years or older with diabetes mellitus, where treatment
with insulin is required.
4.2 Posology and method of administration
The potency of this preparation is stated in units. These units are exclusive to Apidra and are not the
same as IU or the units used to express the potency of other insulin analogues (see section 5.1).
Apidra should be used in regimens that include an intermediate or long acting insulin or basal insulin
analogue and can be used with oral hypoglycaemic agents.
The dose of Apidra should be individually adjusted.
Renal impairment
The pharmacokinetic properties of insulin glulisine are generally maintained in patients with renal
impairment. However, insulin requirements may be reduced in the presence of renal impairment (see
section 5.2).
Hepatic impairment
The pharmacokinetic properties of insulin glulisine have not been investigated in patients with
decreased liver function. In patients with hepatic impairment, insulin requirements may be diminished
due to reduced capacity for gluconeogenesis and reduced insulin metabolism.
Elderly
Limited pharmacokinetic data are available in elderly patients with diabetes mellitus. Deterioration of
renal function may lead to a decrease in insulin requirements.
There is insufficient clinical information on the use of Apidra in children younger than the age of
6 years.
Apidra should be given by subcutaneous injection shortly (0-15 min) before or soon after meals or by
continuous subcutaneous pump infusion.
Apidra should be administered subcutaneously in the abdominal wall, thigh or deltoid or by
continuous infusion in the abdominal wall. Injection sites and infusion sites within an injection area
(abdomen, thigh or deltoid) should be rotated from one injection to the next. The rate of absorption,
and consequently the onset and duration of action, may be affected by the injection site, exercise and
other variables. Subcutaneous injection in the abdominal wall ensures a slightly faster absorption than
other injection sites (see section 5.2).
Care should be taken to ensure that a blood vessel has not been entered. After injection, the site of
injection should not be massaged. Patients must be educated to use proper injection techniques.
Mixing with insulins
When administered as a subcutaneous injection, Apidra must not be mixed with other medicinal
products except NPH human insulin.
For further details on handling, see section 6.6.
Hypersensitivity to the active substance or to any of the excipients.
4.4 Special warnings and precautions for use
Transferring a patient to another type or brand of insulin should be done under strict medical
supervision. Changes in strength, brand (manufacturer), type (regular, neutral protamine Hagedorn
[NPH], lente, long-acting, etc.), origin (animal, human, human insulin analogue) and/or method of
manufacture may result in the need for a change in dose. Concomitant oral antidiabetic treatment may
need to be adjusted.
The use of inadequate doses or discontinuation of treatment, especially in insulin-dependent diabetic,
may lead to hyperglycaemia and diabetic ketoacidosis; conditions which are potentially lethal.
The time of occurrence of hypoglycaemia depends on the action profile of the insulins used and may,
therefore, change when the treatment regimen is changed.
Conditions which may make the early warning symptoms of hypoglycaemia different or less
pronounced include long duration of diabetes, intensified insulin therapy, diabetic nerve disease,
medicinal products such as beta blockers or after transfer from animal-source insulin to human insulin.
Adjustment of dose may be also necessary if patients undertake increased physical activity or change
their usual meal plan. Exercise taken immediately after a meal may increase the risk of
hypoglycaemia.
When compared with soluble human insulin, if hypoglycaemia occurs after an injection with rapid
acting analogues, it may occur earlier.
Uncorrected hypoglycaemic or hyperglycaemic reactions can cause loss of consciousness, coma, or
death.
Insulin requirements may be altered during illness or emotional disturbances.
Pens to be used with Apidra cartridges
The Apidra cartridges should only be used with the following pens: OptiPen, ClikSTAR, Tactipen and
Autopen 24 and should not be used with any other reusable pen as the dosing accuracy has only been
established with the listed pens.
Medication errors have been reported in which other insulins, particularly long-acting insulins, have
been accidentally administered instead of insulin glulisine. Insulin label must always be checked
before each injection to avoid medication errors between insulin glulisine and other insulins.
This medicinal product contains less than 1 mmol (23 mg) sodium per dose, i.e. it is essentially
‘sodium-free’.
Apidra contains metacresol, which may cause allergic reactions.
Combination of Apidra with pioglitazone
Cases of cardiac failure have been reported when pioglitazone was used in combination with insulin,
especially in patients with risk factors for development of cardiac heart failure. This should be kept in
mind if treatment with the combination of pioglitazone and Apidra is considered. If the combination is
used, patients should be observed for signs and symptoms of heart failure, weight gain and oedema.
Pioglitazone should be discontinued if any deterioration in cardiac symptoms occurs.
4.5 Interaction with other medicinal products and other forms of interaction
Studies on pharmacokinetic interactions have not been performed. Based on empirical knowledge
from similar medicinal products, clinically relevant pharmacokinetic interactions are unlikely to occur.
A number of substances affect glucose metabolism and may require dose adjustment of insulin
glulisine and particularly close monitoring.
Substances that may enhance the blood-glucose-lowering activity and increase susceptibility to
hypoglycaemia include oral antidiabetic agents, angiotensin converting enzyme (ACE) inhibitors,
disopyramide, fibrates, fluoxetine, monoamine oxidase inhibitors (MAOIs), pentoxifylline,
propoxyphene, salicylates and sulfonamide antibiotics.
Substances that may reduce the blood-glucose-lowering activity include corticosteroids, danazol,
diazoxide, diuretics, glucagon, isoniazid, phenothiazine derivatives, somatropin, sympathomimetic
agents (e.g. epinephrine [adrenaline], salbutamol, terbutaline), thyroid hormones, estrogens, progestins
(e.g. in oral contraceptives), protease inhibitors and atypical antipsychotic medicinal products (e.g.
olanzapine and clozapine).
Beta-blockers, clonidine, lithium salts or alcohol may either potentiate or weaken the
blood-glucose-lowering activity of insulin. Pentamidine may cause hypoglycaemia, which may
sometimes be followed by hyperglycaemia.
In addition, under the influence of sympatholytic medicinal products such as beta-blockers, clonidine,
guanethidine and reserpine, the signs of adrenergic counter-regulation may be reduced or absent.
4.6 Pregnancy and lactation
There are no adequate data on the use of insulin glulisine in pregnant women.
Animal reproduction studies have not revealed any differences between insulin glulisine and human
insulin regarding pregnancy, embryonal/foetal development, parturition or postnatal development (see
section 5.3).
Caution should be exercised when prescribing to pregnant women. Careful monitoring of glucose
control is essential.
It is essential for patients with pre-existing or gestational diabetes to maintain good metabolic control
throughout pregnancy. Insulin requirements may decrease during the first trimester and generally
increase during the second and third trimesters. Immediately after delivery, insulin requirements
decline rapidly.
It is unknown whether insulin glulisine is excreted in human milk, but in general insulin does not pass
into breast milk and is not absorbed after oral administration.
Breastfeeding mothers may require adjustments in insulin dose and diet.
4.7 Effects on ability to drive and use machines
The patient's ability to concentrate and react may be impaired as a result of hypoglycaemia or
hyperglycaemia or, for example, as a result of visual impairment. This may constitute a risk in
situations where these abilities are of special importance (e.g. driving a car or operating machines).
Patients should be advised to take precautions to avoid hypoglycaemia whilst driving. This is
particularly important in those who have reduced or absent awareness of the warning symptoms of
hypoglycaemia or have frequent episodes of hypoglycaemia. The advisability of driving should be
considered in these circumstances.
Hypoglycaemia, the most frequent undesirable effect of insulin therapy, may occur if the insulin dose
is too high in relation to the insulin requirement.
The following related adverse reactions from clinical studies were listed below by system organ class
and in order of decreasing incidence (very common: 1/10; common: 1/100 to <1/10; uncommon:
1/1,000 to <1/100; rare: 1/10,000 to <1/1,000; very rare: <1/10,000), not known (cannot be
estimated from the available data).
Within each frequency grouping, undesirable effects are presented in order of decreasing seriousness.
MedDRA
Organ system
classes
Metabolism
and nutrition
disorders
Skin and
subcutaneous
tissue
disorders
Injection site
reactions
Local
hypersensitivity
reactions
General
disorders and
administratio
n site
conditions
Systemic
hypersensitivity
reactions
Metabolism and nutrition disorders
Symptoms of hypoglycaemia usually occur suddenly. They may include cold sweats, cool pale skin,
fatigue, nervousness or tremor, anxiousness, unusual tiredness or weakness, confusion, difficulty in
concentration, drowsiness, excessive hunger, vision changes, headache, nausea and palpitation.
Hypoglycaemia can become severe and may lead to unconsciousness and/or convulsions and may
result in temporary or permanent impairment of brain function or even death.
Skin and subcutaneous tissue disorders
Local hypersensitivity reactions (redness, swelling and itching at the injection site) may occur during
treatment with insulin. These reactions are usually transitory and normally they disappear during
continued treatment.
Lipodystrophy may occur at the injection site as a consequence of failure to rotate injection sites
within an area.
General disorders and administration site conditions
Systemic hypersensitivity reactions may include urticaria, chest tightness, dyspnea, allergic dermatitis
and pruritus. Severe cases of generalized allergy, including anaphylactic reaction, may be
life-threatening.
Hypoglycaemia may occur as a result of an excess of insulin activity relative to food intake and energy
expenditure.
There are no specific data available concerning overdoses with insulin glulisine. However,
hypoglycaemia may develop over sequential stages.
Mild hypoglycaemic episodes can be treated by oral administration of glucose or sugary products. It is
therefore recommended that the diabetic patient constantly carries some sugar lumps, sweets, biscuits
or sugary fruit juice.
Severe hypoglycaemic episodes, where the patient has become unconscious, can be treated by
glucagon (0.5 mg to 1 mg) given intramuscularly or subcutaneously by a person who has received











appropriate instruction, or by glucose given intravenously by a healthcare professional. Glucose must
also be given intravenously, if the patient does not respond to glucagon within 10 to 15 minutes.
Upon regaining consciousness, administration of oral carbohydrate is recommended for the patient in
order to prevent relapse.
After an injection of glucagon, the patient should be monitored in a hospital in order to find the reason
for this severe hypoglycaemia and prevent other similar episodes.
PHARMACOLOGICAL PROPERTIES
5.1 Pharmacodynamic properties
Pharmacotherapeutic group: Insulins and analogues for injection, fast-acting. ATC code: A10AB06
Insulin glulisine is a recombinant human insulin analogue that is equipotent to regular human insulin.
Insulin glulisine has a more rapid onset of action and a shorter duration of action than regular human
insulin.
The primary activity of insulins and insulin analogues, including insulin glulisine, is regulation of
glucose metabolism. Insulins lower blood glucose levels by stimulating peripheral glucose uptake,
especially by skeletal muscle and fat, and by inhibiting hepatic glucose production. Insulin inhibits
lipolysis in the adipocyte, inhibits proteolysis and enhances protein synthesis.
Studies in healthy volunteers and patients with diabetes demonstrated that insulin glulisine is more
rapid in onset of action and of shorter duration of action than regular human insulin when given
subcutaneously. When insulin glulisine is injected subcutaneously, the glucose lowering activity will
begin within 10 – 20 minutes. The glucose-lowering activities of insulin glulisine and regular human
insulin are equipotent when administered by intravenous route. One unit of insulin glulisine has the
same glucose - lowering activity as one unit of regular human insulin.
In a study with 18 male subjects with diabetes mellitus type 1 aged 21 to 50 years, insulin glulisine
displayed dose-proportional glucose lowering effect in the therapeutic relevant dose range 0.075 to
0.15 Units/kg,
and less than proportional increase in glucose lowering effect with 0.3 Units/kg or
higher, like human insulin.
Insulin glulisine takes effect about twice as fast as regular human insulin and completes the glucose
lowering effect about 2 hours earlier than regular human insulin.
Obesity
A phase I study carried out with insulin glulisine, lispro and regular human insulin in an obese
population has demonstrated that insulin glulisine maintains its rapid-acting properties. In this study,
the time to 20 % of total AUC and the AUC (0-2h) representing the early glucose lowering activity
were respectively of 114 minutes and 427 mg/kg
for insulin glulisine, 121 minutes and 354 mg/kg for
lispro, 150 minutes and 197 mg/kg for regular human insulin (see figure 2).
Figure 2: Glucose infusion rates (GIR) after subcutaneous injection of 0.3 Units/kg of insulin glulisine
(GLULISINE) or insulin lispro (LISPRO) or regular human insulin (REGULAR) in an obese
population.
Another phase I study with insulin glulisine and insulin lispro in a non-diabetic population in
80 subjects with a wide range of body mass indices (18-46 kg/m²) has demonstrated that rapid action
is generally maintained across a wide range of body mass indices (BMI), while total glucose lowering
effect decreases with increasing obesity.
The average total GIR AUC between 0–1 hour was 102±75 mg/kg and 158±100 mg/kg with 0.2 and
0.4 Units/kg insulin glulisine,
respectively, and was 83.1±72.8 mg/kg and 112.3±70.8 mg/kg with 0.2
and 0.4 Units/kg
insulin
lispro respectively.
A phase I study in 18 obese patients with type 2 diabetes mellitus (BMI between 35 and 40 kg/m
2
)
with insulin glulisine and insulin lispro [90% CI:0.81, 0.95 (p=<0.01)]has shown that insulin glulisine
effectively controls diurnal post-prandial blood glucose excursions.
Type 1 diabetes mellitus-Adults
In a 26-week phase III clinical study comparing insulin glulisine with insulin lispro both injected
subcutaneously shortly (0-15 minutes) before a meal in patients with type 1 diabetes mellitus using
insulin glargine as basal insulin, insulin glulisine was comparable to insulin lispro for glycemic control
as reflected by changes in glycated haemoglobin (expressed as HbA
1c
equivalent) from baseline to
endpoint. Comparable self-monitored blood glucose values were observed. No increase in the basal
insulin dose was needed with insulin glulisine, in contrast to insulin lispro.
A 12-week phase III clinical study performed in patients with type 1 diabetes mellitus receiving
insulin glargine as basal therapy indicate that the immediate postmeal administration of insulin
glulisine provides efficacy that was comparable to immediate premeal insulin glulisine (0-15 minutes)
or regular insulin (30-45 minutes).



































































































In the per protocol population there was a significantly larger observed reduction in GHb in the
premeal glulisine group compared with the regular insulin group.
Type 1 diabetes mellitus-Paediatric
A 26-week phase III clinical study compared insulin glulisine with insulin lispro both injected
subcutaneously shortly (0-15 minutes) before a meal in children (4-5 years: n=9; 6-7 years: n=32 and
8-11 years: n=149) and adolescents (12-17 years: n=382) with type 1 diabetes mellitus using insulin
glargine or NPH as basal insulin. Insulin glulisine was comparable to insulin lispro for glycaemic
control as reflected by changes in glycated haemoglobin (GHb expressed as HbA
1c
equivalent) from
baseline to endpoint and by self-monitored blood glucose values.
There is insufficient clinical information on the use of Apidra in children younger than the age of
6 years.
Type 2 diabetes mellitus-Adults
A 26-week phase III clinical study followed by a 26-week extension safety study was conducted to
compare insulin glulisine (0-15 minutes before a meal) with regular human insulin (30-45 minutes
before a meal) injected subcutaneously in patients with type 2 diabetes mellitus also using NPH
insulin as basal insulin. The average body mass index (BMI) of patients was 34.55 kg/m
2
. Insulin
glulisine was shown to be comparable to regular human insulin with regard to glycated haemoglobin
(expressed as HbA
1c
equivalent) changes from baseline to the 6-month endpoint (-0.46% for insulin
glulisine and -0.30% for regular human insulin, p=0.0029) and from baseline to the 12-month endpoint
(-0.23% for insulin glulisine and -0.13% for regular human insulin, difference not significant). In this
study, the majority of patients (79%) mixed their short acting insulin with NPH insulin immediately
prior to injection and 58% of subjects used oral hypoglycemic agents at randomization and were
instructed to continue to use them at the same dose.
Race and gender
In controlled clinical trials in adults, insulin glulisine did not show differences in safety and efficacy in
subgroup analyses based on race and gender.
5.2 Pharmacokinetic properties
In insulin glulisine the replacement of the human insulin amino acid asparagine in position B3 by
lysine and the lysine in position B29 by glutamic acid favors more rapid absorption.
In a study with 18 male subjects with diabetes mellitus type 1 aged 21 to 50 years, insulin glulisine
displays dose-proportionality for early, maximum and total exposure in the dose range 0.075 to
0.4 Units/kg
.
Absorption and bioavailability
Pharmacokinetic profiles in healthy volunteers and diabetes patients (type 1 or 2) demonstrated that
absorption of insulin glulisine was about twice as fast with a peak concentration approximately twice
as high as compared to regular human insulin.
In a study in patients with type 1 diabetes mellitus after subcutaneous administration of 0.15 Units/kg,
for insulin glulisine the T
max
was 55 minutes and C
max
was 82 ± 1.3 µUnits/ml compared to a T
max
of
82 minutes and a C
max
of 46 ± 1.3 µUnits/ml for regular human insulin. The mean residence time of
insulin glulisine was shorter (98 min) than for regular human insulin (161 min) (see figure 3).
Figure 3: Pharmacokinetic profile of insulin glulisine and regular human insulin in type 1 diabetes
mellitus patients after a dose of 0.15 Units/kg.
In a study in patients with type 2 diabetes mellitus after subcutaneous administration of 0.2 Units/kg
insulin glulisine, the C
max
was 91 µUnits/ml with the interquartile range from 78 to 104 µUnits/ml.
When insulin glulisine was injected subcutaneously into abdomen, deltoid and thigh, the
concentration-time profiles were similar with a slightly faster absorption when administered in the
abdomen compared to the thigh. Absorption from deltoid sites was in-between (see section 4.2). The
absolute bioavailability (70%) of insulin glulisine was similar between injection sites and of low
intra-subject variability (11%CV). Intravenous bolus administration of insulin glulisine resulted in a
higher systemic exposure when compared to subcutaneous injection, with a C
max
approximately
40-fold higher.
Obesity
Another phase I study with insulin glulisine and insulin lispro in a non-diabetic population in
80 subjects with a wide range of body mass indices (18-46 kg/m²) has demonstrated that rapid
absorption and total exposure is generally maintained across a wide range of body mass indices.
The time to 10% of total INS exposure was reached earlier by approximately 5–6 min with insulin
glulisine.

Distribution and elimination
The distribution and elimination of insulin glulisine and regular human insulin after intravenous
administration is similar with volumes of distribution of 13 l and 22 l and half-lives of 13 and
18 minutes, respectively.
After subcutaneous administration, insulin glulisine is eliminated more rapidly than regular human
insulin with an apparent half-life of 42 minutes compared to 86 minutes. In an across study analysis of
insulin glulisine in either healthy subjects or subjects with type 1 or type 2 diabetes mellitus the
apparent half-life ranged from 37 to 75 minutes (interquartile range).
Insulin glulisine shows low plasma protein binding, similar to human insulin.
Renal impairment
In a clinical study performed in non-diabetic subjects covering a wide range of renal function
(CrCl > 80 ml/min, 30-50 ml/min, < 30 ml/min), the rapid-acting properties of insulin glulisine were
generally maintained. However, insulin requirements may be reduced in the presence of renal
impairment.
Hepatic impairment
The pharmacokinetic properties have not been investigated in patients with impaired liver function.
Elderly
Very limited pharmacokinetic data are available for elderly patients with diabetes mellitus.
Children and adolescents
The pharmacokinetic and pharmacodynamic properties of insulin glulisine were investigated in
children (7-11 years) and adolescents (12-16 years) with type 1 diabetes mellitus. Insulin glulisine was
rapidly absorbed in both age groups, with similar T
max
and C
max
as in adults (see section 4.2).
Administered immediately before a test meal, insulin glulisine provided better postprandial control
than regular human insulin, as in adults (see section 5.1). The glucose excursion (AUC
0-6h
) was
641 mg.h.dl
-1
for insulin glulisine and 801 mg.h.dl
-1
for regular human insulin.
5.3 Preclinical safety data
Non-clinical data did not reveal toxicity findings others than those linked to the blood glucose
lowering pharmacodynamic activity (hypoglycemia), different from regular human insulin or of
clinical relevance for humans.
PHARMACEUTICAL PARTICULARS
Metacresol
Sodium chloride
Trometamol
Polysorbate 20
Hydrochloric acid, concentrated
Sodium hydroxide
Water for injections
In the absence of compatibility studies this medicinal product must not be mixed with other medicinal
products except NPH human insulin.
Shelf life after first use of the cartridge
The product may be stored for a maximum of 4 weeks below 25°C away from direct heat or direct
light.
The pen containing a cartridge must not be stored in the refrigerator.
The pen cap must be put back on the pen after each injection in order to protect from light.
6.4 Special precautions for storage
Store in a refrigerator (2°C - 8°C).
Do not freeze.
Do not put Apidra next to the freezer compartment or a freezer pack.
Keep the cartridge in the outer carton in order to protect from light.
For storage conditions, see section 6.3.
6.5 Nature and contents of container
3 ml solution in a cartridge (type I colourless glass) with a plunger (elastomeric bromobutyl rubber)
and a flanged cap (aluminium) with a stopper (elastomeric bromobutyl rubber). Packs of 1, 3, 4, 5, 6,
8, 9 and 10 cartridges are available.
Not all pack sizes may be marketed.
6.6 Special precautions for disposal and other handling
The Apidra cartridges are to be used only in conjunction with OptiPen, ClikSTAR, Autopen 24 or
Tactipen (see section 4.4). Not all of these pens may be marketed in your country.
The pen should be used as recommended in the information provided by the device manufacturer.
The manufacturer’s instructions for using the pen must be followed carefully for loading the cartridge,
attaching the needle, and administering the insulin injection. Inspect the cartridge before use. It must
only be used if the solution is clear, colourless, with no solid particles visible. Before insertion of the
cartridge into the reusable pen, the cartridge must be stored at room temperature for 1 to 2 hours. Air
bubbles must be removed from the cartridge before injection (see instruction for using pen). Empty
cartridges must not be refilled.
If the pen malfunctions (see instructions for using the pen), the solution may be drawn from the
cartridge into a syringe (suitable for an insulin with 100 Units/ml) and injected. If the insulin pen is
damaged or not working properly (due to mechanical defects) it has to be discarded, and a new insulin
pen has to be used.
To prevent any kind of contamination, the re-usable pen should be used by a single patient only.
Insulin label must always be checked before each injection to avoid medication errors between insulin
glulisine and other insulins (see section 4.4).
When mixed with NPH human insulin, Apidra should be drawn into the syringe first. Injection should
be given immediately after mixing as no data are available regarding the mixtures made up a
significant time before injection.
MARKETING AUTHORISATION HOLDER
Sanofi-Aventis Deutschland GmbH
D-65926 Frankfurt am Main
Germany.
MARKETING AUTHORISATION NUMBER(S)
DATE OF FIRST AUTHORISATION/RENEWAL OF THE AUTHORISATION
Date of first authorisation: 27 September 2004
Date of latest renewal: 20 August 2009
10. DATE OF REVISION OF THE TEXT
Detailed information on this product is available on the website of the European Medicines Agency
http://www.ema.europa.eu/
NAME OF THE MEDICINAL PRODUCT
Apidra 100 Units/ml solution for injection in a cartridge for OptiClik
QUALITATIVE AND QUANTITATIVE COMPOSITION
Each ml contains 100 Units insulin glulisine (equivalent to 3.49 mg).
Each cartridge contains 3 ml of solution for injection, equivalent to 300 Units.
Insulin glulisine is produced by recombinant DNA technology in
Escherichia coli
.
For a full list of excipients, see section 6.1.
Solution for injection in a cartridge for OptiClik.
Clear, colourless, aqueous solution.
4.1 Therapeutic indications
Treatment of adults, adolescents and children, 6 years or older with diabetes mellitus, where treatment
with insulin is required.
4.2 Posology and method of administration
The potency of this preparation is stated in units. These units are exclusive to Apidra and are not the
same as IU or the units used to express the potency of other insulin analogues (see section 5.1).
Apidra should be used in regimens that include an intermediate or long acting insulin or basal insulin
analogue and can be used with oral hypoglycaemic agents.
The dose of Apidra should be individually adjusted.
Renal impairment
The pharmacokinetic properties of insulin glulisine are generally maintained in patients with renal
impairment. However, insulin requirements may be reduced in the presence of renal impairment (see
section 5.2).
Hepatic impairment
The pharmacokinetic properties of insulin glulisine have not been investigated in patients with
decreased liver function. In patients with hepatic impairment, insulin requirements may be diminished
due to reduced capacity for gluconeogenesis and reduced insulin metabolism.
Elderly
Limited pharmacokinetic data are available in elderly patients with diabetes mellitus. Deterioration of
renal function may lead to a decrease in insulin requirements.
Paediatric population
There is insufficient clinical information on the use of Apidra in children younger than the age of
6 years.
Apidra should be given by subcutaneous injection shortly (0-15 min) before or soon after meals or by
continuous subcutaneous pump infusion.
Apidra should be administered subcutaneously in the abdominal wall, thigh or deltoid or by
continuous infusion in the abdominal wall. Injection sites and infusion sites within an injection area
(abdomen, thigh or deltoid) should be rotated from one injection to the next. The rate of absorption,
and consequently the onset and duration of action, may be affected by the injection site, exercise and
other variables. Subcutaneous injection in the abdominal wall ensures a slightly faster absorption than
other injection sites (see section 5.2).
Care should be taken to ensure that a blood vessel has not been entered. After injection, the site of
injection should not be massaged. Patients must be educated to use proper injection techniques.
Mixing with insulins
When administered as a subcutaneous injection, Apidra must not be mixed with other medicinal
products except NPH human insulin.
For further details on handling, see section 6.6.
Hypersensitivity to the active substance or to any of the excipients.
4.4 Special warnings and precautions for use
Transferring a patient to another type or brand of insulin should be done under strict medical
supervision. Changes in strength, brand (manufacturer), type (regular, neutral protamine Hagedorn
[NPH], lente, long-acting, etc.), origin (animal, human, human insulin analogue) and/or method of
manufacture may result in the need for a change in dose. Concomitant oral antidiabetic treatment may
need to be adjusted.
The use of inadequate doses or discontinuation of treatment, especially in insulin-dependent diabetic,
may lead to hyperglycaemia and diabetic ketoacidosis; conditions which are potentially lethal.
The time of occurrence of hypoglycaemia depends on the action profile of the insulins used and may,
therefore, change when the treatment regimen is changed.
Conditions which may make the early warning symptoms of hypoglycaemia different or less
pronounced include long duration of diabetes, intensified insulin therapy, diabetic nerve disease,
medicinal products such as beta blockers or after transfer from animal-source insulin to human insulin.
Adjustment of dose may be also necessary if patients undertake increased physical activity or change
their usual meal plan. Exercise taken immediately after a meal may increase the risk of
hypoglycaemia.
When compared with soluble human insulin, if hypoglycaemia occurs after an injection with rapid
acting analogues, it may occur earlier.
Uncorrected hypoglycaemic or hyperglycaemic reactions can cause loss of consciousness, coma, or
death.
Insulin requirements may be altered during illness or emotional disturbances.
Medication errors have been reported in which other insulins, particularly long-acting insulins, have
been accidentally administered instead of insulin glulisine. Insulin label must always be checked
before each injection to avoid medication errors between insulin glulisine and other insulins.
This medicinal product contains less than 1 mmol (23 mg) sodium per dose, i.e. it is essentially
‘sodium-free’.
Apidra contains metacresol, which may cause allergic reactions.
Combination of Apidra with pioglitazone
Cases of cardiac failure have been reported when pioglitazone was used in combination with insulin,
especially in patients with risk factors for development of cardiac heart failure. This should be kept in
mind if treatment with the combination of pioglitazone and Apidra is considered. If the combination is
used, patients should be observed for signs and symptoms of heart failure, weight gain and oedema.
Pioglitazone should be discontinued if any deterioration in cardiac symptoms occurs.
4.5 Interaction with other medicinal products and other forms of interaction
Studies on pharmacokinetic interactions have not been performed. Based on empirical knowledge
from similar medicinal products, clinically relevant pharmacokinetic interactions are unlikely to occur.
A number of substances affect glucose metabolism and may require dose adjustment of insulin
glulisine and particularly close monitoring.
Substances that may enhance the blood-glucose-lowering activity and increase susceptibility to
hypoglycaemia include oral antidiabetic agents, angiotensin converting enzyme (ACE) inhibitors,
disopyramide, fibrates, fluoxetine, monoamine oxidase inhibitors (MAOIs), pentoxifylline,
propoxyphene, salicylates and sulfonamide antibiotics.
Substances that may reduce the blood-glucose-lowering activity include corticosteroids, danazol,
diazoxide, diuretics, glucagon, isoniazid, phenothiazine derivatives, somatropin, sympathomimetic
agents (e.g. epinephrine [adrenaline], salbutamol, terbutaline), thyroid hormones, estrogens, progestins
(e.g. in oral contraceptives), protease inhibitors and atypical antipsychotic medicinal products (e.g.
olanzapine and clozapine).
Beta-blockers, clonidine, lithium salts or alcohol may either potentiate or weaken the
blood-glucose-lowering activity of insulin. Pentamidine may cause hypoglycaemia, which may
sometimes be followed by hyperglycaemia.
In addition, under the influence of sympatholytic medicinal products such as beta-blockers, clonidine,
guanethidine and reserpine, the signs of adrenergic counter-regulation may be reduced or absent.
4.6 Pregnancy and lactation
There are no adequate data on the use of insulin glulisine in pregnant women.
Animal reproduction studies have not revealed any differences between insulin glulisine and human
insulin regarding pregnancy, embryonal/foetal development, parturition or postnatal development (see
section 5.3).
Caution should be exercised when prescribing to pregnant women. Careful monitoring of glucose
control is essential.
It is essential for patients with pre-existing or gestational diabetes to maintain good metabolic control
throughout pregnancy. Insulin requirements may decrease during the first trimester and generally
increase during the second and third trimesters. Immediately after delivery, insulin requirements
decline rapidly.
It is unknown whether insulin glulisine is excreted in human milk, but in general insulin does not pass
into breast milk and is not absorbed after oral administration.
Breast-feeding mothers may require adjustments in insulin dose and diet.
4.7 Effects on ability to drive and use machines
The patient's ability to concentrate and react may be impaired as a result of hypoglycaemia or
hyperglycaemia or, for example, as a result of visual impairment. This may constitute a risk in
situations where these abilities are of special importance (e.g. driving a car or operating machines).
Patients should be advised to take precautions to avoid hypoglycaemia whilst driving. This is
particularly important in those who have reduced or absent awareness of the warning symptoms of
hypoglycaemia or have frequent episodes of hypoglycaemia. The advisability of driving should be
considered in these circumstances.
Hypoglycaemia, the most frequent undesirable effect of insulin therapy, may occur if the insulin dose
is too high in relation to the insulin requirement.
The following related adverse reactions from clinical studies were listed below by system organ class
and in order of decreasing incidence (very common: 1/10; common: 1/100 to <1/10; uncommon:
1/1,000 to <1/100; rare: 1/10,000 to <1/1,000; very rare: <1/10,000), not known (cannot be
estimated from the available data).
Within each frequency grouping, undesirable effects are presented in order of decreasing seriousness.
MedDRA
Organ system
classes
Metabolism
and nutrition
disorders
Skin and
subcutaneous
tissue
disorders
Injection site
reactions
Local
hypersensitivity
reactions
General
disorders and
administration
site conditions
Systemic
hypersensitivity
reactions
Metabolism and nutrition disorders
Symptoms of hypoglycaemia usually occur suddenly. They may include cold sweats, cool pale skin,
fatigue, nervousness or tremor, anxiousness, unusual tiredness or weakness, confusion, difficulty in
concentration, drowsiness, excessive hunger, vision changes, headache, nausea and palpitation.
Hypoglycaemia can become severe and may lead to unconsciousness and/or convulsions and may
result in temporary or permanent impairment of brain function or even death.
Skin and subcutaneous tissue disorders
Local hypersensitivity reactions (redness, swelling and itching at the injection site) may occur during
treatment with insulin. These reactions are usually transitory and normally they disappear during
continued treatment.
Lipodystrophy may occur at the injection site as a consequence of failure to rotate injection sites
within an area.
General disorders and administration site conditions
Systemic hypersensitivity reactions may include urticaria, chest tightness, dyspnea, allergic dermatitis
and pruritus. Severe cases of generalized allergy, including anaphylactic reaction, may be
life-threatening.
Hypoglycaemia may occur as a result of an excess of insulin activity relative to food intake and energy
expenditure.
There are no specific data available concerning overdoses with insulin glulisine. However,
hypoglycaemia may develop over sequential stages:
Mild hypoglycaemic episodes can be treated by oral administration of glucose or sugary products. It is
therefore recommended that the diabetic patient constantly carries some sugar lumps, sweets, biscuits
or sugary fruit juice.
Severe hypoglycaemic episodes, where the patient has become unconscious, can be treated by
glucagon (0.5 mg to 1 mg) given intramuscularly or subcutaneously by a person who has received
appropriate instruction, or by glucose given intravenously by a healthcare professional. Glucose must
also be given intravenously, if the patient does not respond to glucagon within 10 to 15 minutes.











Upon regaining consciousness, administration of oral carbohydrate is recommended for the patient in
order to prevent relapse.
After an injection of glucagon, the patient should be monitored in a hospital in order to find the reason
for this severe hypoglycaemia and prevent other similar episodes.
PHARMACOLOGICAL PROPERTIES
5.1 Pharmacodynamic properties
Pharmacotherapeutic group: Insulins and analogues for injection, fast-acting. ATC code: A10AB06
Insulin glulisine is a recombinant human insulin analogue that is equipotent to regular human insulin.
Insulin glulisine has a more rapid onset of action and a shorter duration of action than regular human
insulin.
The primary activity of insulins and insulin analogues, including insulin glulisine, is regulation of
glucose metabolism. Insulins lower blood glucose levels by stimulating peripheral glucose uptake,
especially by skeletal muscle and fat, and by inhibiting hepatic glucose production. Insulin inhibits
lipolysis in the adipocyte, inhibits proteolysis and enhances protein synthesis.
Studies in healthy volunteers and patients with diabetes demonstrated that insulin glulisine is more
rapid in onset of action and of shorter duration of action than regular human insulin when given
subcutaneously. When insulin glulisine is injected subcutaneously, the glucose lowering activity will
begin within 10 – 20 minutes. The glucose-lowering activities of insulin glulisine and regular human
insulin are equipotent when administered by intravenous route. One unit of insulin glulisine has the
same glucose - lowering activity as one unit of regular human insulin.
Dose proportionality
In a study with 18 male subjects with diabetes mellitus type 1 aged 21 to 50 years, insulin glulisine
displayed dose-proportional glucose lowering effect in the therapeutic relevant dose range 0.075 to
0.15 Units/kg,
and less than proportional increase in glucose lowering effect with 0.3 Units/kg or
higher, like human insulin.
Insulin glulisine takes effect about twice as fast as regular human insulin and completes the glucose
lowering effect about 2 hours earlier than regular human insulin.
A phase I study carried out with insulin glulisine, lispro and regular human insulin in an obese
population has demonstrated that insulin glulisine maintains its rapid-acting properties. In this study,
the time to 20 % of total AUC and the AUC (0-2h) representing the early glucose lowering activity
were respectively of 114 minutes and 427 mg/kg
for insulin glulisine, 121 minutes and 354 mg/kg for
lispro, 150 minutes and 197 mg/kg for regular human insulin (see figure 2).
Figure 2: Glucose infusion rates (GIR) after subcutaneous injection of 0.3 Units/kg of insulin glulisine
(GLULISINE) or insulin lispro (LISPRO) or regular human insulin (REGULAR) in an obese
population.
Another phase I study with insulin glulisine and insulin lispro in a non-diabetic population in 80
subjects with a wide range of body mass indices (18-46 kg/m²) has demonstrated that rapid action is
generally maintained across a wide range of body mass indices (BMI), while total glucose lowering
effect decreases with increasing obesity.
The average total GIR AUC between 0–1 hour was 102±75 mg/kg and 158±100 mg/kg with 0.2 and
0.4 Units/kg
insulin glulisine,
respectively, and was 83.1±72.8 mg/kg and 112.3±70.8 mg/kg with 0.2
and 0.4 Units/kg
insulin
lispro respectively.
A phase I study in 18 obese patients with type 2 diabetes mellitus (BMI between 35 and 40 kg/m
2
)
with insulin glulisine and insulin lispro [90% CI:0.81, 0.95 (p=<0.01)]has shown that insulin glulisine
effectively controls diurnal post-prandial blood glucose excursions.
Type 1 diabetes mellitus-Adults
In a 26-week phase III clinical study comparing insulin glulisine with insulin lispro both injected
subcutaneously shortly (0-15 minutes) before a meal in patients with type 1 diabetes mellitus using
insulin glargine as basal insulin, insulin glulisine was comparable to insulin lispro for glycemic control
as reflected by changes in glycated haemoglobin (expressed as HbA
1c
equivalent) from baseline to
endpoint. Comparable self-monitored blood glucose values were observed. No increase in the basal
insulin dose was needed with insulin glulisine, in contrast to insulin lispro.
A 12-week phase III clinical study performed in patients with type 1 diabetes mellitus receiving
insulin glargine as basal therapy indicate that the immediate postmeal administration of insulin
glulisine provides efficacy that was comparable to immediate premeal insulin glulisine (0-15 minutes)
or regular insulin (30-45 minutes).




































































































In the per protocol population there was a significantly larger observed reduction in GHb in the
premeal glulisine group compared with the regular insulin group.
Type 1 diabetes mellitus-Paediatric
A 26-week phase III clinical study compared insulin glulisine with insulin lispro both injected
subcutaneously shortly (0-15 minutes) before a meal in children (4-5 years: n=9; 6-7 years: n=32 and
8-11 years: n=149) and adolescents (12-17 years: n=382) with type 1 diabetes mellitus using insulin
glargine or NPH as basal insulin. Insulin glulisine was comparable to insulin lispro for glycaemic
control as reflected by changes in glycated haemoglobin (GHb expressed as HbA
1c
equivalent) from
baseline to endpoint and by self-monitored blood glucose values.
There is insufficient clinical information on the use of Apidra in children younger than the age of
6 years.
Type 2 diabetes mellitus-Adults
A 26-week phase III clinical study followed by a 26-week extension safety study was conducted to
compare insulin glulisine (0-15 minutes before a meal) with regular human insulin (30-45 minutes
before a meal) injected subcutaneously in patients with type 2 diabetes mellitus also using NPH
insulin as basal insulin. The average body mass index (BMI) of patients was 34.55 kg/m
2
. Insulin
glulisine was shown to be comparable to regular human insulin with regard to glycated haemoglobin
(expressed as HbA
1c
equivalent) changes from baseline to the 6-month endpoint (-0.46% for insulin
glulisine and -0.30% for regular human insulin, p=0.0029) and from baseline to the 12-month endpoint
(-0.23% for insulin glulisine and -0.13% for regular human insulin, difference not significant). In this
study, the majority of patients (79%) mixed their short acting insulin with NPH insulin immediately
prior to injection and 58% of subjects used oral hypoglycemic agents at randomization and were
instructed to continue to use them at the same dose.
Race and gender
In controlled clinical trials in adults, insulin glulisine did not show differences in safety and efficacy in
subgroup analyses based on race and gender.
5.2 Pharmacokinetic properties
In insulin glulisine the replacement of the human insulin amino acid asparagine in position B3 by
lysine and the lysine in position B29 by glutamic acid favors more rapid absorption.
In a study with 18 male subjects with diabetes mellitus type 1, aged 21 to 50 years, insulin glulisine
displays dose-proportionality for early, maximum and total exposure in the dose range 0.075 to
0.4 Units/kg
.
Absorption and bioavailability
Pharmacokinetic profiles in healthy volunteers and diabetes patients (type 1 or 2) demonstrated that
absorption of insulin glulisine was about twice as fast with a peak concentration approximately twice
as high as compared to regular human insulin.
In a study in patients with type 1 diabetes mellitus after subcutaneous administration of 0.15 Units/kg,
for insulin glulisine the T
max
was 55 minutes and C
max
was 82 ± 1.3 µUnits/ml compared to a T
max
of
82 minutes and a C
max
of 46 ± 1.3 µUnits/ml for regular human insulin. The mean residence time of
insulin glulisine was shorter (98 min) than for regular human insulin (161 min) (see figure 3).
Figure 3: Pharmacokinetic profile of insulin glulisine and regular human insulin in type 1 diabetes
mellitus patients after a dose of 0.15 Units/kg.
In a study in patients with type 2 diabetes mellitus after subcutaneous administration of 0.2 Units/kg
insulin glulisine, the C
max
was 91 µUnits/ml with the interquartile range from 78 to 104 µUnits/ml.
When insulin glulisine was injected subcutaneously into abdomen, deltoid and thigh, the
concentration-time profiles were similar with a slightly faster absorption when administered in the
abdomen compared to the thigh. Absorption from deltoid sites was in-between (see section 4.2). The
absolute bioavailability (70%) of insulin glulisine was similar between injection sites and of low
intra-subject variability (11%CV). Intravenous bolus administration of insulin glulisine resulted in a
higher systemic exposure when compared to subcutaneous injection, with a C
max
approximately
40-fold higher.
Another phase I study with insulin glulisine and insulin lispro in a non-diabetic population in
80 subjects with a wide range of body mass indices (18-46 kg/m²) has demonstrated that rapid
absorption and total exposure is generally maintained across a wide range of body mass indices.
The time to 10% of total INS exposure was reached earlier by approximately 5–6 min with insulin
glulisine.

Distribution and elimination
The distribution and elimination of insulin glulisine and regular human insulin after intravenous
administration is similar with volumes of distribution of 13 l and 22 l and half-lives of 13 and
18 minutes, respectively.
After subcutaneous administration, insulin glulisine is eliminated more rapidly than regular human
insulin with an apparent half-life of 42 minutes compared to 86 minutes. In an across study analysis of
insulin glulisine in either healthy subjects or subjects with type 1 or type 2 diabetes mellitus the
apparent half-life ranged from 37 to 75 minutes (interquartile range).
Insulin glulisine shows low plasma protein binding, similar to human insulin.
Renal impairment
In a clinical study performed in non-diabetic subjects covering a wide range of renal function
(CrCl > 80 ml/min, 30-50 ml/min, < 30 ml/min), the rapid-acting properties of insulin glulisine were
generally maintained. However, insulin requirements may be reduced in the presence of renal
impairment.
Hepatic impairment
The pharmacokinetic properties have not been investigated in patients with impaired liver function.
Elderly
Very limited pharmacokinetic data are available for elderly patients with diabetes mellitus.
Children and adolescents
The pharmacokinetic and pharmacodynamic properties of insulin glulisine were investigated in
children (7-11 years) and adolescents (12-16 years) with type 1 diabetes mellitus. Insulin glulisine was
rapidly absorbed in both age groups, with similar T
max
and C
max
as in adults (see section 4.2).
Administered immediately before a test meal, insulin glulisine provided better postprandial control
than regular human insulin, as in adults (see section 5.1). The glucose excursion (AUC
0-6h
) was
641 mg.h.dl
-1
for insulin glulisine and 801 mg.h.dl
-1
for regular human insulin.
5.3 Preclinical safety data
Non-clinical data did not reveal toxicity findings others than those linked to the blood glucose
lowering pharmacodynamic activity (hypoglycemia), different from regular human insulin or of
clinical relevance for humans.
PHARMACEUTICAL PARTICULARS
Metacresol
Sodium chloride
Trometamol
Polysorbate 20
Hydrochloric acid, concentrated
Sodium hydroxide
Water for injections
In the absence of compatibility studies, this medicinal product must not be mixed with other medicinal
products except NPH human insulin.
Shelf life after first use of the cartridge
The product may be stored for a maximum of 4 weeks below 25°C away from direct heat or direct
light. The pen containing the cartridge must not be stored in the refrigerator. The pen cap must be put
back on the pen after each injection in order to protect from light.
6.4 Special precautions for storage
Store in a refrigerator (2°C - 8°C).
Do not freeze.
Do not put Apidra next to the freezer compartment or a freezer pack..
Keep the cartridge in the outer carton in order to protect from light.
For storage conditions, see section 6.3.
6.5 Nature and contents of container
3 ml solution in a cartridge (type I colourless glass cartridge) with a plunger (elastomeric bromobutyl
rubber) and a flanged cap (aluminium) with a stopper (elastomeric bromobutyl rubber).
The glass cartridge is irreversibly integrated in a transparent container and attached to a plastic
mechanism by a threaded rod at one extremity.
Packs of 1, 3, 4, 5, 6, 8, 9 and 10 cartridges for OptiClik are available. Not all pack sizes may be
marketed.
6.6 Special precautions for disposal and other handling
The cartridges for OptiClik are to be used in conjunction with OptiClik only, and as recommended in
the information provided by the device manufacturer.
The manufacturer’s instructions for using the pen must be followed carefully for loading the cartridge,
attaching the needle, and administering the insulin injection.
If OptiClik is damaged or not working properly (due to mechanical defects) it has to be discarded, and
a new OptiClik has to be used.
Before insertion of the cartridge into the reusable pen, the cartridge must be stored at room
temperature for 1 to 2 hours. Inspect the cartridge before use. It must only be used if the cartridge is
intact and the solution is clear, colourless, with no solid particles visible.
Air bubbles must be removed from the cartridge before injection (see instruction for using pen).
Empty cartridges must not be refilled.
If the pen malfunctions, (see instructions for using the pen) the solution may be drawn from the
cartridge into a syringe (suitable for an insulin with 100 Units/ml) and injected.
To prevent any kind of contamination, the re-usable pen should be used by a single patient only.
Insulin label must always be checked before each injection to avoid medication errors between insulin
glulisine and other insulins (see section 4.4).
When mixed with NPH human insulin, Apidra should be drawn into the syringe first. Injection should
be given immediately after mixing as no data are available regarding the mixtures made up a
significant time before injection.
MARKETING AUTHORISATION HOLDER
Sanofi-Aventis Deutschland GmbH
D-65926 Frankfurt am Main
Germany.
MARKETING AUTHORISATION NUMBER(S)
DATE OF FIRST AUTHORISATION/RENEWAL OF THE AUTHORISATION
Date of first authorisation: 27 September 2004
Date of latest renewal: 20 August 2009
10. DATE OF REVISION OF THE TEXT
Detailed information on this product is available on the website of the European Medicines Agency
http://www.ema.europa.eu/
The active substance is insulin glulisine. Each ml of the solution contains 100 Units of insulin
glulisine (equivalent to 3.49 mg).
The other ingredients are: metacresol, sodium chloride, trometamol, polysorbate 20,
concentrated hydrochloric acid, sodium hydroxide, water for injections
What Apidra looks like and contents of the pack
Apidra SoloStar 100 Units/ml, solution for injection in a pre-filled pen. It is a clear, colourless,
aqueous solution with no particles visible.
Each pen contains 3 ml solution, equivalent to 300 Units. Packs of 1, 3, 4, 5, 6, 8, 9 and 10 pre-filled
pens are available. Not all pack sizes may be marketed.
Marketing Authorisation Holder and Manufacturer
Marketing Authorisation Holder
:
Sanofi-Aventis Deutschland GmbH
D-65926 Frankfurt am Main
Germany.
Manufacturer:
Sanofi-Aventis Deutschland GmbH
Industriepark Höchst, D- 65926 Frankfurt
Germany
For any information about this medicine, please contact the local representative of the Marketing
Authorisation Holder.
België/Belgique/Belgien
sanofi-aventis Belgium
Tél/Tel: +32 (0)2 710 54 00
Luxembourg/Luxemburg
sanofi-aventis Belgium
Tél/Tel: +32 (0)2 710 54 00 (Belgique/Belgien)
България
sanofi-aventis Bulgaria EOOD
Тел.: +359 (0)2 970 53 00
Magyarország
sanofi-aventis zrt., Magyarország
Tel.: +36 1 505 0050
Česká republika
sanofi-aventis, s.r.o.
Tel: +420 233 086 111
Malta
sanofi-aventis Malta Ltd.
Tel: +356 21493022
Danmark
sanofi-aventis Denmark A/S
Tlf: +45 45 16 70 00
Nederland
sanofi-aventis Netherlands B.V.
Tel: +31 (0)182 557 755
Deutschland
Sanofi-Aventis Deutschland GmbH
Tel: +49 (0)180 2 222010
Norge
sanofi-aventis Norge AS
Tlf: +47 67 10 71 00
Eesti
sanofi-aventis Estonia OÜ
Tel: +372 627 34 88
Österreich
sanofi-aventis GmbH
Tel: +43 1 80 185 – 0
Ελλάδα
sanofi-aventis AEBE
Τηλ: +30 210 900 16 00
Polska
sanofi-aventis Sp. z o.o.
Tel.: +48 22 280 00 00
España
sanofi-aventis, S.A.
Tel: +34 93 485 94 00
Portugal
sanofi-aventis - Produtos Farmacêuticos, Lda.
Tel: +351 21 35 89 400
France
sanofi-aventis France
Tél: 0 800 222 555
Appel depuis l’étranger : +33 1 57 63 23 23
România
sanofi-aventis România S.R.L.
Tel: +40 (0) 21 317 31 36
Ireland
sanofi-aventis Ireland Ltd.
Tel: +353 (0) 1 403 56 00
Slovenija
sanofi-aventis d.o.o.
Tel: +386 1 560 48 00
Ísland
Vistor hf.
Sími: +354 535 7000
Slovenská republika
sanofi-aventis Pharma Slovakia s.r.o.
Tel: +421 2 33 100 100
Italia
sanofi-aventis S.p.A.
Tel: 800 13 12 12 (domande di tipo tecnico)
+39 02 393 91 (altre domande e chiamate
dall'estero)
Suomi/Finland
sanofi-aventis Oy
Puh/Tel: +358 (0) 201 200 300
Κύπρος
sanofi-aventis Cyprus Ltd.
Τηλ: +357 22 871600
Sverige
sanofi-aventis AB
Tel: +46 (0)8 634 50 00
Latvija
sanofi-aventis Latvia SIA
Tel: +371 67 33 24 51
United Kingdom
sanofi-aventis
Tel: +44 (0) 1483 505 515
Lietuva
UAB sanofi-aventis Lietuva
Tel: +370 5 2755224
This leaflet was last approved in
Detailed information on this medicine is available on the European Medicines Agency web site:
http://www.ema.europa.eu/
HYPERGLYCAEMIA AND HYPOGLYCAEMIA
Always carry some sugar (at least 20 grams) with you.
Carry some information with you to show you are a person with diabetes.
HYPERGLYCAEMIA (high blood sugar levels)
If your blood sugar is too high (hyperglycaemia), you may not have injected enough insulin.
Why does hyperglycaemia occur?
Examples include:
-
you have not injected your insulin or not injected enough, or if it has become less effective, for
example through incorrect storage,
you are doing less exercise than usual, you are under stress (emotional distress, excitement), or
you have an injury, operation, infection or fever,
you are taking or have taken certain other medicines (see section 2, "Using other medicines").
Warning symptoms of hyperglycaemia
Thirst, increased need to urinate, tiredness, dry skin, reddening of the face, loss of appetite, low blood
pressure, fast heart beat and glucose and ketone bodies in urine. Stomach pain, fast and deep
breathing, sleepiness or even loss of consciousness may be signs of a serious condition (ketoacidosis)
resulting from lack of insulin.
What should you do if you experience hyperglycaemia?
Test your blood sugar level and your urine for ketones as soon as any of the above symptoms
occur
. Severe hyperglycaemia or ketoacidosis must always be treated by a doctor, normally in a
hospital.
HYPOGLYCAEMIA (low blood sugar levels)
If your blood sugar level falls too much you may become unconscious. Serious hypoglycaemia may
cause a heart attack or brain damage and may be life-threatening. You normally should be able to
recognise when your blood sugar is falling too much so that you can take the right actions.
Why does hypoglycaemia occurs?
Examples include:
-
you inject too much insulin,
you do not eat enough, or eat food containing less carbohydrate than normal (sugar and
substances similar to sugar are called carbohydrates; however, artificial sweeteners are NOT
you drink alcohol, particularly if you are not eating much,
you are doing more exercise than usual or a different type of physical activity,
you are recovering from an injury or operation or other stress,
you are recovering from an illness or from fever,
you are taking or have stopped taking certain other medicines (see section 2, "Using other
medicines").
you miss meals or delay them,
you lose carbohydrates due to vomiting or diarrhoea,



Hypoglycaemia is also more likely to occur if:
-
you have just begun insulin treatment or changed to another insulin preparation,
your blood sugar levels are almost normal or are unstable,
you change the area of skin where you inject insulin (for example from the thigh to the upper
arm),
you suffer from severe kidney or liver disease, or some other disease such as hypothyroidism.
Warning symptoms of hypoglycaemia
Examples of symptoms that tell you that your blood sugar level is falling too much or too fast :
sweating, clammy skin, anxiety, fast heart beat, high blood pressure, palpitations and irregular
heartbeat. These symptoms often develop before the symptoms of a low sugar level in the brain.
Examples of symptoms that indicate a low sugar level in the brain: headaches, intense hunger, nausea,
vomiting, tiredness, sleepiness, sleep disturbances, restlessness, aggressive behaviour, lapses in
concentration, impaired reactions, depression, confusion, speech disturbances (sometimes total loss of
speech), visual disorders, trembling, paralysis, tingling sensations (paraesthesia), numbness and
tingling sensations in the area of the mouth, dizziness, loss of self-control, inability to look after
yourself, convulsions and loss of consciousness.
The first symptoms which alert you to hypoglycaemia ("warning symptoms") may change, be weaker
or may be missing altogether if:
-
you have had diabetes for a long time,
you suffer from a certain type of nervous disease (diabetic autonomic neuropathy),
you have recently suffered hypoglycaemia (for example the day before) or if it develops slowly,
you have almost normal or, at least, greatly improved blood sugar levels,
you are taking or have taken certain other medicines (see section 2, "Using other medicines.)
In such a case, you may develop severe hypoglycaemia (and even faint) before you are aware of the
problem. Be familiar with your warning symptoms. If necessary, more frequent blood sugar testing
can help to identify mild hypoglycaemic episodes that may otherwise be overlooked. If you are not
confident about recognising your warning symptoms, avoid situations (such as driving a car) in which
you or others would be put at risk by hypoglycaemia.
What should you do if you experience hypoglycaemia?
Do not inject insulin. Immediately take about 10 to 20 g sugar, such as glucose, sugar cubes or a
sugar-sweetened beverage. Caution: Artificial sweeteners and foods with artificial sweeteners
(such as diet drinks) are of no help in treating hypoglycaemia.
Then eat something that has a long-acting effect in raising your blood sugar (such as bread or
pasta). Your doctor or nurse should have discussed this with you previously.
If the hypoglycaemia comes back again take another 10 to 20 g sugar.
Speak to a doctor immediately if you are not able to control the hypoglycaemia or if it recurs.
Tell your relatives, friends and close colleagues the following:
If you are not able to swallow or if you are unconscious, you will require an injection of glucose or
glucagon (a medicine which increases blood sugar). These injections are justified even if it is not
certain that you have hypoglycaemia.
It is advisable to test your blood sugar immediately after taking glucose to check that you really have
hypoglycaemia.



Apidra SoloStar solution for injection in a pre-filled pen. INSTRUCTIONS FOR USE
SoloStar is a prefilled pen for the injection of insulin. Your healthcare professional has decided that
SoloStar is right for you. Talk with your healthcare professional about proper injection technique
before using SoloStar.
Read these instructions carefully before using your SoloStar. If you are not able to follow all the
instructions completely on your own, use SoloStar only if you have help from a person who is able to
follow the instructions. Hold the pen as shown in this leaflet. To ensure that you read the dose
correctly, hold the pen horizontally, with the needle on the left and the dosage selector to the right as
shown in the illustrations below.
You can set doses from 1 to 80 units in steps of 1 unit. Each pen contains multiple doses.
Keep this leaflet for future reference.
If you have any questions about SoloStar or about diabetes, ask your healthcare professional or call the
local sanofi-aventis number on the front of this leaflet.
Schematic diagram of the pen
Important information for use of SoloStar:
Always attach a new needle before each use. Only use needles that are compatible for use with
SoloStar.
Always perform the safety test before each injection (see Step 3).
This pen is only for your use. Do not share it with anyone else.
If your injection is given by another person, special caution must be taken by this person to
avoid accidental needle injury and transmission of infection.
Never use SoloStar if it is damaged or if you are not sure that it is working properly.
Always have a spare SoloStar in case your SoloStar is lost or damaged.
Step 1. Check the insulin
A
. Check the label on your SoloStar to make sure you have the correct insulin. The Apidra
SoloStar is blue. It has a dark blue injection button with a raised ring on the top.
B.
Take off the pen cap.
C.
Check the appearance of your insulin. Apidra is a clear insulin. Do not use this SoloStar if the
insulin is cloudy, coloured or has particles.
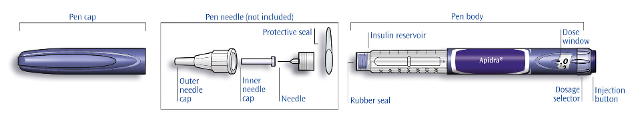
Step 2. Attach the needle
Always use a new sterile needle for each injection. This helps prevent contamination, and potential
needle blocks.
A
. Remove the protective seal from a new needle.
B.
Line up the needle with the pen, and keep it straight as you attach it (screw or push on,
depending on the needle type).
If the needle is not kept straight while you attach it, it can damage the rubber seal and cause
leakage, or damage the needle.
Step 3. Perform a Safety test
Always perform the safety test before each injection. This ensures that you get an accurate dose by:
ensuring that pen and needle work properly
A
. Select a dose of 2 units by turning the dosage selector.
B.
Take off the outer needle cap and keep it to remove the used needle after injection. Take off the
inner needle cap and discard it.
C.
Hold the pen with the needle pointing upwards.
D.
Tap the insulin reservoir so that any air bubbles rise up towards the needle.


E
. Press the injection button all the way in. Check if insulin comes out of the needle tip.
You may have to perform the safety test several times before insulin is seen.
If no insulin comes out, check for air bubbles and repeat the safety test two more times to
remove them.
If still no insulin comes out, the needle may be blocked. Change the needle and try again.
If no insulin comes out after changing the needle, your SoloStar may be damaged. Do not use
this SoloStar.
You can set the dose in steps of 1 unit, from a minimum of 1 unit to a maximum of 80 units. If you
need a dose greater than 80 units, you should give it as two or more injections.
A
. Check that the dose window shows “0” following the safety test.
B
. Select your required dose (in the example below, the selected dose is 30 units). If you turn past
your dose, you can turn back down.
Do not push the injection button while turning, as insulin will come out.
You cannot turn the dosage selector past the number of units left in the pen. Do not force the
dosage selector to turn. In this case, either you can inject what is remaining in the pen and
complete your dose with a new SoloStar or use a new SoloStar for your full dose.
A
. Use the injection method as instructed by your healthcare professional.
B
. Insert the needle into the skin.

C
. Deliver the dose by pressing the injection button in all the way. The number in the dose
window will return to “0” as you inject.
D
. Keep the injection button pressed all the way in. Slowly count to 10 before you withdraw the
needle from the skin. This ensures that the full dose will be delivered.
The pen plunger moves with each dose. The plunger will reach the end of the cartridge when the
total of 300 units of insulin have been used.
Step 6.
Remove
and discard the needle
Always remove the needle after each injection and store SoloStar without a needle attached.
This helps prevent:
Contamination and/or infection,
Entry of air into the insulin reservoir and leakage of insulin, which can cause inaccurate dosing.
A
. Put the outer needle cap back on the needle, and use it to unscrew the needle from the pen. To
reduce the risk of accidental needle injury, never replace the inner needle cap.
B.
Dispose of the needle safely, as instructed by your healthcare professional.
C
. Always put the pen cap back on the pen, then store the pen until your next injection.
Please check the reverse (insulin) side of this leaflet for instructions on how to store SoloStar.
If your SoloStar is in cool storage, take it out 1 to 2 hours before you inject to allow it to warm up.
Cold insulin is more painful to inject.
Discard your used SoloStar as required by your local authorities.
Protect your SoloStar from dust and dirt.
You can clean the outside of your SoloStar by wiping it with a damp cloth.
Do not soak, wash or lubricate the pen as this may damage it.
Your SoloStar is designed to work accurately and safely. It should be handled with care. Avoid
situations where SoloStar might be damaged. If you are concerned that your SoloStar may be
damaged, use a new one.

Source: European Medicines Agency
 - Please bookmark this page (add it to your favorites).
- Please bookmark this page (add it to your favorites).
- If you wish to link to this page, you can do so by referring to the URL address below this line.
https://theodora.com/drugs/eu/apidra.html
Copyright © 1995-2021 ITA all rights reserved.
|








































































































































































































































































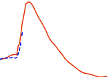










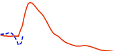




















































































































































































































































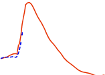








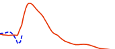




















































































































































































































































 - Please bookmark this page (add it to your favorites).
- Please bookmark this page (add it to your favorites).










































