Product Characteristics
ANNEX I
SUMMARY OF PRODUCT CHARACTERISTICS
1. NAME OF THE MEDICINAL PRODUCT
LUMIGAN 0.1 mg/ml eye drops, solution
2. QUALITATIVE AND QUANTITATIVE COMPOSITION
One ml of solution contains 0.1 mg bimatoprost.
Excipient:
One ml of solution contains 0.2 mg benzalkonium chloride.
For a full list of excipients, see section 6.1.
Colourless to slightly yellow solution.
4.1 Therapeutic indications
Reduction of elevated intraocular pressure in chronic open-angle glaucoma and ocular hypertension in
adults
(
as monotherapy or as adjunctive therapy to beta-blockers).
4.2 Posology and method of administration
The recommended dose is one drop in the affected eye(s) once daily, administered in the evening. The
dose should not exceed once daily as more frequent administration may lessen the intraocular pressure
lowering effect.
If more than one topical ophthalmic medicinal product is being used, each one should be administered
at least 5 minutes apart.
Use in children and adolescents (under the age of 18):
LUMIGAN is not recommended for use in children below 18 years, due to a lack of data on safety and
efficacy.
Use in hepatic and renal impairment:
LUMIGAN has not been studied in patients with renal or moderate to severe hepatic impairment and
should therefore be used with caution in such patients. In patients with a history of mild liver disease
or abnormal alanine aminotransferase (ALT), aspartate aminotransferase (AST) and/or bilirubin at
baseline, bimatoprost 0.3 mg/ml eye drops, solution had no adverse effect on liver function over 24
months.
Hypersensitivity to the active substance or to any of the excipients.
LUMIGAN 0.1 mg/ml is contraindicated in patients who have had a suspected previous adverse
reaction to benzalkonium chloride that has led to discontinuation.
4.4 Special warnings and precautions for use
Before treatment is initiated, patients should be informed of the possibility of eyelash growth,
darkening of the eyelid skin and increased iris pigmentation since these have been observed during
treatment with LUMIGAN. Some of these changes may be permanent, and may lead to differences in
appearance between the eyes when only one eye is treated. The change in iris pigmentation occurs
slowly and may not be noticeable for several months or years. . At 12 months, there was one report of
iris hyperpigmentation with bimatoprost 0.1 mg/ml eye drops, solution (incidence of 0.5%). At
12 months, the incidence with bimatoprost 0.3 mg/ml eye drops, solution was 1.5% and did not
increase following 3 years treatment (see section 4.8). Periorbital tissue pigmentation has been
reported to be reversible in some patients.
LUMIGAN has not been studied in patients with compromised respiratory function and should
therefore be used with caution in such patients. In clinical studies, in those patients with a history of a
compromised respiratory function, no significant untoward respiratory effects have been seen.
LUMIGAN has not been studied in patients with heart block more severe than first degree or
uncontrolled congestive heart failure. There have been a limited number of spontaneous reports of
bradycardia or hypotension with bimatoprost 0.3 mg/ml eye drops, solution. LUMIGAN should be
used with caution in patients predisposed to low heart rate or low blood pressure.
LUMIGAN has not been studied in patients with inflammatory ocular conditions, neovascular,
inflammatory, angle-closure glaucoma, congenital glaucoma or narrow-angle glaucoma.
Cystoid macular oedema has been uncommonly reported (≥1/1000 to <1/100) following treatment
with bimatoprost 0.3 mg/ml eye drops, solution. Therefore, LUMIGAN should be used with caution in
patients with known risk factors for macular oedema (e.g. aphakic patients, pseudophakic patients with
a torn posterior lens capsule).
There have been rare spontaneous reports of reactivation of previous corneal infiltrates or ocular
infections with bimatoprost 0.3 mg/ml eye drops, solution. LUMIGAN should be used with caution in
patients with a prior history of significant ocular viral infections (e.g. herpes simplex) or uveitis/iritis.
LUMIGAN 0.1 mg/ml contains the preservative benzalkonium chloride (200 ppm), which may be
absorbed by soft contact lenses. Eye irritation and discolouration of the soft contact lenses may also
occur because of the presence of benzalkonium chloride. Contact lenses should be removed prior to
instillation and may be reinserted 15 minutes following administration.
Benzalkonium chloride, which is commonly used as a preservative in ophthalmic products, has been
reported to cause punctate keratopathy and/or toxic ulcerative keratopathy. Since
LUMIGAN 0.1 mg/ml contains 200 ppm benzalkonium chloride (four times the concentration in
bimatoprost 0.3 mg/ml eye drops), it should be used with caution in dry eye patients, in patients where
the cornea may be compromised and in patients taking multiple BAK-containing eye drops. In
addition, monitoring is required with prolonged use in such patients.
4.5 Interaction with other medicinal products and other forms of interaction
No interaction studies have been performed.
No interactions are anticipated in humans, since systemic concentrations of bimatoprost are extremely
low (less than 0.2 ng/ml) following ocular dosing with bimatoprost 0.3 mg/ml eye drops, solution.
Bimatoprost is biotransformed by any of multiple enzymes and pathways, and no effects on hepatic
drug metabolising enzymes were observed in preclinical studies.
In clinical studies, bimatoprost 0.3 mg/ml, eye drops, solution was used concomitantly with a number
of different ophthalmic beta-blocking agents without evidence of interactions.
Concomitant use of LUMIGAN and antiglaucomatous agents other than topical beta-blockers has not
been evaluated during adjunctive glaucoma therapy.
4.6 Pregnancy and lactation
Pregnancy
There are no adequate data from the use of bimatoprost in pregnant women. Animal studies have
shown reproductive toxicity at high maternotoxic doses (see section 5.3).
LUMIGAN should not be used during pregnancy unless clearly necessary.
Lactation
It is unknown whether bimatoprost is excreted in human breast milk. Animal studies have shown
excretion of bimatoprost in breast milk. A decision on whether to continue/discontinue breast-feeding
or to continue/discontinue therapy with LUMIGAN should be made taking into account the benefit of
breast-feeding to the child and the benefit of LUMIGAN therapy to the woman.
4.7 Effects on ability to drive and use machines
LUMIGAN has negligible influence on the ability to drive and use machines. As with any ocular
treatment, if transient blurred vision occurs at instillation, the patient should wait until the vision clears
before driving or using machines.
In a 12-month Phase III clinical study approximately 38 % of patients treated with
LUMIGAN 0.1 mg/ml eye drops, solution experienced adverse reactions. The most frequently
reported adverse reaction was conjunctival hyperaemia (mostly trace to mild and of a non-
inflammatory nature) occurring in 29 % of patients. Approximately 4 % of patients discontinued due
to any adverse event in the 12-month study.
The following adverse reactions were reported during clinical trials with LUMIGAN 0.1 mg/ml eye
drops, solution. Most were ocular, mild and none was serious.
Very common (≥1/10); common (≥1/100 to <1/10); uncommon (≥1/1,000 to <1/100); rare (≥1/10,000
to <1/1,000); very rare (<1/10,000) adverse reactions are presented according to System Organ Class
in Table 1 in order of decreased seriousness within each frequency grouping.
punctate keratitis, eye irritation,
eye pruritus,
growth of eyelashes
asthenopia, blurred vision,
conjunctival disorder,
conjunctival oedema, iris
hyperpigmentation, madarosis
Gastrointestinal disorders
Skin and subcutaneous tissue
disorders
erythema of eyelid, eyelids
pruritus,
skin hyperpigmentation,
hypertrichosis
dry skin, eyelid margin crusting,
eyelid oedema, pruritus
General disorders and
administration site conditions
instillation site irritation
In clinical studies, over 1800 patients have been treated with LUMIGAN 0.3 mg/ml. On combining
the data from phase III monotherapy and adjunctive LUMIGAN 0.3 mg/ml usage, the most frequently
reported adverse reactions were: growth of eyelashes in up to 45 % in the first year with the incidence
of new reports decreasing to 7 % at 2 years and 2 % at 3 years, conjunctival hyperaemia (mostly trace
to mild and thought to be of a non-inflammatory nature) in up to 44 % in the first year with the
incidence of new reports decreasing to 13 % at 2 years and 12 % at 3 years and ocular pruritus in up to
14 % of patients in the first year with the incidence of new reports decreasing to 3 % at 2 years and
0 % at 3 years. Less than 9 % of patients discontinued due to any adverse event in the first year with
the incidence of additional patient discontinuations being 3 % at both 2 and 3 years.
Additional adverse reactions reported during clinical trials with LUMIGAN 0.3 mg/ml are presented
in Table 2. The table also includes those adverse reactions which occurred with both formulations but
at a different frequency. Most were ocular, mild to moderate, and none was serious: With each
frequency grouping, undesirable effects are presented in order of decreasing seriousness.













ocular pruritus, growth of
eyelashes
corneal erosion, ocular burning,
allergic conjunctivitis,
blepharitis, worsening of visual
acuity, asthenopia, conjunctival
oedema, foreign body sensation,
ocular dryness, eye pain,
photophobia, tearing , eye
discharge, visual disturbance,
increased
iris pigmentation, eyelash
darkening
retinal haemorrhage, uveitis,
cystoid macular oedema, iritis,
blepharospasm, eyelid retraction
Skin and subcutaneous tissue
disorders
pigmentation of periocular skin
General disorders and
administration site conditions
liver function test abnormal
No case of overdose has been reported, and is unlikely to occur after ocular administration.
If overdose occurs, treatment should be symptomatic and supportive. If LUMIGAN is accidentally
ingested, the following information may be useful: in two-week oral rat and mouse studies, doses up to
100 mg/kg/day did not produce any toxicity. This dose expressed as mg/m
2
is at least 210 times higher
than the accidental dose of one bottle of LUMIGAN 0.1 mg/ml eye drops, solution in a 10 kg child.
5. PHARMACOLOGICALPROPERTIES
5.1 Pharmacodynamic properties
Pharmacotherapeutic group: Ophthalmologicals, prostaglandin analogues, ATC code: S01EE03.
The mechanism of action by which bimatoprost reduces intraocular pressure in humans is by
increasing aqueous humour outflow through the trabecular meshwork and enhancing uveoscleral
outflow. Reduction of the intraocular pressure starts approximately 4 hours after the first
















administration and maximum effect is reached within approximately 8 to 12 hours. The duration of
effect is maintained for at least 24 hours.
Bimatoprost is a potent ocular hypotensive agent. It is a synthetic prostamide, structurally related to
prostaglandin F
2α
(PGF
2α
), that does not act through any known prostaglandin receptors. Bimatoprost
selectively mimics the effects of newly discovered biosynthesised substances called prostamides. The
prostamide receptor, however, has not yet been structurally identified.
During a 12-month pivotal study in adults with LUMIGAN 0.1 mg/ml eye drops, the mean diurnal
IOP values measured at any visit over the 12-month study period differed by no more than 1.1 mmHg
throughout the day and were never greater than 17.7 mmHg.
LUMIGAN 0.1 mg/ml eye drops contains BAK in a concentration of 200 ppm.
Limited experience is available with the use of LUMIGAN in patients with open-angle glaucoma with
pseudoexfoliative and pigmentary glaucoma, and chronic angle-closure glaucoma with patent
iridotomy.
No clinically relevant effects on heart rate and blood pressure have been observed in clinical trials.
5.2 Pharmacokinetic properties
Bimatoprost penetrates the human cornea and sclera well
in vitro
. After ocular administration in
adults, the systemic exposure of bimatoprost is very low with no accumulation over time. After once
daily ocular administration of one drop of 0.3 mg/ml bimatoprost to both eyes for two weeks, blood
concentrations peaked within 10 minutes after dosing and declined to below the lower limit of
detection (0.025 ng/ml) within 1.5 hours after dosing. Mean C
max
and AUC
0-24hrs
values were similar
on days 7 and 14 at approximately 0.08 ng/ml and 0.09 ng•hr/ml respectively, indicating that a steady
bimatoprost concentration was reached during the first week of ocular dosing.
Bimatoprost is moderately distributed into body tissues and the systemic volume of distribution in
humans at steady-state was 0.67 l/kg. In human blood, bimatoprost resides mainly in the plasma. The
plasma protein binding of bimatoprost is approximately 88 %.
Bimatoprost is the major circulating species in the blood once it reaches the systemic circulation
following ocular dosing. Bimatoprost then undergoes oxidation, N-deethylation and glucuronidation to
form a diverse variety of metabolites.
Bimatoprost is eliminated primarily by renal excretion, up to 67 % of an intravenous dose
administered to healthy adult volunteers was excreted in the urine, 25 % of the dose was excreted via
the faeces. The elimination half-life, determined after intravenous administration, was approximately
45 minutes; the total blood clearance was 1.5 l/hr/kg.
Characteristics in elderly patients:
After twice daily dosing with bimatoprost 0.3 mg/ml eye drops, solution, the mean AUC
0-24hr
value of
0.0634 ng•hr/ml bimatoprost in the elderly (subjects 65 years or older) were significantly higher than
0.0218 ng•hr/ml in young healthy adults. However, this finding is not clinically relevant as systemic
exposure for both elderly and young subjects remained very low from ocular dosing. There was no
accumulation of bimatoprost in the blood over time and the safety profile was similar in elderly and
young patients.
5.3 Preclinical safety data
Effects in non-clinical studies were observed only at exposures considered sufficiently in excess of the
maximum human exposure indicating little relevance to clinical use.
Monkeys administered ocular bimatoprost concentrations of ≥0.3 mg/ml daily for 1 year had an
increase in iris pigmentation and reversible dose-related periocular effects characterised by a
prominent upper and/or lower sulcus and widening of the palpebral fissure. The increased iris
pigmentation appears to be caused by increased stimulation of melanin production in melanocytes and
not by an increase in melanocyte number. No functional or microscopic changes related to the
periocular effects have been observed, and the mechanism of action for the periocular changes is
unknown.
Bimatoprost was not mutagenic or carcinogenic in a series of
in vitro
and
in vivo
studies.
Bimatoprost did not impair fertility in rats up to doses of 0.6 mg/kg/day (at least 103-times the
intended human exposure). In embryo/foetal developmental studies abortion, but no developmental
effects were seen in mice and rats at doses that were at least 860-times or 1700-times higher than the
dose in humans, respectively. These doses resulted in systemic exposures of at least 33- or 97-times
higher, respectively, than the intended human exposure. In rat peri/postnatal studies, maternal toxicity
caused reduced gestation time, foetal death, and decreased pup body weights at ≥ 0.3 mg/kg/day (at
least 41-times the intended human exposure). Neurobehavioural functions of offspring were not
affected.
6. PHARMACEUTICAL PARTICULARS
Benzalkonium chloride
Sodium chloride
Sodium phosphate dibasic heptahydrate
Citric acid monohydrate
Hydrochloric acid or sodium hydroxide (to adjust pH)
Purified water
4 weeks after first opening.
6.4 Special precautions for storage
This medicinal product does not require any special storage conditions.
6.5
Nature and contents of container
White opaque low density polyethylene bottles with polystyrene screw cap. Each bottle has a fill
volume of 3 ml.
The following pack sizes are available: cartons containing 1 or 3 bottles of 3 ml solution. Not all pack
sizes may be marketed.
6.6 Special precautions for disposal
7. MARKETING AUTHORISATION HOLDER
Allergan Pharmaceuticals Ireland
Castlebar Road
Westport
Co. Mayo
Ireland
8. MARKETING AUTHORISATION NUMBER
EU/1/02/205/003-004
9. DATE OF FIRST AUTHORISATION/RENEWAL OF THE AUTHORISATION
07/01/2010
10. DATE OF REVISION OF THE TEXT
Detailed information on this product is available on the website of the European Medicines Agency
(EMEA) http://www.emea.europa.eu
NAME OF THE MEDICINAL PRODUCT
LUMIGAN 0.3 mg/ml eye drops, solution
QUALITATIVE AND QUANTITATIVE COMPOSITION
One ml of solution contains 0.3 mg bimatoprost.
Excipient
:
One ml of solution contains 0.05 mg benzalkonium chloride.
For a full list of excipients, see section 6.1.
Colourless to slightly yellow solution.
4.1 Therapeutic indications
Reduction of elevated intraocular pressure in chronic open-angle glaucoma and ocular hypertension in
adults (as monotherapy or as adjunctive therapy to beta-blockers).
4.2 Posology and method of administration
The recommended dose is one drop in the affected eye(s) once daily, administered in the evening. The
dose should not exceed once daily as more frequent administration may lessen the intraocular pressure
lowering effect.
If more than one topical ophthalmic medicinal product is being used, each one should be administered
at least 5 minutes apart.
Use in children and adolescents (under the age of 18):
LUMIGAN is not recommended for use in children below 18 years, due to a lack of data on safety
and efficacy.
Use in hepatic and renal impairment:
LUMIGAN has not been studied in patients with renal or moderate to severe hepatic impairment and
should therefore be used with caution in such patients. In patients with a history of mild liver disease
or abnormal alanine aminotransferase (ALT), aspartate aminotransferase (AST) and/or bilirubin at
baseline, bimatoprost 0.3 mg/ml eye drops, solution had no adverse effect on liver function over 24
months.
Hypersensitivity to the active substance or to any of the excipients.
4.4 Special warnings and precautions for use
Before treatment is initiated, patients should be informed of the possibility of eyelash growth,
darkening of the eyelid skin and increased iris pigmentation since these have been observed during
treatment with LUMIGAN. Some of these changes may be permanent, and may lead to differences in
appearance between the eyes when only one eye is treated. The change in iris pigmentation occurs
slowly and may not be noticeable for several months or years. At 12 months, the incidence was 1.5%
and did not increase following 3 years treatment (see section 4.8). Periorbital tissue pigmentation has
been reported to be reversible in some patients.
LUMIGAN has not been studied in patients with compromised respiratory function and should
therefore be used with caution in such patients. In clinical studies, in those patients with a history of a
compromised respiratory function, no significant untoward respiratory effects have been seen.
LUMIGAN has not been studied in patients with heart block more severe than first degree or
uncontrolled congestive heart failure. There have been a limited number of spontaneous reports of
bradycardia or hypotension with bimatoprost 0.3 mg/ml eye drops, solution. LUMIGAN should be
used with caution in patients predisposed to low heart rate or low blood pressure
LUMIGAN has not been studied in patients with inflammatory ocular conditions, neovascular,
inflammatory, angle-closure glaucoma, congenital glaucoma or narrow-angle glaucoma.
Cystoid macular oedema has been uncommonly reported (≥1/1000 to <1/100) following treatment
with bimatoprost 0.3 mg/ml eye drops. Therefore, LUMIGAN should be used with caution in patients
with known risk factors for macular oedema (e.g. aphakic patients, pseudophakic patients with a torn
posterior lens capsule).
There have been rare spontaneous reports of reactivation of previous corneal infiltrates or ocular
infections with bimatoprost 0.3 mg/ml eye drops, solution. LUMIGAN should be used with caution in
patients with a prior history of significant ocular viral infections (e.g. herpes simplex) or uveitis/iritis.
Bimatoprost 0.3 mg/ml eye drops, solution contains the preservative benzalkonium chloride, which
may be absorbed by soft contact lenses. Eye irritation and discolouration of the soft contact lenses
may also occur because of the presence of benzalkonium chloride. Contact lenses should be removed
prior to instillation and may be reinserted 15 minutes following administration.
Benzalkonium chloride, which is commonly used as a preservative in ophthalmic products, has been
reported to cause punctate keratopathy and/or toxic ulcerative keratopathy. Since LUMIGAN contains
benzalkonium chloride, monitoring is required with frequent or prolonged use in dry eye patients or
where the cornea is compromised.
4.5 Interaction with other medicinal products and other forms of interaction
No interaction studies have been performed.
No interactions are anticipated in humans, since systemic concentrations of bimatoprost are extremely
low (less than 0.2 ng/ml) following ocular dosing with bimatoprost 0.3 mg/ml eye drops, solution.
Bimatoprost is biotransformed by any of multiple enzymes and pathways, and no effects on hepatic
drug metabolising enzymes were observed in preclinical studies.
In clinical studies, LUMIGAN was used concomitantly with a number of different ophthalmic
beta-blocking agents without evidence of interactions.
Concomitant use of LUMIGAN and antiglaucomatous agents other than topical beta-blockers has not
been evaluated during adjunctive glaucoma therapy.
4.6 Pregnancy and lactation
Pregnancy
There are no adequate data from the use of bimatoprost in pregnant women. Animal studies have
shown reproductive toxicity at high maternotoxic doses (see section 5.3).
LUMIGAN should not be used during pregnancy unless clearly necessary.
Lactation
It is unknown whether bimatoprost is excreted in human breast milk. Animal studies have shown
excretion of bimatoprost in breast milk. A risk to the suckling child cannot be excluded. A decision on
whether to continue/discontinue breast-feeding or to continue/discontinue therapy with LUMIGAN
should be made, taking into account the benefit of breast-feeding to the child and the benefit of
LUMIGAN therapy to the woman.
4.7 Effects on ability to drive and use machines
LUMIGAN has negligible influence on the ability to drive and use machines. As with any ocular
treatment, if transient blurred vision occurs at instillation, the patient should wait until the vision clears
before driving or using machines.
In clinical studies, over 1800 patients have been treated with LUMIGAN 0.3 mg/ml eye drops,
solution. On combining the data from phase III monotherapy and adjunctive LUMIGAN 0.3 mg/ml
eye drops, solution usage, the most frequently reported treatment-related adverse events were: growth
of eyelashes in up to 45% in the first year with the incidence of new reports decreasing to 7% at 2
years and 2% at 3 years, conjunctival hyperaemia (mostly trace to mild and thought to be of a non-
inflammatory nature) in up to 44% in the first year with the incidence of new reports decreasing to
13% at 2 years and 12% at 3 years and ocular pruritus in up to 14% of patients in the first year with
the incidence of new reports decreasing to 3% at 2 years and 0% at 3 years. Less than 9% of patients
discontinued due to any adverse event in the first year with the incidence of additional patient
discontinuations being 3% at both 2 and 3 years.
The following adverse reactions were reported during clinical trials with LUMIGAN 0.3 mg/ml eye
drops, solution. Most were ocular, mild to moderate, and none was serious:
Very common (≥1/10); common (≥1/100 to <1/10); uncommon (≥1/1,000 to <1/100); rare (≥1/10,000
to <1/1,000); very rare (<1/10,000) and not known (cannot be estimated from available data)
undesirable effects are presented according to System Organ Class in Table 1. Within each frequency
grouping, undesirable effects are presented in order of decreasing seriousness.
conjunctival hyperaemia,
ocular pruritus, growth of
eyelashes
superficial punctate keratitis,
corneal erosion, ocular
burning, ocular irritation,
allergic conjunctivitis,
blepharitis, worsening of
visual acuity, asthenopia,
conjunctival oedema, foreign
body sensation, ocular
dryness, eye pain,
photophobia, tearing, eye
discharge, visual disturbance,
increased iris pigmentation,
eyelash darkening.
retinal haemorrhage, uveitis,
cystoid macular oedema, iritis,
blepharospasm, eyelid retraction
Gastrointestinal disorders
Skin and subcutaneous tissue
disorders
eyelid erythema, eyelid pruritus,
pigmentation of periocular skin
eyelid oedema, hirsutism.
General disorders and
administration site conditions
liver function test abnormal
No case of overdose has been reported, and is unlikely to occur after ocular administration.
If overdose occurs, treatment should be symptomatic and supportive. If LUMIGAN is accidentally
ingested, the following information may be useful: in two-week oral rat and mouse studies, doses up to
100 mg/kg/day did not produce any toxicity. This dose expressed as mg/m
2
is at least 70-times higher
than the accidental dose of one bottle of LUMIGAN 0.3 mg/ml eye drops, solution in a 10 kg child.
PHARMACOLOGICAL PROPERTIES
5.1 Pharmacodynamic properties
Pharmacotherapeutic group: Ophthalmologicals, prostaglandin analogues, ATC code: S01EE03.
The mechanism of action by which bimatoprost reduces intraocular pressure in humans is by
increasing aqueous humour outflow through the trabecular meshwork and enhancing uveoscleral
outflow. Reduction of the intraocular pressure starts approximately 4 hours after the first


















administration and maximum effect is reached within approximately 8 to 12 hours. The duration of
effect is maintained for at least 24 hours.
Bimatoprost is a potent ocular hypotensive agent. It is a synthetic prostamide, structurally related to
prostaglandin F
2α
(PGF
2α
), that does not act through any known prostaglandin receptors. Bimatoprost
selectively mimics the effects of newly discovered biosynthesised substances called prostamides. The
prostamide receptor, however, has not yet been structurally identified.
During 12 months’ monotherapy treatment with LUMIGAN 0.3 mg/ml in adults, versus timolol, mean
change from baseline in morning (08:00) intraocular pressure ranged from -7.9 to -8.8 mm Hg. At any
visit, the mean diurnal IOP values measured over the 12-month study period differed by no more than
1.3 mmHg throughout the day and were never greater than 18.0 mmHg.
In a 6-month clinical study with LUMIGAN 0.3 mg/ml, versus latanoprost, a statistically superior
reduction in morning mean IOP (ranging from -7.6 to -8.2 mmHg for bimatoprost versus –6.0 to
-7.2 mmHg for latanoprost) was observed at all visits throughout the study. Conjunctival hyperaemia,
growth of eyelashes, and eye pruritus were statistically significantly higher with bimatoprost than with
latanoprost, however, the discontinuation rates due to adverse events were low with no statistically
significant difference.
Compared to treatment with beta-blocker alone, adjunctive therapy with beta-blocker and
LUMIGAN 0.3 mg/ml lowered mean morning (08:00) intraocular pressure by -6.5 to -8.1 mmHg.
Limited experience is available in patients with open-angle glaucoma with pseudoexfoliative and
pigmentary glaucoma, and chronic angle-closure glaucoma with patent iridotomy.
No clinically relevant effects on heart rate and blood pressure have been observed in clinical trials.
5.2 Pharmacokinetic properties
Bimatoprost penetrates the human cornea and sclera well
in vitro
. After ocular administration in
adults, the systemic exposure of bimatoprost is very low with no accumulation over time. After once
daily ocular administration of one drop of LUMIGAN 0.3 mg/ml to both eyes for two weeks, blood
concentrations peaked within 10 minutes after dosing and declined to below the lower limit of
detection (0.025 ng/ml) within 1.5 hours after dosing. Mean C
max
and AUC
0-24hrs
values were similar
on days 7 and 14 at approximately 0.08 ng/ml and 0.09 ng•hr/ml respectively, indicating that a steady
bimatoprost concentration was reached during the first week of ocular dosing.
Bimatoprost is moderately distributed into body tissues and the systemic volume of distribution in
humans at steady-state was 0.67 l/kg. In human blood, bimatoprost resides mainly in the plasma. The
plasma protein binding of bimatoprost is approximately 88%.
Bimatoprost is the major circulating species in the blood once it reaches the systemic circulation
following ocular dosing. Bimatoprost then undergoes oxidation, N-deethylation and glucuronidation
to form a diverse variety of metabolites.
Bimatoprost is eliminated primarily by renal excretion, up to 67% of an intravenous dose administered
to healthy adult volunteers was excreted in the urine, 25% of the dose was excreted via the faeces.
The elimination half-life, determined after intravenous administration, was approximately 45 minutes;
the total blood clearance was 1.5 l/hr/kg.
Characteristics in elderly patients:
After twice daily dosing of LUMIGAN 0.3 mg/ml, the mean AUC
0-24hr
value of 0.0634 ng•hr/ml
bimatoprost in the elderly (subjects 65 years or older) were significantly higher than 0.0218 ng•hr/ml
in young healthy adults. However, this finding is not clinically relevant as systemic exposure for both
elderly and young subjects remained very low from ocular dosing. There was no accumulation of
bimatoprost in the blood over time and the safety profile was similar in elderly and young patients.
5.3 Preclinical safety data
Effects in non-clinical studies were observed only at exposures considered sufficiently in excess of the
maximum human exposure indicating little relevance to clinical use.
Monkeys administered ocular bimatoprost concentrations of ≥0.3 mg/ml daily for 1 year had an
increase in iris pigmentation and reversible dose-related periocular effects characterised by a
prominent upper and/or lower sulcus and widening of the palpebral fissure. The increased iris
pigmentation appears to be caused by increased stimulation of melanin production in melanocytes and
not by an increase in melanocyte number. No functional or microscopic changes related to the
periocular effects have been observed, and the mechanism of action for the periocular changes is
unknown.
Bimatoprost was not mutagenic or carcinogenic in a series of
in vitro
and
in vivo
studies.
Bimatoprost did not impair fertility in rats up to doses of 0.6 mg/kg/day (at least 103-times the
intended human exposure). In embryo/foetal developmental studies abortion, but no developmental
effects were seen in mice and rats at doses that were at least 860-times or 1700-times higher than the
dose in humans, respectively. These doses resulted in systemic exposures of at least 33- or 97-times
higher, respectively, than the intended human exposure. In rat peri/postnatal studies, maternal toxicity
caused reduced gestation time, foetal death, and decreased pup body weights at ≥0.3 mg/kg/day (at
least 41-times the intended human exposure). Neurobehavioural functions of offspring were not
affected.
PHARMACEUTICAL PARTICULARS
Benzalkonium chloride
Sodium chloride
Sodium phosphate dibasic heptahydrate
Citric acid monohydrate
Hydrochloric acid or sodium hydroxide (to adjust pH)
Purified water
4 weeks after first opening.
6.4 Special precautions for storage
This medicinal product does not require any special storage conditions.
6.5 Nature and contents of container
White opaque low density polyethylene bottles with polystyrene screw cap. Each bottle has a fill
volume of 3 ml.
The following pack sizes are available: cartons containing 1 or 3 bottles of 3 ml solution. Not all pack
sizes may be marketed.
6.6 Special precautions for disposal
MARKETING AUTHORISATION HOLDER
Allergan Pharmaceuticals Ireland
Castlebar Road
Westport
Co. Mayo
Ireland
MARKETING AUTHORISATION NUMBER
DATE OF FIRST AUTHORISATION/RENEWAL OF THE AUTHORISATION
8 March 2002 / 20 February 2007
10. DATE OF REVISION OF THE TEXT
Detailed information on this product is available on the website of the European Medicines Agency
(EMEA): http://www.emea.europa.eu
A. MANUFACTURING AUTHORISATION HOLDER
RESPONSIBLE FOR BATCH RELEASE
B. CONDITIONS OF THE MARKETING AUTHORISATION
A. MANUFACTURING AUTHORISATION HOLDER RESPONSIBLE FOR BATCH
RELEASE
Name and address of the manufacturer responsible for batch release
Allergan Pharmaceuticals Ireland
Castlebar Road
Westport
County Mayo
Ireland
B. CONDITIONS OF THE MARKETING AUTHORISATION
CONDITIONS OR RESTRICTIONS REGARDING SUPPLY AND USE IMPOSED ON
THE MARKETING AUTHORISATION HOLDER
Medicinal product subject to medical prescription.
CONDITIONS OR RESTRICTIONS WITH REGARD TO THE SAFE AND
EFFECTIVE USE OF THE MEDICINAL PRODUCT
Pharmacovigilance system
The MAH must ensure that the system of pharmacovigilance, as described in version 1.0 presented in
Module 1.8.1 of the Marketing Authorisation Application, is in place and functioning before and
whilst the product is on the market.
Risk Management Plan
The MAH commits to performing the studies and additional pharmacovigilance activities
detailed in the Pharmacovigilance Plan, as agreed in version 4.0 of the Risk Management Plan
(RMP) presented in Module 1.8.2 of the Marketing Authorisation Application and any
subsequent updates of the RMP agreed by the CHMP.
As per the CHMP Guideline on Risk Management Systems for medicinal products for human use, any
updated RMP should be submitted at the same time as the following Periodic Safety Update Report
(PSUR).
In addition, an updated RMP should be submitted:
•
When new information is received that may impact on the current Safety Specification,
Pharmacovigilance Plan or risk minimisation activities
•
Within 60 days of an important (pharmacovigilance or risk minimisation) milestone being
reached
•
At the request of the EMEA
PSURs
PSURS will be submitted annually until otherwise specified by the CHMP
ANNEX III
LABELLING AND PACKAGE LEAFLET
PARTICULARS TO APPEAR ON THE OUTER PACKAGING
NAME OF THE MEDICINAL PRODUCT
LUMIGAN 0.1 mg/ml, eye drops, solution
Bimatoprost
STATEMENT OF ACTIVE SUBSTANCE(S)
One ml of solution contains 0.1 mg bimatoprost
Benzalkonium chloride, sodium phosphate dibasic heptahydrate, citric acid monohydrate, sodium
chloride, hydrochloric acid or sodium hydroxide (to adjust pH) and purified water
PHARMACEUTICAL FORM AND CONTENTS
Eye drops, solution
1 x 3 ml
METHOD AND ROUTE(S) OF ADMINISTRATION
Ocular use.
Read the package leaflet before use.
SPECIAL WARNING THAT THE MEDICINAL PRODUCT MUST BE STORED OUT
OF THE REACH AND SIGHT OF CHILDREN
Keep out of the reach and sight of children.
OTHER SPECIAL WARNING(S), IF NECESSARY
Remove contact lenses before use.
EXP
Discard four weeks after first opening.
Opened:
SPECIAL STORAGE CONDITIONS



























10. SPECIAL PRECAUTIONS FOR DISPOSAL OF UNUSED MEDICINAL PRODUCTS
OR WASTE MATERIALS DERIVED FROM SUCH MEDICINAL PRODUCTS, IF
APPROPRIATE
11. NAME AND ADDRESS OF THE MARKETING AUTHORISATION HOLDER
Allergan Pharmaceuticals Ireland
Castlebar Road
Westport
Co. Mayo
Ireland
12. MARKETING AUTHORISATION NUMBER(S)
14. GENERAL CLASSIFICATION FOR SUPPLY
Medicinal product subject to medical prescription
16. INFORMATION IN BRAILLE

















PARTICULARS TO APPEAR ON THE OUTER PACKAGING
CARTON CONTAINING THREE BOTTLES
NAME OF THE MEDICINAL PRODUCT
LUMIGAN 0.1 mg/ml, eye drops, solution
Bimatoprost
STATEMENT OF ACTIVE SUBSTANCE(S)
One ml of solution contains 0.1 mg bimatoprost
Benzalkonium chloride, sodium phosphate dibasic heptahydrate, citric acid monohydrate, sodium
chloride, hydrochloric acid or sodium hydroxide (to adjust pH) and purified water
PHARMACEUTICAL FORM AND CONTENTS
Eye drops, solution
3 x 3 ml
METHOD AND ROUTE(S) OF ADMINISTRATION
Ocular use.
Read the package leaflet before use.
SPECIAL WARNING THAT THE MEDICINAL PRODUCT MUST BE STORED OUT
OF THE REACH AND SIGHT OF CHILDREN
Keep out of the reach and sight of children.
OTHER SPECIAL WARNING(S), IF NECESSARY
Remove contact lenses before use.
EXP
Discard four weeks after first opening.
Opened (1):
Opened (2):
Opened (3):























SPECIAL STORAGE CONDITIONS
10. SPECIAL PRECAUTIONS FOR DISPOSAL OF UNUSED MEDICINAL PRODUCTS
OR WASTE MATERIALS DERIVED FROM SUCH MEDICINAL PRODUCTS, IF
APPROPRIATE
11. NAME AND ADDRESS OF THE MARKETING AUTHORISATION HOLDER
Allergan Pharmaceuticals Ireland
Castlebar Road
Westport
Co. Mayo
Ireland
12. MARKETING AUTHORISATION NUMBER(S)
14. GENERAL CLASSIFICATION FOR SUPPLY
Medicinal product subject to medical prescription
16. INFORMATION IN BRAILLE



















PARTICULARS TO APPEAR ON THE OUTER PACKAGING
NAME OF THE MEDICINAL PRODUCT
LUMIGAN 0.3 mg/ml, eye drops, solution
Bimatoprost
STATEMENT OF ACTIVE SUBSTANCE(S)
One ml of solution contains 0.3 mg bimatoprost
Benzalkonium chloride, sodium phosphate dibasic heptahydrate, citric acid monohydrate, sodium
chloride, hydrochloric acid or sodium hydroxide (to adjust pH) and purified water
PHARMACEUTICAL FORM AND CONTENTS
Eye drops, solution
1 x 3 ml
METHOD AND ROUTE(S) OF ADMINISTRATION
Ocular use.
Read the package leaflet before use.
SPECIAL WARNING THAT THE MEDICINAL PRODUCT MUST BE STORED OUT
OF THE REACH AND SIGHT OF CHILDREN
Keep out of the reach and sight of children.
OTHER SPECIAL WARNING(S), IF NECESSARY
Remove contact lenses before use.
EXP
Discard four weeks after first opening.
Opened:
SPECIAL STORAGE CONDITIONS



























10. SPECIAL PRECAUTIONS FOR DISPOSAL OF UNUSED MEDICINAL PRODUCTS
OR WASTE MATERIALS DERIVED FROM SUCH MEDICINAL PRODUCTS, IF
APPROPRIATE
11. NAME AND ADDRESS OF THE MARKETING AUTHORISATION HOLDER
Allergan Pharmaceuticals Ireland
Castlebar Road
Westport
Co. Mayo
Ireland
12. MARKETING AUTHORISATION NUMBER(S)
14. GENERAL CLASSIFICATION FOR SUPPLY
Medicinal product subject to medical prescription
16. INFORMATION IN BRAILLE

















PARTICULARS TO APPEAR ON THE OUTER PACKAGING
CARTON CONTAINING THREE BOTTLES
NAME OF THE MEDICINAL PRODUCT
LUMIGAN 0.3 mg/ml, eye drops, solution
Bimatoprost
STATEMENT OF ACTIVE SUBSTANCE(S)
One ml of solution contains 0.3 mg bimatoprost
Benzalkonium chloride, sodium phosphate dibasic heptahydrate, citric acid monohydrate, sodium
chloride, hydrochloric acid or sodium hydroxide (to adjust pH) and purified water
PHARMACEUTICAL FORM AND CONTENTS
Eye drops, solution
3 x 3 ml
METHOD AND ROUTE(S) OF ADMINISTRATION
Ocular use.
Read the package leaflet before use.
SPECIAL WARNING THAT THE MEDICINAL PRODUCT MUST BE STORED OUT
OF THE REACH AND SIGHT OF CHILDREN
Keep out of the reach and sight of children.
OTHER SPECIAL WARNING(S), IF NECESSARY
Remove contact lenses before use.
EXP
Discard four weeks after first opening.
Opened (1):
Opened (2):
Opened (3):























SPECIAL STORAGE CONDITIONS
10. SPECIAL PRECAUTIONS FOR DISPOSAL OF UNUSED MEDICINAL PRODUCTS
OR WASTE MATERIALS DERIVED FROM SUCH MEDICINAL PRODUCTS, IF
APPROPRIATE
11. NAME AND ADDRESS OF THE MARKETING AUTHORISATION HOLDER
Allergan Pharmaceuticals Ireland
Castlebar Road
Westport
Co. Mayo
Ireland
12. MARKETING AUTHORISATION NUMBER(S)
14. GENERAL CLASSIFICATION FOR SUPPLY
Medicinal product subject to medical prescription
16. INFORMATION IN BRAILLE



















PACKAGE LEAFLET: INFORMATION FOR THE USER
LUMIGAN 0.1 mg/ml, eye drops, solution
Bimatoprost
Read all of this leaflet carefully before you start using this medicine.
-
Keep this leaflet. You may need to read it again.
If you have further questions, please ask your doctor or your pharmacist.
This medicine has been prescribed for you. Do not pass it on to others. It may harm them, even
if their symptoms are the same as yours.
If any of the side effects get serious, or if you notice any side effects not listed in this leaflet
please tell your doctor or pharmacist.
In this leaflet
:
1. What LUMIGAN 0.1 mg/ml is and what it is used for
2. Before you use LUMIGAN 0.1 mg/ml
3. How to use LUMIGAN 0.1 mg/ml
4. Possible side effects
5.
How to store LUMIGAN 0.1 mg/ml
6.
Further information
1.
WHAT LUMIGAN 0.1 mg/ml IS AND WHAT IT IS USED FOR
LUMIGAN is an antiglaucoma preparation. It belongs to a group of medicines called prostamides.
LUMIGAN eye drops are used to reduce high pressure in the eye. This high pressure can lead to a
disease called glaucoma. If the high pressure is not reduced, it could eventually damage your sight.
Your eye contains a clear, watery liquid that feeds the inside of the eye. Liquid is constantly being
drained out of the eye and new liquid is made to replace this. If the liquid cannot drain out quickly
enough, the pressure inside the eye builds up. LUMIGAN works by increasing the amount of liquid
that is drained. This reduces the pressure inside the eye.
LUMIGAN may be used on its own or with other drops called beta-blockers which also reduce
pressure.
2.
BEFORE YOU USE LUMIGAN 0.1 mg/ml
Do not use LUMIGAN 0.1 mg/ml:
-
if you are allergic (hypersensitive) to bimatoprost or any of the other ingredients of LUMIGAN.
-
if you have had to stop using eye drops in the past because of a side effect of the preservative
benzalkonium chloride.
Take special care with LUMIGAN 0.1 mg/ml:
Talk to your doctor, if:
-
You have any breathing problems.
You have had a cataract surgery in the past
You have or have had any problems with your cornea (front transparent part of the eye)
You wear contact lenses (see “Important information about some of the ingredients of
LUMIGAN 0.1 mg/ml”)
You have or have had low blood pressure or low heart rate
You have liver or kidney problems.
You have had a viral infection or inflammation of the eye
LUMIGAN may cause your eyelashes to darken and grow, and cause the skin around the eyelid to
darken too. The colour of your iris may also go darker over time. These changes may be permanent.
The change may be more noticeable if you are only treating one eye.
LUMIGAN has not been tested in children under the age of 18 and therefore must not be used by
patients under 18 years.
Using other medicines:
Please tell your doctor or pharmacist if you are taking, or have recently taken, any other medicines,
including medicines obtained without a prescription.
If you use LUMIGAN with another eye drop, leave at least five minutes between putting in
LUMIGAN and then the other drops.
Pregnancy and breast-feeding
Ask your doctor or pharmacist for advice before taking any medicine. LUMIGAN should not be used
during pregnancy unless your doctor still recommends it.
LUMIGAN may get into breast milk so you should not breast-feed while you are taking LUMIGAN.
Driving and using machines:
Your sight may become blurred for a short time just after using LUMIGAN. You should not drive or
use machines until your sight is clear again.
Important information about some of the ingredients of LUMIGAN 0.1 mg/ml
Do not use the drops when you are wearing your lenses. Wait 15 minutes after using the drops before
you put your lenses back in. A preservative in LUMIGAN called benzalkonium chloride may cause
eye irritation and can discolour soft contact lenses.
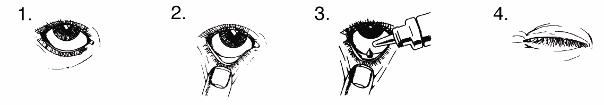
HOW TO USE LUMIGAN 0.1 mg/ml
LUMIGAN should only be applied to the eye. Always use LUMIGAN exactly as your doctor has told
you. You should check with your doctor or pharmacist if you are not sure. The usual dose is one drop
of LUMIGAN in the evening, once daily in each eye that needs treatment.
If you use LUMIGAN with another eye medicine, apply the LUMIGAN eye drop first and wait 5
minutes before applying the second eye medicine.
You must not use the bottle if the tamper-proof seal on the bottle neck is broken before you first use it.
1.
Wash your hands. Tilt your head back and look at the ceiling.
2.
Gently pull down the lower eyelid until there is a small pocket.
3.
Turn the bottle upside down and squeeze it to release one drop into each eye that needs treatment.
4.
Let go of the lower lid, and close your eye for 30 seconds.
If a drop misses your eye, try again.
To help prevent infections, do not let the tip of the bottle touch your eye or anything else. Put the cap
back on and close the bottle straight after you have used it.
If you use more LUMIGAN 0.1 mg/ml than you should
If you use more LUMIGAN than you should, it is unlikely to cause you any serious harm. Put your
next dose in at the usual time. If you are worried, talk to your doctor or pharmacist.
If you forget to use LUMIGAN 0.1 mg/ml
If you forget to use LUMIGAN,
use a single drop as soon as you remember, and then go back to your
regular routine. Do not use a double dose to make up for a forgotten dose.
If you stop using LUMIGAN 0.1 mg/ml
LUMIGAN should be used every day to work properly. If you stop using LUMIGAN the pressure
inside your eye may go up, therefore talk to your doctor before stopping this treatment.
If you have any further questions on the use of this product, ask your doctor or pharmacist.
Like all medicines, LUMIGAN 0.1 mg/ml can have side effects, although not everybody gets them.
Most of the side effects are not serious.
Very common side effects
These may affect one or more users in 10
Affecting the eye
•
Slight redness (up to 29 % of people)
Common side effects
These may affect 1 to 9 users in 100
Affecting the eye
•
Small breaks in the surface of the eye, with or without inflammation
Irritation, when drop is put in the eye
Inflamed, red and itchy eyelids
Darker skin colour around the eye
Hair growth around the eye
Uncommon side effects
These may affect 1 to 9 users in 1000
Swelling of the surface of the eye
Affecting the skin
•
Dry skin
•
Crusting of on the edge of the eyelid
•
Swelling of the eyelid
•
Itching
In addition to the side effects for LUMIGAN 0.1 mg/ml, the following side effects have been seen with
another medicine containing a higher strength of bimatoprost (0.3 mg/ml):
An allergic reaction in the eye; sensitivity to light; darker eyelashes; pain; a feeling that something is in
your eye; sticky eyes; difficulty in seeing clearly; burning; tears; dryness; an increase in blood-test
results that show how your liver is working, increased blood pressure; cystoid macular oedema
(swelling of the retina within the eye leading to worsening vision); inflammation within the eye; retinal
bleeding.
If any of the side effects get serious, or if you notice any side effects not mentioned in this leaflet,
please tell your doctor or pharmacist.
HOW TO STORE LUMIGAN 0.1 mg/ml
Keep out of the reach and sight of children.
Do not use LUMIGAN after the expiry date which is stated on the bottle label and the carton after
EXP. The expiry date refers to the last day of that month.
You must throw away the bottle at the latest four weeks after you first opened it, even if there are still
some drops left. This will prevent infections. To help you remember, write down the date you opened
it in the space on the box.
Medicines should not be disposed of via wastewater or household waste. Ask your pharmacist how to
dispose of medicines no longer required. These measures will help to protect the environment.
What LUMIGAN 0.1 mg/ml contains
The other ingredients are benzalkonium chloride (preservative), sodium chloride, sodium
phosphate dibasic heptahydrate, citric acid monohydrate and purified water. Small amounts of
hydrochloric acid or sodium hydroxide may be added to keep the level of acid (pH levels)
normal.
What LUMIGAN 0.1 mg/ml looks like and contents of the pack
LUMIGAN is a colourless to slightly yellow, clear eye drop solution in a pack containing either
1 plastic bottle or 3 plastic bottles each with a screw cap. Each bottle is approximately half full and
contains 3 millilitres of solution. This is enough for 4 weeks’ usage. Not all pack sizes may be
marketed.
Marketing Authorisation Holder and Manufacturer
Allergan Pharmaceuticals Ireland
Castlebar Road
Westport
Co. Mayo
Ireland.
For any information about this medicine, please contact the local representative of the marketing
authorisation holder.
België/Belgique/Belgien
Allergan NV/SA
Terhulpsesteenweg 6D
B-1560 Hoeilaart
Tél/Tel: + 32 (0)2 351 24 24
E-mail: uk_medinfo@allergan.com
Luxembourg/Luxemburg
Allergan NV/SA
Terhulpsesteenweg 6D
B-1560 Hoeilaart
Belgique/Belgien
Tél/Tel: + 32 (0)2 351 24 24
E-mail: uk_medinfo@allergan.com
България
Eвофарма АГ
Представителство ул. Персенк 73, ап. 27, ет. 8
1164 София
Тел.: +359 2 962 12 00
E-mail: uk_medinfo@allergan.com
Magyarország
Ewopharma AG Magyarországi Kereskedelmi
Képviselete
1021 Budapest
Budakeszi út 73/F
Hungary
Tel: +36 1 200 4650
E-mail: uk_medinfo@allergan.com
Česká republika
NEOMED s.r.o.
Sodomkova 1474/6, Praha 10
CZ-102 00
Tel.: +420 274 008 411
E-mail: uk_medinfo@allergan.com
Malta
Allergan Ltd
1st Floor
Marlow International
The Parkway
Marlow
Bucks, SL7 1YL-UK
United Kingdom/Renju Unit
Tel: + 44 (0)1628 494026
E-mail: uk_medinfo@allergan.com
Danmark
Allergan Norden AB
The active substance is bimatoprost. One ml of solution contains 0.1 mg bimatoprost.
Johanneslundsvägen 3-5
S-194 81 Upplands Väsby
Sverige
Tlf: + 46 (0)8 594 100 00
E-mail: uk_medinfo@allergan.com
Terhulpsesteenweg 6D
B-1560 Hoeilaart
België
Tel: + 32 (0)2 351 24 24
E-mail: uk_medinfo@allergan.com
Deutschland
Pharm-Allergan GmbH
Pforzheimer Straße 160
D-76275 Ettlingen
Tel: + 49 (0)7243 501 0
E-mail: uk_medinfo@allergan.com
Norge
Allergan Norden AB
Johanneslundsvägen 3-5
S-194 81 Upplands Väsby
Sverige
Tlf: + 46 (0)8 594 100 00
E-mail: uk_medinfo@allergan.com
Eesti
Allergan Ltd
1st Floor
Marlow International
The Parkway
Marlow
Bucks, SL7 1YL-UK
Ühendkuningriik
Tel: + 44 (0)1628 494026
E-mail: uk_medinfo@allergan.com
Österreich
Pharm-Allergan GmbH
Pforzheimer Straße 160
D-76275 Ettlingen
Deutschland
Tel: + 49 (0)7243 501 0
E-mail: uk_medinfo@allergan.com
Ελλάδα
Nexus Medicals S.A.
Λεωφ. Μαρκοπούλου - Σουνίου
Θέση Βγέντζι
GR-190 03 Μαρκόπουλο Μεσογαίας - Αττική
Tηλ: +30 22990.41350
E-mail: uk_medinfo@allergan.com
Polska
Allergan Sp. z o.o.
Aleje Jerozolimskie 94
00-807 Warszawa
Tel: +48 22 256 37 00
Fax: +48 22 256 37 01
E-mail: uk_medinfo@allergan.com
España
Allergan S.A.U
Edificio la Encina
Plaza de la Encina, 10-11
E-28760 Tres Cantos
Madrid
Tel: + 34 91 807 6130
E-mail: uk_medinfo@allergan.com
Portugal
Profarin Lda.
Rua da Quinta dos Grilos, 30
P-2790-476 Carnaxide
Tel : + 351 21 425 3242
E-mail: uk_medinfo@allergan.com
France
Allergan France SAS
12, Place de la Défense
92400 Courbevoie
Tel : +33 (0)1 49 07 83 00
E -mail: uk_medinfo@allergan.com
România
Ewopharma AG România
B-dul Primăverii, nr. 19-21,
Bucureşti 011972-RO
Tel.: +40 21 260 13 44
E-mail: uk_medinfo@allergan.com
Ireland
Allergan Ltd
1st Floor
Marlow International
The Parkway
Marlow
Bucks, SL7 1YL-UK
United Kingdom
Tel: + 44 (0)1628 494026
E-mail: uk_medinfo@allergan.com
Slovenija
Ewopharma d.o.o.
Cesta 24. junija 23
SI-1231 Ljubljana – Črnuče
Tel: + 386 (0) 590 848 40
E-mail: uk_medinfo@allergan.com
Ísland
Vistor hf.
Hörgatún 2
IS-212 Garðabær
Sími: + 354 535 7000
Netfang: uk_medinfo@allergan.com
Slovenská republika
NEOMED,s.r.o., pobočka Bratislava
Šťastná 11
SK-821 05 Bratislava
Tel.: +421 2 434 150 12
E-mail: uk_medinfo@allergan.com
Italia
Allergan S.p.A
Via S.Quasimodo 134/138
I-00144 Roma
Tel: + 39 06 509 561
E-mail: uk_medinfo@allergan.com
Suomi/Finland
Allergan Norden AB
Johanneslundsvägen 3-5
S-194 81 Upplands Väsby
Ruotsi/Sverige
Puh/Tel: + 46 (0)8 594 100 00
E-mail: uk_medinfo@allergan.com
Κύπρος
Allergan Ltd
1st Floor
Marlow International
The Parkway
Marlow
Bucks, SL7 1YL-UK
Ηνωμένο Βασίλειο
Τηλ: + 44 (0)1628 494026
E-mail: uk_medinfo@allergan.com
Sverige
Allergan Norden AB
Johanneslundsvägen 3-5
S-194 81 Upplands Väsby
Tel: +46 (0)8 594 100 00
E-mail: uk_medinfo@allergan.com
Latvija
Allergan Ltd
1st Floor
Marlow International
The Parkway
Marlow
Bucks, SL7 1YL-UK
Lielbritānija
Tel: + 44 (0)1628 494026
E-mail: uk_medinfo@allergan.com
United Kingdom
Allergan Ltd
1st Floor
Marlow International
The Parkway
Marlow
Bucks, SL7 1YL-UK
Tel: + 44 (0)1628 494026
E-mail: uk_medinfo@allergan.com
Lietuva
Allergan Ltd
1st Floor
Marlow International
The Parkway
Marlow
Bucks, SL7 1YL-UK
Jungtinė Karalystė
Tel. + 44 (0)1628 494026
E-mail: uk_medinfo@allergan.com
This leaflet was last approved on
Detailed inform
ation on this medicinal product is available on the website of the European Medicines
Agency (EMEA) http://www.emea.europa.eu/
PACKAGE LEAFLET: INFORMATION FOR THE USER
LUMIGAN 0.3 mg/ml, eye drops, solution
Bimatoprost
Read all of this leaflet carefully before you start using this medicine.
-
Keep this leaflet. You may need to read it again.
This medicine has been prescribed for you. Do not pass it on to others. It may harm them, even
if their symptoms are the same as yours.
If any of the side effects get serious, or if you notice any side effects not listed in this leaflet
please tell your doctor or pharmacist.
In this leaflet
:
1. What LUMIGAN 0.3 mg/ml is and what it is used for
2. Before you use LUMIGAN 0.3 mg/ml
3. How to use LUMIGAN 0.3 mg/ml
4. Possible side effects
7.
How to store LUMIGAN 0.3 mg/ml
8.
Further information
1.
WHAT
LUMIGAN 0.3 mg/ml
IS AND WHAT IT IS USED FOR
LUMIGAN is an antiglaucoma preparation. It belongs to a group of medicines called prostamides.
LUMIGAN eye drops are used to reduce high pressure in the eye. This high pressure can lead to a
disease called glaucoma. If the high pressure is not reduced, it could eventually damage your sight.
Your eye contains a clear, watery liquid that feeds the inside of the eye. Liquid is constantly being
drained out of the eye and new liquid is made to replace this. If the liquid cannot drain out quickly
enough, the pressure inside the eye builds up. LUMIGAN works by increasing the amount of liquid
that is drained. This reduces the pressure inside the eye.
LUMIGAN may be used on its own or with other drops called beta-blockers which also reduce
pressure.
2.
BEFORE YOU USE LUMIGAN 0.3 mg/ml
Do not use LUMIGAN 0.3 mg/ml:
-
if you are allergic (hypersensitive) to bimatoprost or any of the other ingredients of LUMIGAN.
Take special care with LUMIGAN 0.3 mg/ml:
-
Talk to your doctor, if:
-
You have any breathing problems.
You have liver or kidney problems.
You have had a cataract surgery in the past
You wear contact lenses (see “Important information about some of the ingredients of
LUMIGAN 0.3 mg/ml”)
You have or have had low blood pressure or low heart rate
You have had a viral infection or inflammation of the eye
If you have further questions, please ask your doctor or your pharmacist.
LUMIGAN may cause your eyelashes to darken and grow, and cause the skin around the eyelid to
darken too. The colour of your iris may also go darker over time. These changes may be permanent.
The change may be more noticeable if you are only treating one eye.
LUMIGAN has not been tested in children under the age of 18 and therefore LUMIGAN must not be
used by patients under 18 years.
Using other medicines:
Please tell your doctor or pharmacist if you are taking, or have recently taken, any other medicines,
including medicines obtained without a prescription.
If you use LUMIGAN with another eye drop, leave at least five minutes between putting in
LUMIGAN and then the other drops.
Pregnancy and breast feeding
Ask your doctor or pharmacist for advice before taking any medicine. LUMIGAN should not be used
during pregnancy unless your doctor still recommends it.
LUMIGAN may get into breast milk so you should not breast-feed while you are taking LUMIGAN.
Driving and using machines:
Your sight may become blurred for a short time just after using LUMIGAN. You should not drive or
use machines until your sight is clear again.
Important information about some of the ingredients of LUMIGAN 0.3 mg/ml
Do not use the drops when you are wearing your lenses. Wait 15 minutes after using the drops before
you put your lenses back in. A preservative in LUMIGAN called benzalkonium chloride may cause
eye irritation and can discolour soft contact lenses.
HOW TO USE LUMIGAN 0.3 mg/ml
LUMIGAN should only be applied to the eye. Always use LUMIGAN exactly as your doctor has told
you. You should check with your doctor or pharmacist if you are not sure. The usual dose is one drop
of LUMIGAN in the evening, once daily in each eye that needs treatment.
If you use LUMIGAN with another eye medicine, apply the LUMIGAN eye drop first and wait
5 minutes before applying the second eye medicine.
You must not use the bottle if the tamper-proof seal on the bottle neck is broken before you first use it.
1.
Wash your hands. Tilt your head back and look at the ceiling.
2.
Gently pull down the lower eyelid until there is a small pocket.

3.
Turn the bottle upside down and squeeze it to release one drop into each eye that needs
treatment.
4.
Let go of the lower lid, and close your eye for 30 seconds.
If a drop misses your eye, try again.
To help prevent infections, do not let the tip of the bottle touch your eye or anything else. Put the cap
back on and close the bottle straight after you have used it.
If you use more LUMIGAN 0.3 mg/ml than you should
If you use more LUMIGAN than you should, it is unlikely to cause you any serious harm. Put your
next dose in at the usual time. If you are worried, talk to your doctor or pharmacist.
If you forget to use LUMIGAN 0.3 mg/ml
If you forget to use LUMIGAN,
use a single drop as soon as you remember, and then go back to your
regular routine. Do not use a double dose to make up for a forgotten dose.
If you stop using LUMIGAN 0.3 mg/ml
LUMIGAN should be used every day to work properly. If you stop using LUMIGAN the pressure
inside your eye may go up, therefore talk to your doctor before stopping this treatment.
If you have any further questions on the use of this product, ask your doctor or pharmacist.
Like all medicines, LUMIGAN 0.3 mg/ml can have side effects, although not everybody gets them.
Most of the side effects are not serious.
Very common side effects
These may affect one or more users in 10
Affecting the eye
•
Longer eyelashes (up to 45% of people)
Slight redness (up to 44% of people)
Itchiness (up to 14% of people)
Common side effects
These may affect 1 to 9 users in 100
Affecting the eye
•
An allergic reaction in the eye
Darker skin colour around the eye
A feeling that something is in your eye
Difficulty in seeing clearly
Inflamed, red and itchy eyelids
Swelling of the see-through layer which covers the surface of the eye
Small breaks in the surface of the eye, with or without inflammation
An increase in blood-test results that show how your liver is working
Uncommon side effects
These may affect 1 to 9 users in 1000
Affecting the eye
•
Cystoid macular oedema (swelling of the retina within the eye leading to worsening vision)
Inflammation within the eye
Side effects where the occurrence is unknown
If any of the side effects get serious, or if you notice any side effects not mentioned in this leaflet,
please tell your doctor or pharmacist.
HOW TO STORE LUMIGAN 0.3 mg/ml
Keep out of the reach and sight of children.
Do not use LUMIGAN after the expiry date which is stated on the bottle label and the carton after
EXP:. The expiry date refers to the last day of that month.
You must throw away the bottle, at the latest, four weeks after you first opened it, even if there are still
some drops left. This will prevent infections. To help you remember, write down the date you opened
it in the space on the box.
Medicines should not be disposed of via wastewater or household waste. Ask your pharmacist how to
dispose of medicines no longer required. These measures will help to protect the environment.
What LUMIGAN 0.3 mg/ml contains
-
The active substance is Bimatoprost. One ml of solution contains 0.3 mg bimatoprost.
The other ingredients are benzalkonium chloride (preservative), sodium chloride, sodium
phosphate dibasic heptahydrate, citric acid monohydrate and purified water. Small amounts of
hydrochloric acid or sodium hydroxide may be added to keep the level of acid (pH levels)
normal.
What LUMIGAN 0.3 mg/ml looks like and contents of the pack
LUMIGAN is a colourless to slightly yellow, clear eye drop solution in a pack containing either
1 plastic bottle or 3 plastic bottles each with a screw cap. Each bottle is approximately half full and
contains 3 millilitres of solution. This is enough for 4 weeks’ usage. Not all pack sizes may be
marketed.
Marketing Authorisation Holder and Manufacturer
Allergan Pharmaceuticals Ireland
Castlebar Road
Westport
Co. Mayo
Ireland.
For any information about this medicine, please contact the local representative of the marketing
authorisation holder.
België/Belgique/Belgien
Allergan NV/SA
Terhulpsesteenweg 6D
B-1560 Hoeilaart
Tél/Tel: + 32 (0)2 351 24 24
E-mail: uk_medinfo@allergan.com
Luxembourg/Luxemburg
Allergan NV/SA
Terhulpsesteenweg 6D
B-1560 Hoeilaart
Belgique/Belgien
Tél/Tel: + 32 (0)2 351 24 24
E-mail: uk_medinfo@allergan.com
България
Eвофарма АГ
Представителство ул. Персенк 73, ап. 27, ет. 8
1164 София
Тел.: +359 2 962 12 00
E-mail: uk_medinfo@allergan.com
Magyarország
Ewopharma AG Magyarországi Kereskedelmi
Képviselete
1021 Budapest
Budakeszi út 73/F
Hungary
Tel: +36 1 200 4650
E-mail: uk_medinfo@allergan.com
Česká republika
NEOMED s.r.o.
Sodomkova 1474/6, Praha 10
CZ-102 00
Tel.: +420 274 008 411
E-mail: uk_medinfo@allergan.com
Malta
Allergan Ltd
1st Floor
Marlow International
The Parkway
Marlow
Bucks, SL7 1YL-UK
United Kingdom/Renju Unit
Tel: + 44 (0)1628 494026
E-mail: uk_medinfo@allergan.com
Danmark
Allergan Norden AB
Johanneslundsvägen 3-5
S-194 81 Upplands Väsby
Sverige
Tlf: + 46 (0)8 594 100 00
E-mail: uk_medinfo@allergan.com
Nederland
Allergan n.v.
Terhulpsesteenweg 6D
B-1560 Hoeilaart
België
Tel: + 32 (0)2 351 24 24
E-mail: uk_medinfo@allergan.com
Deutschland
Pharm-Allergan GmbH
Pforzheimer Straße 160
D-76275 Ettlingen
Tel: + 49 (0)7243 501 0
E-mail: uk_medinfo@allergan.com
Norge
Allergan Norden AB
Johanneslundsvägen 3-5
S-194 81 Upplands Väsby
Sverige
Tlf: + 46 (0)8 594 100 00
E-mail: uk_medinfo@allergan.com
Eesti
Allergan Ltd
1st Floor
Marlow International
The Parkway
Marlow
Bucks, SL7 1YL-UK
Ühendkuningriik
Tel: + 44 (0)1628 494026
E-mail: uk_medinfo@allergan.com
Österreich
Pharm-Allergan GmbH
Pforzheimer Straße 160
D-76275 Ettlingen
Deutschland
Tel: + 49 (0)7243 501 0
E-mail: uk_medinfo@allergan.com
Ελλάδα
Nexus Medicals S.A.
Λεωφ. Μαρκοπούλου - Σουνίου
Θέση Βγέντζι
GR-190 03 Μαρκόπουλο Μεσογαίας - Αττική
Tηλ: +30 22990.41350
E-mail: uk_medinfo@allergan.com
Polska
Allergan Sp. z o.o.
Aleje Jerozolimskie 94
00-807 Warszawa
Tel: +48 22 256 37 00
Fax: +48 22 256 37 01
E-mail: uk_medinfo@allergan.com
España
Allergan S.A.U
Edificio la Encina
Plaza de la Encina, 10-11
E-28760 Tres Cantos
Madrid
Tel: + 34 91 807 6130
E-mail: uk_medinfo@allergan.com
Portugal
Profarin Lda.
Rua da Quinta dos Grilos, 30
P-2790-476 Carnaxide
Tel : + 351 21 425 3242
E-mail: uk_medinfo@allergan.com
France
Allergan France SAS
12, Place de la Défense
92400 Courbevoie
Tel : +33 (0)1 49 07 83 00
E-mail: uk_medinfo@allergan.com
România
Ewopharma AG România
B-dul Primăverii, nr. 19-21,
Bucureşti 011972-RO
Tel.: +40 21 260 13 44
E-mail: uk_medinfo@allergan.com
Ireland
Allergan Ltd
1st Floor
Marlow International
The Parkway
Marlow
Bucks, SL7 1YL-UK
United Kingdom
Tel: + 44 (0)1628 494026
E-mail: uk_medinfo@allergan.com
Slovenija
Ewopharma d.o.o.
Cesta 24. junija 23
SI-1231 Ljubljana – Črnuče
Tel: + 386 (0) 590 848 40
E-mail: uk_medinfo@allergan.com
Ísland
Vistor hf.
Hörgatúni 2
IS-212 Garðabæ
Sími: + 354 535 7000
Netfang: uk_medinfo@allergan.com
Slovenská republika
NEOMED,s.r.o., pobočka Bratislava
Šťastná 11
SK-821 05 Bratislava
Tel.: +421 2 434 150 12
E-mail: uk_medinfo@allergan.com
Italia
Allergan S.p.A
Via S.Quasimodo 134/138
I-00144 Roma
Tel: + 39 06 509 561
E-mail: uk_medinfo@allergan.com
Suomi/Finland
Allergan Norden AB
Johanneslundsvägen 3-5
S-194 81 Upplands Väsby
Ruotsi/Sverige
Puh/Tel: + 46 (0)8 594 100 00
E-mail: uk_medinfo@allergan.com
Κύπρος
Allergan Ltd
1st Floor
Marlow International
The Parkway
Marlow
Bucks, SL7 1YL-UK
Ηνωμένο Βασίλειο
Τηλ: + 44 (0)1628 494026
E-mail: uk_medinfo@allergan.com
Sverige
Allergan Norden AB
Johanneslundsvägen 3-5
S-194 81 Upplands Väsby
Tel: +46 (0)8 594 100 00
E-mail: uk_medinfo@allergan.com
Latvija
Allergan Ltd
1st Floor
Marlow International
The Parkway
Marlow
Bucks, SL7 1YL-UK
Lielbritānija
Tel: + 44 (0)1628 494026
E-mail: uk_medinfo@allergan.com
United Kingdom
Allergan Ltd
1st Floor
Marlow International
The Parkway
Marlow
Bucks, SL7 1YL-UK
Tel: + 44 (0)1628 494026
E-mail: uk_medinfo@allergan.com
Lietuva
Allergan Ltd
1st Floor
Marlow International
The Parkway
Marlow
Bucks, SL7 1YL-UK
Jungtinė Karalystė
Tel. + 44 (0)1628 494026
E-mail: uk_medinfo@allergan.com
This leaflet was last approved on
Detailed information on this medicinal product is available on the website of the European Medicines
Agency (EMEA) http://www.emea.europa.eu/
Source: European Medicines Agency
 - Please bookmark this page (add it to your favorites).
- Please bookmark this page (add it to your favorites).
- If you wish to link to this page, you can do so by referring to the URL address below this line.
https://theodora.com/drugs/eu/lumigan.html
Copyright © 1995-2021 ITA all rights reserved.
|



































































































































































































































































 - Please bookmark this page (add it to your favorites).
- Please bookmark this page (add it to your favorites).










































