Product Characteristics
ANNEX I
SUMMARY OF PRODUCT CHARACTERISTICS
NAME OF THE MEDICINAL PRODUCT
Simponi 50 mg solution for injection in pre-filled pen.
QUALITATIVE AND QUANTITATIVE COMPOSITION
One 0.5 ml pre-filled pen contains 50 mg of golimumab*.
* Human IgG1κ monoclonal antibody produced by a murine hybridoma cell line with recombinant
DNA technology.
Excipient:
Each pre-filled pen contains 20.5 mg sorbitol per 50 mg dose.
For a full list of excipients, see section 6.1.
Solution for injection in pre-filled pen (injection), SmartJect
The solution is clear to slightly opalescent, colourless to light yellow.
4.1 Therapeutic indications
Rheumatoid arthritis (RA)
Simponi, in combination with methotrexate (MTX), is indicated for:
•
the treatment of moderate to severe, active rheumatoid arthritis in adults when the response to
disease-modifying anti-rheumatic drug (DMARD) therapy including MTX has been
inadequate.
the treatment of severe, active and progressive rheumatoid arthritis in adults not previously
treated with MTX.
Simponi, in combination with MTX, has been shown to reduce the rate of progression of joint
damage as measured by X-ray and to improve physical function.
Psoriatic arthritis (PsA)
Simponi, alone or in combination with MTX, is indicated for the treatment of active and progressive
psoriatic arthritis in adults when the response to previous disease-modifying anti-rheumatic drug
(DMARD) therapy has been inadequate. Simponi has also been shown to improve physical function
in this patient population.
Ankylosing spondylitis (AS)
Simponi is indicated for the treatment of severe, active ankylosing spondylitis in adults who have
responded inadequately to conventional therapy
4.2
Posology and method of administration
Simponi treatment is to be initiated and supervised by qualified physicians experienced in the
diagnosis and treatment of rheumatoid arthritis, psoriatic arthritis or ankylosing spondylitis. Patients
treated with Simponi should be given the Patient Alert Card.
Rheumatoid arthritis
Simponi 50 mg given once a month, on the same date each month.
Simponi should be given concomitantly with MTX.
Psoriatic arthritis
Simponi 50 mg given once a month, on the same date each month.
Ankylosing spondylitis
Simponi 50 mg given once a month, on the same date each month.
Available data suggest that clinical response is usually achieved within 12 to 14 weeks of treatment
(after 3-4 doses). Continued therapy should be reconsidered in patients who show no evidence of
therapeutic benefit within this time period.
In patients weighing more than 100 kg who do not achieve an adequate clinical response after 3 or 4
doses, increasing the dose of golimumab to 100 mg once a month may be considered, taking into
account the increased risk of certain serious adverse drug reactions with the 100 mg dose compared
with the 50 mg dose (see section 4.8). Continued therapy should be reconsidered in patients who
show no evidence of therapeutic benefit after receiving 3 to 4 additional doses of 100 mg.
Missed dose
If a patient forgets to inject Simponi on the planned date, the forgotten dose should be injected as
soon as the patient remembers. Patients should be instructed not to inject a double dose to make up
for the forgotten dose.
The next dose should be administered based on the following guidance:
•
if the dose is less than 2 weeks late, the patient should inject his/her forgotten dose and stay
on his/her original monthly schedule.
•
if the dose is more than 2 weeks late, the patient should inject his/her forgotten dose and a
new once-monthly schedule should be established from the date of this injection.
Elderly patients (≥ 65 years)
No dose adjustment is required in the elderly.
Renal and hepatic impairment
Simponi has not been studied in these patient populations. No dose recommendations can be made.
Paediatric population
The safety and efficacy of Simponi in patients aged less than 18 have not yet been established. No
data are available.
Method of administration
For subcutaneous use. After proper training in subcutaneous injection technique, patients may
self-inject with Simponi if their physician determines that this is appropriate, with medical follow-up
as necessary. Patients should be instructed to inject the full amount of Simponi according to the
comprehensive instructions for administration provided in the package leaflet. For administration
instructions, see section 6.6.
Hypersensitivity to the active substance or to any of the excipients (see section 6.1).
Active tuberculosis (TB) or other severe infections such as sepsis, and opportunistic infections
(see section 4.4).
Moderate or severe heart failure (NYHA class III/IV) (see section 4.4).
4.4 Special warnings and precautions for use
Infections
Patients must be monitored closely for infections including tuberculosis before, during and after
treatment with Simponi. Because the elimination of golimumab may take up to 5 months, monitoring
should be continued throughout this period. Further treatment with Simponi must not be given if a
patient develops a serious infection or sepsis (see section 4.3).
Simponi should not be given to patients with a clinically important, active infection. Caution should
be exercised when considering the use of Simponi in patients with a chronic infection or a history of
recurrent infection. Patients should be advised of, and avoid exposure to, potential risk factors for
infection as appropriate.
Patients taking TNF-blockers are more susceptible to serious infections.
Bacterial (including sepsis and pneumonia), mycobacterial (including TB), invasive fungal and
opportunistic infections, including fatalities, have been reported in patients receiving Simponi. Some
of these serious infections have occurred in patients on concomitant immunosuppressive therapy that,
in addition to their underlying disease, could predispose them to infections. Patients who develop a
new infection while undergoing treatment with Simponi should be monitored closely and undergo a
complete diagnostic evaluation. Administration of Simponi should be discontinued if a patient
develops a new serious infection or sepsis, and appropriate antimicrobial or antifungal therapy should
be initiated until the infection is controlled. For patients who have resided in or travelled to regions
where invasive fungal infections such as histoplasmosis, coccidioidomycosis, or blastomycosis are
endemic, the benefits and risks of Simponi treatment should be carefully considered before initiation
of Simponi therapy.
Tuberculosis
There have been reports of tuberculosis in patients receiving Simponi. It should be noted that in the
majority of these reports, tuberculosis was extrapulmonary presenting as either local or disseminated
disease.
Before starting treatment with Simponi, all patients must be evaluated for both active and inactive
(‘latent’) tuberculosis. This evaluation should include a detailed medical history with personal history
of tuberculosis or possible previous contact with tuberculosis and previous and/or current
immunosuppressive therapy. Appropriate screening tests, i.e. tuberculin skin or blood test and chest
X-ray, should be performed in all patients (local recommendations may apply). It is recommended
that the conduct of these tests should be recorded in the patient’s alert card. Prescribers are reminded
of the risk of false negative tuberculin skin test results, especially in patients who are severely ill or
immunocompromised.
If active tuberculosis is diagnosed, Simponi therapy must not be initiated (see section 4.3).
If latent tuberculosis is suspected, a physician with expertise in the treatment of tuberculosis should
be consulted. In all situations described below, the benefit/risk balance of Simponi therapy should be
very carefully considered.
If inactive (‘latent’) tuberculosis is diagnosed, treatment for latent tuberculosis must be started with
anti-tuberculosis therapy before the initiation of Simponi, and in accordance with local
recommendations.
In patients who have several or significant risk factors for tuberculosis and have a negative test for
latent tuberculosis, anti-tuberculosis therapy should be considered before the initiation of Simponi.
Use of anti-tuberculosis therapy should also be considered before the initiation of Simponi in patients
with a past history of latent or active tuberculosis in whom an adequate course of treatment cannot be
confirmed.
All patients should be informed to seek medical advice if signs/symptoms suggestive of tuberculosis
(e.g. persistent cough, wasting/weight loss, low-grade fever) appear during or after Simponi
treatment.
Hepatitis B virus reactivation
Reactivation of hepatitis B has occurred in patients receiving a TNF-antagonist including Simponi,
who are chronic carriers of this virus (i.e., surface antigen positive). Some cases have had fatal
outcome.
Patients should be tested for HBV infection before initiating treatment with Simponi. For patients
who test positive for HBV infection, consultation with a physician with expertise in the treatment of
hepatitis B is recommended.
Carriers of HBV who require treatment with Simponi should be closely monitored for signs and
symptoms of active HBV infection throughout therapy and for several months following termination
of therapy. Adequate data of treating patients who are carriers of HBV with anti-viral therapy in
conjunction with TNF-antagonist therapy to prevent HBV reactivation are not available. In patients
who develop HBV reactivation, Simponi should be stopped and effective anti-viral therapy with
appropriate supportive treatment should be initiated.
Malignancies and lymphoproliferative disorders
The potential role of TNF-blocking therapy in the development of malignancies is not known. Based
on the current knowledge, a possible risk for the development of lymphomas, leukaemia or other
malignancies in patients treated with a TNF-antagonist cannot be excluded. Caution should be
exercised when considering TNF-blocking therapy for patients with a history of malignancy or when
considering continuing treatment in patients who develop malignancy.
Paediatric malignancy
Malignancies, some fatal, have been reported among children, adolescents and young adults (up to 22
years of age) treated with TNF-blocking agents (initiation of therapy ≤ 18 years of age) in the post
marketing setting. Approximately half the cases were lymphomas. The other cases represented a
variety of different malignancies and included rare malignancies usually associated with
immunosuppression. A risk for the development of malignancies in children and adolescents treated
with TNF-blockers cannot be excluded.
Lymphoma and leukaemia
In the controlled portions of clinical trials of all the TNF-blocking agents including Simponi, more
cases of lymphoma have been observed among patients receiving anti-TNF treatment compared with
control patients. During the Simponi Phase IIb and Phase III clinical trials, the incidence of
lymphoma in Simponi-treated patients was higher than expected in the general population. In the
post-marketing setting, cases of leukaemia have been reported in patients treated with a TNF-
antagonist. There is an increased background risk for lymphoma and leukaemia in rheumatoid
arthritis patients with long-standing, highly active, inflammatory disease, which complicates risk
estimation.
Malignancies other than lymphoma
In the controlled portions of the Simponi Phase IIb and Phase III clinical trials in RA, PsA, and AS,
the incidence of non-lymphoma malignancies (excluding non-melanoma skin cancer) was similar
between the Simponi and the control groups.
In an exploratory clinical trial evaluating the use of Simponi in patients with severe persistent
asthma, more malignancies were reported in patients treated with Simponi compared with control
patients (see section 4.8). The significance of this finding is unknown.
In an exploratory clinical trial evaluating the use of another anti-TNF agent, infliximab, in patients
with moderate to severe chronic obstructive pulmonary disease (COPD), more malignancies, mostly
in the lung or head and neck, were reported in infliximab-treated patients compared with control
patients. All patients had a history of heavy smoking. Therefore, caution should be exercised when
using any TNF-antagonist in COPD patients, as well as in patients with an increased risk of
malignancy due to heavy smoking.
Congestive heart failure (CHF)
Cases of worsening congestive heart failure (CHF) and new onset CHF have been reported with TNF
blockers, including Simponi. In a clinical trial with another TNF-antagonist worsening congestive
heart failure and increased mortality due to CHF have been observed. Simponi has not been studied
in patients with CHF. Simponi should be used with caution in patients with mild heart failure
(NYHA class I/II). Patients should be closely monitored and Simponi must be discontinued in
patients who develop new or worsening symptoms of heart failure (see section 4.3).
Neurological events
Use of TNF-blocking agents, including Simponi, has been associated with cases of new onset or
exacerbation of clinical symptoms and/or radiographic evidence of central nervous system
demyelinating disorders, including multiple sclerosis and peripheral demyelinating disorders. In
patients with pre-existing or recent onset of demyelinating disorders, the benefits and risks of
anti-TNF treatment should be carefully considered before initiation of Simponi therapy.
Discontinuation of Simponi should be considered if these disorders develop.
Surgery
There is limited safety experience of Simponi treatment in patients who have undergone surgical
procedures, including arthroplasty. The long half-life should be taken into consideration if a surgical
procedure is planned. A patient who requires surgery while on Simponi should be closely monitored
for infections, and appropriate actions should be taken.
Immunosuppression
The possibility exists for TNF-blocking agents, including Simponi, to affect host defences against
infections and malignancies since TNF mediates inflammation and modulates cellular immune
responses.
Autoimmune processes
The relative deficiency of TNF
α
caused by anti-TNF therapy may result in the initiation of an
autoimmune process. If a patient develops symptoms suggestive of a lupus-like syndrome following
treatment with Simponi and is positive for antibodies against double-stranded DNA, treatment with
Simponi should be discontinued (see section 4.8).
Haematologic reactions
There have been post-marketing reports of pancytopaenia, leucopaenia, neutropaenia, aplastic
anaemia, and thrombocytopaenia in patients receiving TNF-blockers. Cytopaenias including
pancytopaenia have been infrequently reported with Simponi in clinical trials. All patients should be
advised to seek immediate medical attention if they develop signs and symptoms suggestive of blood
dyscrasias (e.g. persistent fever, bruising, bleeding, pallor). Discontinuation of Simponi therapy
should be considered in patients with confirmed significant haematologic abnormalities.
Concurrent administration of TNF-antagonists and anakinra
Serious infections and neutropenia were seen in clinical studies with concurrent use of anakinra and
another TNF-blocking agent, etanercept, with no added clinical benefit. Because of the nature of the
adverse events seen with this combination therapy, similar toxicities may also result from the
combination of anakinra and other TNF-blocking agents. The combination of Simponi and anakinra
is not recommended.
Concurrent administration of TNF-antagonists and abatacept
In clinical studies concurrent administration of TNF-antagonists and abatacept has been associated
with an increased risk of infections including serious infections compared to TNF-antagonists alone,
without increased clinical benefit. The combination of Simponi and abatacept is not recommended.
Switching between biological DMARDs
When switching from one biologic to another, patients should be monitored for signs of infection.
Vaccinations
Patients treated with Simponi may receive concurrent vaccinations, except for live vaccines (see
section 4.5). No data are available on the response to vaccination, risk of infection or transmission of
infection with the administration of live vaccines to patients receiving Simponi.
Allergic reactions
In post-marketing experience, serious systemic hypersensitivity reactions (including anaphylactic
reaction) have been reported following Simponi administration. Some of these reactions occurred
after the first administration of Simponi. If an anaphylactic reaction or other serious allergic reactions
occur, administration of Simponi should be discontinued immediately and appropriate therapy
initiated.
Latex sensitivity
The needle cover on the pre-filled pen is manufactured from dry natural rubber containing latex, and
may cause allergic reactions in individuals sensitive to latex.
Elderly patients (≥ 65 years)
In the Phase III studies in RA, PsA, and AS, no overall differences in adverse events (AEs), serious
adverse events (SAEs) , and serious infections in patients age 65 or older (n=155) who received
Simponi were observed compared with younger patients. However, caution should be exercised when
treating the elderly and particular attention paid with respect to occurrence of infections.
Renal and hepatic impairment
Specific studies of Simponi have not been conducted in patients with renal or hepatic impairment.
Simponi should be used with caution in subjects with impaired hepatic function (see section 4.2).
Excipients
Simponi contains sorbitol (E420). Patients with rare hereditary problems of fructose intolerance
should not take Simponi.
4.5 Interaction with other medicinal products and other forms of interaction
No interaction studies have been performed.
Concurrent use with anakinra and abatacept
The combination of Simponi and anakinra or abatacept is not recommended (see section 4.4).
Live vaccines
Live vaccines should not be given concurrently with Simponi (see section 4.4).
Methotrexate
Although concomitant use of MTX results in higher steady-state trough concentrations of Simponi in
patients with RA, PsA or AS, the data do not suggest the need for dose adjustment of either Simponi
or MTX (see section 5.2).
4.6 Fertility, pregnancy and lactation
Women of childbearing potential
Women of childbearing potential must use adequate contraception to prevent pregnancy and continue
its use for at least 6 months after the last golimumab treatment.
Pregnancy
There are no adequate data on the use of golimumab in pregnant women. Due to its inhibition of
TNF, golimumab administered during pregnancy could affect normal immune responses in the
newborn. Studies in animals do not indicate direct or indirect harmful effects with respect to
pregnancy, embryonal/foetal development, parturition or postnatal development (see section 5.3).
The use of golimumab in pregnant women is not recommended; golimumab should be given to a
pregnant woman only if clearly needed.
Breastfeeding
It is not known whether golimumab is excreted in human milk or absorbed systemically after
ingestion. Golimumab was shown to pass over to breast milk in monkeys, and because human
immunoglobulins are excreted in milk, women must not breast feed during and for at least 6 months
after golimumab treatment.
Fertility
No animal fertility studies have been conducted with golimumab. A fertility study in mice, using an
analogous antibody that selectively inhibits the functional activity of mouse TNFα, showed no
relevant effects on fertility (see section 5.3).
4.7 Effects on ability to drive and use machines
Simponi may have a minor influence on the ability to drive and use machines. Dizziness may occur
following administration of Simponi (see section 4.8).
Upper respiratory tract infection was the most common adverse drug reaction (ADR) reported in the
controlled Phase III RA, PsA, and AS studies through week 16, occurring in 7.2% of golimumab-
treated patients as compared with 5.8% of control patients. The most serious ADRs that have been
reported for Simponi include serious infections (including sepsis, pneumonia, TB, invasive fungal
and opportunistic infections), demyelinating disorders, lymphoma, HBV reactivation, CHF,
autoimmune processes (lupus-like syndrome) and haematologic reactions (see section 4.4).
ADRs observed in clinical studies and reported from world-wide post-marketing use of golimumab
are listed in Table 1. Within the designated system organ classes, the adverse drug reactions are listed
under headings of frequency and using the following convention: Very common (≥ 1/10); Common
(≥ 1/100 to < 1/10); Uncommon (≥ 1/1,000 to < 1/100); Rare (≥ 1/10,000 to < 1/1,000); Very rare
(< 1/10,000); Not known (cannot be estimated from the available data).
Table 1
Tabulated list of ADRs
Infections and infestations
Very common: Upper respiratory tract infection (nasopharyngitis, pharyngitis,
laryngitis and rhinitis)
Common: Bacterial infections (such as cellulitis), viral infections (such as
influenza and herpes), bronchitis, sinusitis, superficial fungal
infections
Uncommon: Septic shock, sepsis, tuberculosis, lower respiratory tract
infection (such as pneumonia), opportunistic infections (such as
invasive fungal infections [histoplasmosis, coccidioidomycosis,
pneumocytosis], bacterial, atypical mycobacterial infection and
protozoal), pyelonephritis, abscess, bacterial arthritis, infective
bursitis
Rare: Hepatitis B reactivation
Neoplasms, benign, malignant and
unspecified
Uncommon: Neoplasms (such as skin cancer, squamous cell carcinoma and
melanocytic naevus)
Rare: Lymphoma
Not known: Leukaemia*
Blood and lymphatic system
disorders
Common: Anaemia
Uncommon: Leucopaenia, thrombocytopenia
Rare: Pancytopaenia
Not known: Aplastic anaemia *
Immune system disorders
Common: Allergic reactions (bronchospasm, hypersensitivity, urticaria),
autoantibody positive
Rare: Serious systemic hypersensitivity reactions (including
anaphylactic reaction), vasculitis (systemic)
Endocrine disorders
Uncommon: Thyroid disorder (such as hypothyroidism, hyperthyroidism and
goitre)
Metabolism and nutrition disorders
Uncommon: Blood glucose increased, lipids increased
Psychiatric disorders
Common: Depression, insomnia
Nervous system disorders
Common: Dizziness, paraesthesia, headache
Uncommon: Demyelinating disorders (central and peripheral), balance
disorders, dysguesia
Uncommon: Visual disorders (such as blurred vision and decreased visual
acuity), conjunctivitis, eye allergy (such as pruritis and
irritation)
Uncommon: Congestive heart failure (new onset or worsening), arrhythmia,
ischemic coronary artery disorders
Common: Hypertension
Uncommon: Thrombosis (such as deep venous and aortic), Raynaud’s
phenomenon, flushing
Respiratory, thoracic and mediastinal
disorders
Uncommon: Asthma and related symptoms (such as wheezing and bronchial
hyperactivity)













Rare: Interstitial lung disease
Gastrointestinal disorders
Common: Constipation, dyspepsia, gastrointestinal and abdominal pain
Uncommon: Gastrointestinal inflammatory disorders (such as gastritis and
colitis), gastro-oesophageal reflux disease, stomatitis
Hepatobiliary disorders
Common: Alanine aminotransferase increased, aspartate aminotransferase
increased
Uncommon: Cholelithiasis, hepatic disorders
Skin and subcutaneous tissue
disorders
Common: Alopecia, dermatitis, pruritus, rash
Uncommon: Psoriasis (new onset
or worsening of pre-existing psoriasis,
palmar/plantar and pustular), urticaria, vasculitis (cutaneous)
Musculoskeletal and connective
tissue disorders
Rare: Lupus-like syndrome
Renal and urinary disorders
Uncommon: Bladder disorders
Rare: Renal disorders
Reproductive system and breast
disorders
Uncommon: Breast disorders, menstrual disorders
General disorders and administration
site conditions
Common: Pyrexia, asthenia, injection site reaction (such as injection site
erythema, urticaria, induration, pain, bruising, pruritus, irritation
and paraesthesia), impaired healing, chest discomfort
Injury, poisoning and procedural
complications
Observed with other TNF-blocking agents, but not observed in clinical studies with golimumab.
Description of selected adverse drug reactions
Infections
Upper respiratory tract infection was the most common adverse reaction reported in the combined
Phase III RA, PsA, and AS studies through week 16, occurring in 7.2% of golimumab-treated
patients (incidence per patient-year: 0.26; 95% CI: 0.22, 0.31) as compared with 5.8% of control
patients (incidence per patient-year: 0.23; 95% CI: 0.17, 0.31). The incidence per patient year (95%
confidence interval; CI) of upper respiratory tract infections through 1 year of follow up was 0.23
events (0.21, 0.25) for golimumab-treated patients and 0.25 events (0.20, 0.31) for control patients.
In controlled Phase III trials through week 16 in RA, PsA, and AS, infections were observed in
28.3% of golimumab-treated patients (incidence per patient-year: 1.28; 95% CI: 1.18, 1.38)
compared with 24.7% of control patients (incidence per patient-year: 1.17; 95% CI: 1.02, 1.33). The
incidence per patient year (95% CI) of infections through 1 year of follow up was 1.32 events (1.27,
1.38) for golimumab-treated patients and 1.31 events (1.18, 1.44) for control patients.
In controlled Phase III trials through week 16 in RA, PsA, and AS, serious infections were observed
in 1.4% of golimumab-treated patients (incidence per patient-year: 0.06; 95% CI: 0.04, 0.08) and
1.3% of control patients (incidence per patient-year: 0.04; 95% CI: 0.02, 0.08). Serious infections
observed in golimumab-treated patients included tuberculosis, bacterial infections including sepsis
and pneumonia, invasive fungal infections and other opportunistic infections. Some of these
infections have been fatal. The incidence per patient year (95% CI) of serious infections through 1
year of follow up was 0.05 events (0.04, 0.06) for golimumab-treated patients and 0.06 events (0.04,
0.09) for control patients (see section 4.4). In the controlled and uncontrolled portions of the Phase II











RA and the Phase III RA, PsA, and AS trials with a mean follow-up of 1.6 years, a greater incidence
of TB was observed in the golimumab 100 mg treatment group compared with the golimumab 50 mg
group.
Lymphoma
The incidence of lymphoma in Simponi treated patients with RA, PsA and AS during the controlled
portions of phase IIb and III clinical trials and through 1 year of follow up was higher than expected
in the general population. Lymphoma was diagnosed in 2 subjects (both in golimumab 100 mg
treatment groups) with an incidence (95%, CI) per 100 subject-years of follow up of 0.10 (0.01, 0.37)
events for golimumab and 0.00 (0.00, 0.90) events for placebo. In the controlled and uncontrolled
portions of these trials through a median follow-up of 2.5 years, a greater incidence of lymphoma
was observed in the golimumab 100 mg treatment group compared with the golimumab 50 mg group.
The majority of lymphomas occurred in study GO-AFTER, which enrolled patients previously
exposed to anti-TNF agents who had longer disease duration and more refractory disease. See section
4.4.
Malignancies other than lymphoma
In the controlled portions of the Simponi Phase IIb and Phase III clinical trials in RA, PsA, and AS,
and through 1 year of follow up, the incidence of non-lymphoma malignancies (excluding
non-melanoma skin cancer) was similar between the Simponi and the control groups.
Through 1 year of follow up,of the Phase IIb and Phase III studies in rheumatologic indications,
nonmelanoma skin cancer was diagnosed in 19 subjects (5 in placebo, 6 in golimumab 50 mg and 8
in golimumab 100 mg treatment groups) with an incidence (95% CI) per 100 subject-years of follow
up of 0.72 (0.39, 1.20) events for golimumab and 1.51 (0.49, 3.52) events for placebo.
Through 1 year of follow up, of the Phase IIb and Phase III studies in rheumatologic indications,
malignancies besides nonmelanoma skin cancer and lymphoma were diagnosed in 12 subjects (2 in
placebo, 6 in golimumab 50 mg and 4 in golimumab 100 mg treatment groups) with an incidence
(95%CI) per 100 subject-years of follow up of 0.51 (0.24, 0.94) events for golimumab and 0.60
(0.07, 2.17) events for placebo. See section 4.4.
Cases reported in clinical studies in asthma
In an exploratory clinical study, patients with severe persistent asthma received a golimumab loading
dose (150% of the assigned treatment dose) subcutaneously at week 0 followed by golimumab 200
mg, golimumab 100 mg or golimumab 50 mg every 4 weeks subcutaneously through week 52. Eight
malignancies in the combined golimumab treatment group (n=230) and none in the placebo treatment
group (n=79). Lymphoma was reported in 1 patient, non-melanoma skin cancer in 2 patients, and
other malignancies in 5 patients. There was no specific clustering of any type of malignancy.
During the placebo-controlled portion of the study, the incidence (95% CI) of all malignancies per
100 subject-years of follow-up was 3.19 (1.38, 6.28) in the golimumab group. In this study, the
incidence (95% CI) per 100 subject-years of follow-up in golimumab-treated subjects was 0.40 (0.01,
2.20) for lymphoma, 0.79 (0.10, 2.86) for non-melanoma skin cancers, and 1.99 (0.64, 4.63) for other
malignancies. For placebo subjects, the incidence (95% CI) per 100 subject-years of follow-up of
these malignancies was 0.00 (0.00, 2.94). The significance of this finding is unknown.
Liver enzyme elevations
In controlled Phase III trials through week 16, mild ALT elevations (> 1 and < 3 x upper limit of
normal (ULN)) occurred in similar proportions of golimumab and control patients in the RA and PsA
studies (22.1% to 27.4% of patients); in the AS study, more golimumab-treated patients (25.6%) than
control patients (3.9 %) had mild ALT elevations. Through 1 year of follow-up the incidence of mild
ALT elevations was similar in golimumab-treated and control patients in RA and PsA studies. In the
AS population, the incidence of mild ALT elevations was higher in golimumab-treated patients than
in control patients.
In the RA and AS studies through week 16, ALT elevations ≥ 5 x ULN were uncommon and seen in
more golimumab-treated patients (0.4% to 0.9%) than control patients (0.0%). This trend was not
observed in the PsA population. Through 1 year of follow-up, the incidence of ALT elevations ≥5 x
ULN was similar in both golimumab-treated and control patients in the Phase III RA, PsA and AS
studies. In general these elevations were asymptomatic and the abnormalities decreased or resolved
with either continuation or discontinuation of golimumab or modification of concomitant
medications.
Within the Phase II and Phase III programme in RA, PsA and AS, one patient with pre-existing liver
abnormalities and confounding medication treated with golimumab developed non-infectious fatal
hepatitis with jaundice. The role of golimumab as a contributing or aggravation factor cannot be
excluded.
Injection site reactions
In controlled Phase III trials through week 16 in RA, PsA and AS, 5.8% of golimumab-treated
patients had injection site reactions compared with 2.2% in control patients. The presence of
antibodies to golimumab may increase the risk of injection site reactions. The majority of the
injection site reactions were mild and moderate and the most frequent manifestation was injection
site erythema. Injection site reactions generally did not necessitate discontinuation of the medicinal
product.
In controlled phase IIb and III trials in RA, PsA, AS and severe persistent asthma, no patients treated
with golimumab developed anaphylactic reactions.
Autoimmune antibodies
In Phase III trials in RA, PsA, and AS through 1 year of follow up, 4.0% of golimumab-treated
patients and 2.6% of control patients were newly ANA-positive (at titres of 1:160 or greater). The
frequency of anti-dsDNA antibodies at 1 year of follow up in patients anti-dsDNA negative at
baseline was uncommon.
Single doses up to 10 mg/kg intravenously have been administered in a clinical study without
dose-limiting toxicity. In case of an overdose, it is recommended that the patient be monitored for
any signs or symptoms of adverse effects and appropriate symptomatic treatment be instituted
immediately.
PHARMACOLOGICAL PROPERTIES
5.1 Pharmacodynamic properties
Pharmacotherapeutic group: Tumour necrosis factor alpha (TNF-α) inhibitors, ATC code: L04AB06
Mechanism of action
Golimumab is a human monoclonal antibody that forms high affinity, stable complexes with both the
soluble and transmembrane bioactive forms of human TNF-α, which prevents the binding of TNF-α
to its receptors.
Pharmacodynamic effects
The binding of human TNF by golimumab was shown to neutralise TNF-α -induced cell-surface
expression of the adhesion molecules E-selectin, vascular cell adhesion molecule (VCAM)-1 and
intercellular adhesion molecule (ICAM)-1 by human endothelial cells.
In vitro
, TNF-induced
secretion of interleukin (IL)-6, IL-8 and granulocyte-macrophage colony stimulating factor
(GM-CSF) by human endothelial cells was also inhibited by golimumab.
Improvement in C-reactive protein (CRP) levels were observed relative to placebo groups and
treatment with Simponi resulted in significant reductions from baseline in serum levels of IL-6,
ICAM-1, matrix-metalloproteinase (MMP)-3 and vascular endothelial growth factor (VEGF)
compared to control treatment. In addition, levels of TNF-α were reduced in RA and AS patients and
levels of IL-8 were reduced in PsA patients. These changes were observed at the first assessment
(week 4) after the initial Simponi administration and were generally maintained through week 24.
Rheumatoid arthritis
The efficacy of Simponi was demonstrated in three multi-centre, randomised, double-blind,
placebo-controlled studies in over 1500 patients ≥18 years of age with moderately to severely active
RA diagnosed according to American College of Rheumatology (ACR) criteria for at least 3 months
prior to screening. Patients had at least 4 swollen and 4 tender joints. Simponi or placebo were
subcutaneously administered every 4 weeks.
GO-FORWARD evaluated 444 patients who had active RA despite a stable dose of at least
15 mg/week of MTX and who had not been previously treated with an anti-TNF agent. Patients were
randomised to receive placebo + MTX, Simponi 50 mg + MTX, Simponi 100 mg + MTX or Simponi
100 mg + placebo. Patients receiving placebo + MTX were switched to Simponi 50 mg + MTX after
week 24. At week 52, patients entered an open label long-term extension.
GO-AFTER evaluated 445 patients who were previously treated with one or more of the anti-TNF
agents adalimumab, etanercept, or infliximab. Patients were randomised to receive placebo, Simponi
50 mg, or Simponi 100 mg. Patients were allowed to continue concomitant DMARD therapy with
MTX, sulfasalazine (SSZ), and/or hydroxychloroquine (HCQ) during the study. The stated reasons
for discontinuation of prior anti TNF therapies were lack of efficacy (58%), intolerance (13%),
and/or reasons other than safety or efficacy (29%, mostly for financial reasons).
GO-BEFORE evaluated 637 patients with active RA who were MTX-naïve and had not previously
been treated with an anti-TNF agent. Patients were randomised to receive placebo + MTX, Simponi
50 mg + MTX, Simponi 100 mg + MTX or Simponi 100 mg + placebo. At week 52, patients entered
an open label long-term extension in which patients receiving placebo + MTX who had at least 1
tender or swollen joint were switched to Simponi 50 mg + MTX.
In GO-FORWARD, the (co-)primary endpoints were the percentage of patients achieving an ACR 20
response at week 14 and the improvement from baseline in Health Assessment Questionnaire (HAQ)
at week 24. In GO-AFTER, the primary endpoint was the percentage of patients achieving an ACR
20 response at week 14. In GO-BEFORE, the co-primary endpoints were the percentage of patients
achieving ACR 50 response at week 24 and the change from baseline in the van der Heijde-modified
Sharp (vdH-S) score at week 52. In addition to the primary endpoint(s), additional assessments of the
impact of Simponi treatment on the signs and symptoms of arthritis, physical function and
health-related quality of life were performed.
In general, no clinically meaningful differences in measures of efficacy were observed between the
Simponi 50 mg and 100 mg dosing regimens with concomitant MTX.
Signs and symptoms
Key ACR results for the Simponi 50 mg dose at weeks 14,24 and 52 for GO-FORWARD, GO-
AFTER and GO-BEFORE are shown in Table 2 and are described below. Responses were observed
at the first assessment (week 4) after the initial Simponi administration.
In GO-FORWARD, among 89 subjects randomised to Simponi 50 mg + MTX, 48 were still on this
treatment at week 104. Among those, 40, 33 and 24 patients had ACR 20/50/70 response,
respectively at week 104.
In GO-AFTER, the percentage of patients achieving an ACR 20 response was greater for patients
receiving Simponi than for patients receiving placebo regardless of the reason reported for
discontinuation of one or more prior anti-TNF therapies.
Table 2
Key efficacy outcomes from the controlled portions of GO-FORWARD, GO-AFTER and
GO-BEFORE.
GO-FORWARD
Active RA despite MTX
GO-AFTER
Active RA, previously
treated with one or more
anti-TNF agent(s)
GO-BEFORE
Active RA, MTX Naïve
Responders, % of patients
ACR 20
Week 14
ACR 70
Week 14 4% 14% p=0.008 2% 10% p=0.005 NA NA
Week 24 5% 20%* 2% 9% p=0.009 16% 24%
Week 52
NA NA NA NA 22% 28%
a n reflects randomised patients; actual number of patients evaluable for each endpoint may
vary by timepoint.
* p ≤ 0.001
NA: Not Applicable
In GO-BEFORE the primary analysis in patients with moderate to severe rheumatoid arthritis
(combined Simponi 50 and 100 mg + MTX groups vs MTX alone for ACR50) was not statistically
significant at week 24 (p=0.053). At week 52 in the overall population, the percentage of patients in
the Simponi 50 mg + MTX group who achieved an ACR response was generally higher but not
significantly different when compared with MTX alone (see Table 2). Additional analyses were
performed in subsets representative of the indicated population of patients with severe, active and
progressive RA. A generally greater effect of Simponi 50 mg + MTX versus MTX alone was
demonstrated in the indicated population compared with the overall population.
In GO-FORWARD and GO-AFTER, clinically meaningful and statistically significant responses in
Disease Activity Scale (DAS)28 were observed at each prespecified time point, at week 14 and at
week 24 ( p ≤ 0.001). Among patients who remained on the Simponi treatment to which they were
randomised at study start, DAS28 responses were maintained through week 104.
In GO-BEFORE, major clinical response, defined as the maintenance of an ACR 70 response over a
continuous 6-month period, was measured. At week 52, 15% of patients in the Simponi 50 mg +
MTX group achieved a major clinical response compared with 7% of patients in the placebo + MTX
group (p = 0.018). Among 159 subjects randomised to Simponi 50 mg + MTX, 96 were still on this
treatment at week 104. Among those, 85, 66 and 53 patients had ACR 20/50/70 response,
respectively, at week 104.

























In GO-BEFORE the change from baseline in the vdH-S score, a composite score of structural
damage that radiographically measures the number and size of joint erosions and the degree of joint
space narrowing in hands/wrists and feet, was used to assess the degree of structural damage. Key
results for the Simponi 50 mg dose at week 52 are presented in Table 3.
The number of patients with no new erosions or a change from baseline in total vdH-S Score ≤ 0 was
significantly higher in the Simponi treatment group than in the control group (p = 0.003). The
radiographic effects observed at week 52 were maintained through week 104.
Table 3
Radiographic Mean (SD) Changes from Baseline in Total vdH-S Score at week 52 in the overall
population of GO-BEFORE
a
n reflects randomized patients
*
p = 0.015
**
p = 0.044
Physical function and health-related quality of life
Physical function and disability were assessed as a separate endpoint in GO-FORWARD and
GO-AFTER using the disability index of the HAQ. In these studies, Simponi demonstrated clinically
meaningful and statistically significant improvement in HAQ from baseline versus control at week
24. Among patients who remained on the Simponi treatment to which they were randomised at study
start, improvement in HAQ was maintained through week 104.
In GO-FORWARD clinically meaningful and statistically significant improvements were
demonstrated in health-related quality of life as measured by the physical component score of the
SF-36 in patients treated with Simponi versus placebo at week 24. Among patients who remained on
the Simponi treatment to which they were randomised at study start, improvement of the SF-36
physical component was maintained through week 104. In GO-FORWARD and GO-AFTER,
statistically significant improvements were observed in fatigue as measured by functional assessment
of chronic illness therapy-fatigue scale (FACIT-F).
Psoriatic arthritis
The safety and efficacy of Simponi were evaluated in a multi-centre, randomised, double-blind,
placebo-controlled study (GO-REVEAL) in 405 adult patients with active PsA (≥ 3 swollen joints
and ≥ 3 tender joints) despite non-steroidal anti-inflammatory (NSAID) or DMARD therapy. Patients
in this study had a diagnosis of PsA for at least 6 months and had at least mild psoriatic disease.
Patients with each sub-type of psoriatic arthritis were enrolled, including polyarticular arthritis with
no rheumatoid nodules (43%), asymmetric peripheral arthritis (30%), distal interphalangeal (DIP)
joint arthritis (15%), spondylitis with peripheral arthritis (11%), and arthritis mutilans (1%). Previous
treatment with an anti-TNF agent was not allowed. Simponi or placebo were administered
subcutaneously every 4 weeks. Patients were randomly assigned to placebo, Simponi 50 mg, or
Simponi 100 mg.
















Approximately forty-eight percent of patients continued on stable doses of methotrexate (≤25
mg/week). The primary endpoint was the percentage of patients achieving ACR 20 response at week
14. Placebo-controlled efficacy data were collected and analysed through week 24.
Key results for the 50 mg dose are shown in Table 4 and described below. In general, no clinically
meaningful differences in measures of efficacy were observed between the Simponi 50 mg and
100 mg dosing regimens.
Table 4
Key efficacy outcomes from GO-REVEAL
Responders, % of patients
ACR 20
Week 14 1 % 12 %
Week 24 1 % 19 %
PASI
b
75
c
Week 14
3 %
40 %
Week 24
1 %
56 %
* p < 0.05 for all comparisons; p-value calculations are based on comparisons
of median values for continuous variables
a
n reflects randomised patients; actual number of patients evaluable for each
endpoint may vary by timepoint
b Psoriasis Area and Severity Index
c
Based on the subset of patients with ≥ 3% BSA involvement at baseline, 79
patients (69.9%) in the placebo group and 109 (74.3%) in the Simponi 50 mg
group.
Statistically significant responses in DAS28 were also observed at weeks 14 and 24 (p < 0.05).
Improvements in key measures of disease activity were observed at the first assessment (week 4)
after the initial Simponi administration and were maintained through week 24. Similar ACR 20
responses at week 14 were observed in patients with polyarticular arthritis with no rheumatoid
nodules and asymmetric peripheral arthritis PsA subtypes. The number of patients with other PsA
subtypes was too small to allow meaningful assessment. Responses observed in the Simponi treated
groups were similar in patients receiving and not receiving concomitant MTX.
Improvements in parameters of peripheral activity characteristic of psoriatic arthritis (e.g. number of
swollen joints, number of painful/tender joints, dactylitis and enthesitis) were seen in the
Simponi-treated patients.
Simponi treatment resulted in significant improvement in physical function as assessed by HAQ, as
well as significant improvements in health-related quality of life as measured by the physical and
mental component summary scores of the SF-36.
Ankylosing spondylitis
The safety and efficacy of Simponi were evaluated in a multi-centre, randomised, double-blind,
placebo-controlled study (GO-RAISE) in 356 adult patients with active ankylosing spondylitis
(defined as a Bath Ankylosing Spondylitis Disease Activity Index (BASDAI) ≥4 and a VAS for total
back pain of ≥4, on a scale of 0 to 10 cm). Patients enrolled in this study had active disease despite




















current or previous NSAID or DMARD therapy and had not previously been treated with anti-TNF
therapy. Simponi or placebo were administered subcutaneously every 4 weeks. Patients were
randomly assigned to placebo, Simponi 50 mg and Simponi 100 mg and were allowed to continue
concomitant DMARD therapy (MTX, SSZ and/or HCQ). The primary endpoint was the percentage
of patients achieving Ankylosing Spondylitis Assessment Study Group (ASAS) 20 response at week
14. Placebo-controlled efficacy data were collected and analysed through week 24.
Key results for the 50 mg dose are shown in Table 5 and described below. In general, no clinically
meaningful differences in measures of efficacy were observed between the Simponi 50 mg and
100 mg dosing regimens.
Table 5
Key efficacy outcomes from GO-RAISE.
Responders, % of patients
ASAS 20
* p ≤ 0.001 for all comparisons
a
n reflects randomised patients; actual number of patients evaluable for each endpoint
may vary by timepoint
Statistically significant responses in BASDAI 50, 70 and 90 (p ≤ 0.017) were also seen at weeks 14
and 24. Improvements in key measures of disease activity were observed at the first assessment
(week 4) after the initial Simponi administration and were maintained through week 24. Consistent
efficacy was seen in patients regardless of use of DMARDs (MTX, sulfasalazine and/or
hydroxychloroquine), HLA-B27 antigen status or baseline CRP levels as assessed by ASAS 20
responses at week 14.
Simponi treatment resulted in significant improvements in physical function as assessed by changes
from baseline in BASFI at weeks 14 and 24. Health-related quality of life as measured by the
physical component score of the SF-36 was also improved significantly at weeks 14 and 24.
Immunogenicity
Across the Phase III RA, PsA and AS studies through week 52, antibodies to golimumab were
detected in 5% (105/2115) of golimumab treated patients and, where tested, nearly all antibodies
were neutralising
in vitro
. Similar rates were shown across rheumatologic indications. Treatment
with concomitant MTX resulted in a lower proportion of patients with antibodies to golimumab than
patients receiving golimumab without MTX (approximately 3% [41/1262] versus 8% [64/853],
respectively).
The presence of antibodies to golimumab may increase the risk of injection site reactions (see section
4.4).
The small number of patients positive for antibodies to golimumab limits the ability to draw
definitive conclusions regarding the relationship between antibodies to golimumab and clinical
efficacy or safety measures.
Because immunogenicity analyses are product- and assay-specific, comparison of antibody rates with
those from other products is not appropriate.






























Paediatric population
The European Medicines Agency has waived the obligation to conduct studies with Simponi in all
subsets of the paediatric population in ankylosing spondylitis and rheumatoid arthritis
(see section
4.2 for information on paediatric use).
The European Medicines Agency has deferred the obligation to submit the results of studies with
Simponi in one or more subsets of the paediatric population in juvenile idiopathic arthritis and
psoriatic arthritis (see section 4.2 for information on paediatric use).
Pharmacokinetic properties
Following a single subcutaneous administration of golimumab
to healthy subjects or patients with
RA, the median time to reach maximum serum concentrations (T
max
) ranged from 2 to 6 days. A
subcutaneous injection of 50 mg golimumab to healthy subjects produced a mean ± standard
deviation maximum serum concentration (C
max
) of 3.1 ± 1.4 μg/ml.
Golimumab exhibited approximately dose-proportional pharmacokinetics in patients with RA over
the dose range of 0.1 to 10.0 mg/kg following a single intravenous dose. The systemic clearance of
golimumab was estimated to be 6.9 ± 2.0 ml/day/kg, and mean volume of distribution was
115 ± 19 ml/kg. Terminal half-life value was estimated to be approximately 12 ± 3 days in healthy
subjects and similar values were observed in patients with RA, PsA or AS.
Following a single subcutaneous injection of 100 mg, the absorption of golimumab was similar in the
upper arm, abdomen, and thigh, with a mean absolute bioavailability of 51%. Since golimumab
exhibited approximately dose proportional PK following a subcutaneous administration, the absolute
bioavailability of the golimumab 50 mg dose is expected to be similar.
When 50 mg golimumab was administered subcutaneously to patients with RA, PsA or AS every 4
weeks, serum concentrations reached steady state by week 12. With concomitant use of MTX,
treatment with 50 mg golimumab subcutaneous every 4 weeks resulted in a mean (± standard
deviation) steady-state trough serum concentration of approximately 0.6 ± 0.4 μg/ml in RA patients
with active RA despite MTX therapy, and approximately 0.5 ± 0.4 μg/ml
in patients with active PsA
and approximately 0.8 ± 0.4 μg/ml in patients with AS.
Patients with RA, PsA or AS who did not receive concomitant MTX had approximately 30% lower
steady-state trough concentrations of golimumab than those who received golimumab with MTX. In
a limited number of RA patients treated with subcutaneous golimumab over a 6-month period,
concomitant use of MTX reduced the apparent clearance of golimumab by approximately 36%.
However, population pharmacokinetic analysis indicated that concomitant use of NSAIDs, oral
corticosteroids or sulfasalazine did not influence the apparent clearance of golimumab.
There was a trend toward higher apparent clearance of golimumab with increasing weight (see
section 4.2).
Patients who developed anti-golimumab antibodies generally had low trough steady-state serum
concentrations of golimumab (see section 5.1).
5.3 Preclinical safety data
Non-clinical data reveal no special hazard for humans based on conventional studies of safety
pharmacology, repeated dose toxicity, toxicity to reproduction and development.
No mutagenicity studies, animal fertility studies nor long-term carcinogenic studies have been
conducted with golimumab.
In a fertility and general reproductive function study in mouse, using an analogous antibody that
selectively inhibits the functional activity of mouse TNFα, the number of pregnant mice was reduced.
It is not known whether this finding was due to effects on the males and/or the females. In a
developmental toxicity study conducted in mice following administration of the same analogous
antibody, and in cynomolgus monkeys using golimumab, there was no indication of maternal
toxicity, embryotoxicity or teratogenicity.
PHARMACEUTICAL PARTICULARS
Sorbitol(E420)
L-histidine
L-histidine monohydrochloride monohydrate
Polysorbate 80
Water for injections.
In the absence of compatibility studies, this medicinal product must not be mixed with other
medicinal products.
6.4 Special precautions for storage
Store in a refrigerator (2°C – 8°C).
Do not freeze.
Keep the pre-filled pen in the outer carton in order to protect it from light.
6.5
Nature and contents of container
0.5 ml solution in a pre-filled syringe (1.0 ml Type 1 glass) with a fixed needle (stainless steel) and a
needle cover (rubber containing latex) in a pre-filled pen. Simponi is available in packs containing 1
pre-filled pen and multipacks containing 3 (3 packs of 1) pre-filled pens.
Not all pack sizes may be marketed.
Simponi should not be used if the solution is discoloured, cloudy or containing visible foreign
particles.
6.6 Special precautions for disposal and other handling
Simponi is supplied in a single use pre-filled pen called SmartJect. Each Simponi pack is provided
with instructions for use that fully describes the use of the pen. After removing the pre-filled pen
from the refrigerator this should be allowed to reach room temperature by waiting for 30 minutes,
before injecting Simponi. The pen should not be shaken.
The solution is clear to slightly opalescent, colourless to light yellow and may contain a few small
translucent or white particles of protein. This appearance is not unusual for solutions containing
protein.
Comprehensive instructions for the preparation and administration of Simponi in a pre-filled pen are
given in the package leaflet.
Any unused product or waste material should be disposed of in accordance with local requirements.
MARKETING AUTHORISATION HOLDER
Janssen Biologics B.V.
Einsteinweg 101
2333 CB Leiden
The Netherlands
MARKETING AUTHORISATION NUMBER(S)
EU/1/09/546/001 1 pre-filled pen
EU/1/09/546/002 3 pre-filled pens
DATE OF FIRST AUTHORISATION
10. DATE OF REVISION OF THE TEXT
Detailed information on this medicinal product is available on the website of the European Medicines
Agency
http://www.ema.europa.eu
.
NAME OF THE MEDICINAL PRODUCT
Simponi 50 mg solution for injection in pre-filled syringe.
QUALITATIVE AND QUANTITATIVE COMPOSITION
One 0.5 ml pre-filled syringe contains 50 mg of golimumab*.
* Human IgG1κ monoclonal antibody produced by a murine hybridoma cell line with recombinant
DNA technology.
Excipient:
Each pre-filled syringe contains 20.5 mg sorbitol per 50 mg dose.
For a full list of excipients, see section 6.1.
Solution for injection in pre-filled syringe (injection)
The solution is clear to slightly opalescent, colourless to light yellow.
4.1 Therapeutic indications
Rheumatoid arthritis (RA)
Simponi, in combination with methotrexate (MTX), is indicated for:
•
the treatment of moderate to severe, active rheumatoid arthritis in adults when the response to
disease-modifying anti-rheumatic drug (DMARD) therapy including MTX has been
inadequate.
the treatment of severe, active and progressive rheumatoid arthritis in adults not previously
treated with MTX.
Simponi, in combination with MTX, has been shown to reduce the rate of progression of joint
damage as measured by X-ray and to improve physical function.
Psoriatic arthritis (PsA)
Simponi, alone or in combination with MTX, is indicated for the treatment of active and progressive
psoriatic arthritis in adults when the response to previous disease-modifying anti-rheumatic drug
(DMARD) therapy has been inadequate. Simponi has also been shown to improve physical function
in this patient population.
Ankylosing spondylitis (AS)
Simponi is indicated for the treatment of severe, active ankylosing spondylitis in adults who have
responded inadequately to conventional therapy.
4.2
Posology and method of administration
Simponi treatment is to be initiated and supervised by qualified physicians experienced in the
diagnosis and treatment of rheumatoid arthritis, psoriatic arthritis or ankylosing spondylitis. Patients
treated with Simponi should be given the Patient Alert Card.
Rheumatoid arthritis
Simponi 50 mg given once a month, on the same date each month.
Simponi should be given concomitantly with MTX.
Psoriatic arthritis
Simponi 50 mg given once a month, on the same date each month.
Ankylosing spondylitis
Simponi 50 mg given once a month, on the same date each month.
Available data suggest that clinical response is usually achieved within 12 to 14 weeks of treatment
(after 3-4 doses). Continued therapy should be reconsidered in patients who show no evidence of
therapeutic benefit within this time period.
In patients weighing more than 100 kg who do not achieve an adequate clinical response after 3 or 4
doses, increasing the dose of golimumab to 100 mg once a month may be considered, taking into
account the increased risk of certain serious adverse drug reactions with the 100 mg dose compared
with the 50 mg dose (see section 4.8). Continued therapy should be reconsidered in patients who
show no evidence of therapeutic benefit after receiving 3 to 4 additional doses of 100 mg.
Missed dose
If a patient forgets to inject Simponi on the planned date, the forgotten dose should be injected as
soon as the patient remembers. Patients should be instructed not to inject a double dose to make up
for the forgotten dose.
The next dose should be administered based on the following guidance:
•
if the dose is less than 2 weeks late, the patient should inject his/her forgotten dose and stay
on his/her original monthly schedule.
•
if the dose is more than 2 weeks late, the patient should inject his/her forgotten dose and a
new once-monthly schedule should be established from the date of this injection.
Elderly patients (≥ 65 years)
No dose adjustment is required in the elderly.
Renal and hepatic impairment
Simponi has not been studied in these patient populations. No dose recommendations can be made.
Paediatric population
The safety and efficacy of Simponi in patients aged less than 18 have not yet been established. No
data are available.
Method of administration
For subcutaneous use. After proper training in subcutaneous injection technique, patients may
self-inject with Simponi if their physician determines that this is appropriate, with medical follow-up
as necessary. Patients should be instructed to inject the full amount of Simponi according to the
comprehensive instructions for administration provided in the package leaflet. For administration
instructions, see section 6.6.
Hypersensitivity to the active substance or to any of the excipients (see section 6.1).
Active tuberculosis (TB) or other severe infections such as sepsis, and opportunistic infections
(see section 4.4).
Moderate or severe heart failure (NYHA class III/IV) (see section 4.4).
4.4 Special warnings and precautions for use
Infections
Patients must be monitored closely for infections including tuberculosis before, during and after
treatment with Simponi. Because the elimination of golimumab may take up to 5 months, monitoring
should be continued throughout this period. Further treatment with Simponi must not be given if a
patient develops a serious infection or sepsis (see section 4.3).
Simponi should not be given to patients with a clinically important, active infection. Caution should
be exercised when considering the use of Simponi in patients with a chronic infection or a history of
recurrent infection. Patients should be advised of, and avoid exposure to, potential risk factors for
infection as appropriate.
Patients taking TNF-blockers are more susceptible to serious infections.
Bacterial (including sepsis and pneumonia), mycobacterial (including TB), invasive fungal and
opportunistic infections, including fatalities, have been reported in patients receiving Simponi. Some
of these serious infections have occurred in patients on concomitant immunosuppressive therapy that,
in addition to their underlying disease, could predispose them to infections. Patients who develop a
new infection while undergoing treatment with Simponi should be monitored closely and undergo a
complete diagnostic evaluation. Administration of Simponi should be discontinued if a patient
develops a new serious infection or sepsis, and appropriate antimicrobial or antifungal therapy should
be initiated until the infection is controlled. For patients who have resided in or travelled to regions
where invasive fungal infections such as histoplasmosis, coccidioidomycosis, or blastomycosis are
endemic, the benefits and risks of Simponi treatment should be carefully considered before initiation
of Simponi therapy.
Tuberculosis
There have been reports of tuberculosis in patients receiving Simponi. It should be noted that in the
majority of these reports, tuberculosis was extrapulmonary presenting as either local or disseminated
disease.
Before starting treatment with Simponi, all patients must be evaluated for both active and inactive
(‘latent’) tuberculosis. This evaluation should include a detailed medical history with personal history
of tuberculosis or possible previous contact with tuberculosis and previous and/or current
immunosuppressive therapy. Appropriate screening tests, i.e. tuberculin skin or blood test and chest
X-ray, should be performed in all patients (local recommendations may apply). It is recommended
that the conduct of these tests should be recorded in the patient’s alert card. Prescribers are reminded
of the risk of false negative tuberculin skin test results, especially in patients who are severely ill or
immunocompromised.
If active tuberculosis is diagnosed, Simponi therapy must not be initiated (see section 4.3).
If latent tuberculosis is suspected, a physician with expertise in the treatment of tuberculosis should
be consulted. In all situations described below, the benefit/risk balance of Simponi therapy should be
very carefully considered.
If inactive (‘latent’) tuberculosis is diagnosed, treatment for latent tuberculosis must be started with
anti-tuberculosis therapy before the initiation of Simponi, and in accordance with local
recommendations.
In patients who have several or significant risk factors for tuberculosis and have a negative test for
latent tuberculosis, anti-tuberculosis therapy should be considered before the initiation of Simponi.
Use of anti-tuberculosis therapy should also be considered before the initiation of Simponi in patients
with a past history of latent or active tuberculosis in whom an adequate course of treatment cannot be
confirmed.
All patients should be informed to seek medical advice if signs/symptoms suggestive of tuberculosis
(e.g. persistent cough, wasting/weight loss, low-grade fever) appear during or after Simponi
treatment.
Hepatitis B virus reactivation
Reactivation of hepatitis B has occurred in patients receiving a TNF-antagonist including Simponi,
who are chronic carriers of this virus (i.e., surface antigen positive). Some cases have had fatal
outcome.
Patients should be tested for HBV infection before initiating treatment with Simponi. For patients
who test positive for HBV infection, consultation with a physician with expertise in the treatment of
hepatitis B is recommended.
Carriers of HBV who require treatment with Simponi should be closely monitored for signs and
symptoms of active HBV infection throughout therapy and for several months following termination
of therapy. Adequate data of treating patients who are carriers of HBV with anti-viral therapy in
conjunction with TNF-antagonist therapy to prevent HBV reactivation are not available. In patients
who develop HBV reactivation, Simponi should be stopped and effective anti-viral therapy with
appropriate supportive treatment should be initiated.
Malignancies and lymphoproliferative disorders
The potential role of TNF-blocking therapy in the development of malignancies is not known. Based
on the current knowledge, a possible risk for the development of lymphomas, leukaemia or other
malignancies in patients treated with a TNF-antagonist cannot be excluded. Caution should be
exercised when considering TNF-blocking therapy for patients with a history of malignancy or when
considering continuing treatment in patients who develop malignancy.
Paediatric malignancy
Malignancies, some fatal, have been reported among children, adolescents and young adults (up to 22
years of age) treated with TNF-blocking agents (initiation of therapy ≤ 18 years of age) in the post
marketing setting. Approximately half the cases were lymphomas. The other cases represented a
variety of different malignancies and included rare malignancies usually associated with
immunosuppression. A risk for the development of malignancies in children and adolescents treated
with TNF-blockers cannot be excluded.
Lymphoma and leukaemia
In the controlled portions of clinical trials of all the TNF-blocking agents including Simponi, more
cases of lymphoma have been observed among patients receiving anti-TNF treatment compared with
control patients. During the Simponi Phase IIb and Phase III clinical trials, the incidence of
lymphoma in Simponi-treated patients was higher than expected in the general population. In the
post-marketing setting, cases of leukaemia have been reported in patients treated with a TNF-
antagonist. There is an increased background risk for lymphoma and leukaemia in rheumatoid
arthritis patients with long-standing, highly active, inflammatory disease, which complicates risk
estimation.
Malignancies other than lymphoma
In the controlled portions of the Simponi Phase IIb and Phase III clinical trials in RA, PsA, and AS,
the incidence of non-lymphoma malignancies (excluding non-melanoma skin cancer) was similar
between the Simponi and the control groups.
In an exploratory clinical trial evaluating the use of Simponi in patients with severe persistent
asthma, more malignancies were reported in patients treated with Simponi compared with control
patients (see section 4.8). The significance of this finding is unknown.
In an exploratory clinical trial evaluating the use of another anti-TNF agent, infliximab, in patients
with moderate to severe chronic obstructive pulmonary disease (COPD), more malignancies, mostly
in the lung or head and neck, were reported in infliximab-treated patients compared with control
patients. All patients had a history of heavy smoking. Therefore, caution should be exercised when
using any TNF-antagonist in COPD patients, as well as in patients with an increased risk of
malignancy due to heavy smoking.
Congestive heart failure (CHF)
Cases of worsening congestive heart failure (CHF) and new onset CHF have been reported with TNF
blockers, including Simponi. In a clinical trial with another TNF-antagonist worsening congestive
heart failure and increased mortality due to CHF have been observed. Simponi has not been studied
in patients with CHF. Simponi should be used with caution in patients with mild heart failure
(NYHA class I/II). Patients should be closely monitored and Simponi must be discontinued in
patients who develop new or worsening symptoms of heart failure (see section 4.3).
Neurological events
Use of TNF-blocking agents, including Simponi, has been associated with cases of new onset or
exacerbation of clinical symptoms and/or radiographic evidence of central nervous system
demyelinating disorders, including multiple sclerosis and peripheral demyelinating disorders. In
patients with pre-existing or recent onset of demyelinating disorders, the benefits and risks of
anti-TNF treatment should be carefully considered before initiation of Simponi therapy.
Discontinuation of Simponi should be considered if these disorders develop.
Surgery
There is limited safety experience of Simponi treatment in patients who have undergone surgical
procedures, including arthroplasty. The long half-life should be taken into consideration if a surgical
procedure is planned. A patient who requires surgery while on Simponi should be closely monitored
for infections, and appropriate actions should be taken.
Immunosuppression
The possibility exists for TNF-blocking agents, including Simponi, to affect host defences against
infections and malignancies since TNF mediates inflammation and modulates cellular immune
responses.
Autoimmune processes
The relative deficiency of TNF
α
caused by anti-TNF therapy may result in the initiation of an
autoimmune process. If a patient develops symptoms suggestive of a lupus-like syndrome following
treatment with Simponi and is positive for antibodies against double-stranded DNA, treatment with
Simponi should be discontinued (see section 4.8).
Hematologic reactions
There have been post-marketing reports of pancytopaenia, leucopaenia, neutropaenia, aplastic
anaemia, and thrombocytopaenia in patients receiving TNF-blockers. Cytopaenias including
pancytopaenia have been infrequently reported with Simponi in clinical trials. All patients should be
advised to seek immediate medical attention if they develop signs and symptoms suggestive of blood
dyscrasias (e.g. persistent fever, bruising, bleeding, pallor). Discontinuation of Simponi therapy
should be considered in patients with confirmed significant haematologic abnormalities.
Concurrent administration of TNF-antagonists and anakinra
Serious infections and neutropenia were seen in clinical studies with concurrent use of anakinra and
another TNF-blocking agent, etanercept, with no added clinical benefit. Because of the nature of the
adverse events seen with this combination therapy, similar toxicities may also result from the
combination of anakinra and other TNF-blocking agents. The combination of Simponi and anakinra
is not recommended.
Concurrent administration of TNF-antagonists and abatacept
In clinical studies concurrent administration of TNF-antagonists and abatacept has been associated
with an increased risk of infections including serious infections compared to TNF-antagonists alone,
without increased clinical benefit. The combination of Simponi and abatacept is not recommended.
Switching between biological DMARDs
When switching from one biologic to another, patients should be monitored for signs of infection.
Vaccinations
Patients treated with Simponi may receive concurrent vaccinations, except for live vaccines (see
section 4.5). No data are available on the response to vaccination, risk of infection or transmission of
infection with the administration of live vaccines to patients receiving Simponi.
Allergic reactions
In post-marketing experience, serious systemic hypersensitivity reactions (including anaphylactic
reaction) have been reported following Simponi administration. Some of these reactions occurred
after the first administration of Simponi. If an anaphylactic reaction or other serious allergic reactions
occur, administration of Simponi should be discontinued immediately and appropriate therapy
initiated.
Latex sensitivity
The needle cover on the pre-filled syringe is manufactured from dry natural rubber containing latex,
and may cause allergic reactions in individuals sensitive to latex.
Elderly patients (≥ 65 years
)
In the Phase III studies in RA, PsA, and AS, no overall differences in adverse events (AEs), serious
adverse events (SAEs) , and serious infections in patients age 65 or older (n=155) who received
Simponi were observed compared with younger patients. However, caution should be exercised when
treating the elderly and particular attention paid with respect to occurrence of infections.
Renal and hepatic impairment
Specific studies of Simponi have not been conducted in patients with renal or hepatic impairment.
Simponi should be used with caution in subjects with impaired hepatic function (see section 4.2).
Excipients
Simponi contains sorbitol (E420). Patients with rare hereditary problems of fructose intolerance
should not take Simponi.
4.5 Interaction with other medicinal products and other forms of interaction
No interaction studies have been performed.
Concurrent use with anakinra and abatacept
The combination of Simponi and anakinra or abatacept is not recommended (see section 4.4).
Live vaccines
Live vaccines should not be given concurrently with Simponi (see section 4.4).
Methotrexate
Although concomitant use of MTX results in higher steady-state trough concentrations of Simponi in
patients with RA, PsA or AS, the data do not suggest the need for dose adjustment of either Simponi
or MTX (see section 5.2).
4.6 Fertility, pregnancy and lactation
Women of childbearing potential
Women of childbearing potential must use adequate contraception to prevent pregnancy and continue
its use for at least 6 months after the last golimumab treatment.
Pregnancy
There are no adequate data on the use of golimumab in pregnant women. Due to its inhibition of
TNF, golimumab administered during pregnancy could affect normal immune responses in the
newborn. Studies in animals do not indicate direct or indirect harmful effects with respect to
pregnancy, embryonal/foetal development, parturition or postnatal development (see section 5.3).
The use of golimumab in pregnant women is not recommended; golimumab should be given to a
pregnant woman only if clearly needed.
Breastfeeding
It is not known whether golimumab is excreted in human milk or absorbed systemically after
ingestion. Golimumab was shown to pass over to breast milk in monkeys, and because human
immunoglobulins are excreted in milk, women must not breast feed during and for at least 6 months
after golimumab treatment.
Fertility
No animal fertility studies have been conducted with golimumab. A fertility study in mice, using an
analogous antibody that selectively inhibits the functional activity of mouse TNFα, showed no
relevant effects on fertility (see section 5.3).
4.7 Effects on ability to drive and use machines
Simponi may have a minor influence on the ability to drive and use machines. Dizziness may occur
following administration of Simponi (see section 4.8).
Upper respiratory tract infection was the most common adverse drug reaction (ADR) reported in the
controlled Phase III RA, PsA, and AS studies through week 16, occurring in 7.2% of golimumab-
treated patients as compared with 5.8% of control patients. The most serious ADRs that have been
reported for Simponi include serious infections (including sepsis, pneumonia, TB, invasive fungal
and opportunistic infections), demyelinating disorders, lymphoma, HBV reactivation, CHF,
autoimmune processes (lupus-like syndrome) and haematologic reactions (see section 4.4).
ADRs observed in clinical studies and reported from world-wide post-marketing use of golimumab
are listed in Table 1. Within the designated system organ classes, the adverse drug reactions are listed
under headings of frequency and using the following convention: Very common (≥ 1/10); Common
(≥ 1/100 to < 1/10); Uncommon (≥ 1/1,000 to < 1/100); Rare (≥ 1/10,000 to < 1/1,000); Very rare
(< 1/10,000); Not known (cannot be estimated from the available data).
Table 1
Tabulated list of ADRs
Infections and infestations
Very common: Upper respiratory tract infection (nasopharyngitis, pharyngitis,
laryngitis and rhinitis)
Common: Bacterial infections (such as cellulitis), viral infections (such as
influenza and herpes), bronchitis, sinusitis, superficial fungal
infections
Uncommon: Septic shock, sepsis, tuberculosis, lower respiratory tract
infection (such as pneumonia), opportunistic infections (such as
invasive fungal infections [histoplasmosis, coccidioidomycosis,
pneumocytosis], bacterial, atypical mycobacterial infection and
protozoal), pyelonephritis, abscess, bacterial arthritis, infective
bursitis
Rare: Hepatitis B reactivation
Neoplasms, benign, malignant and
unspecified
Uncommon: Neoplasms (such as skin cancer, squamous cell carcinoma and
melanocytic naevus)
Rare: Lymphoma
Not known: Leukaemia*
Blood and lymphatic system
disorders
Common:
Anaemia
Uncommon: Leucopaenia, thrombocytopaenia
Rare: Pancytopaenia
Not known: Aplastic anaemia *
Immune system disorders
Common: Allergic reactions (bronchospasm, hypersensitivity, urticaria),
autoantibody positive
Rare: Serious systemic hypersensitivity reactions (including
anaphylactic reaction), vasculitis (systemic)
Endocrine disorders
Uncommon: Thyroid disorder (such as hypothyroidism, hyperthyroidism and
goitre)
Metabolism and nutrition disorders
Uncommon: Blood glucose increased, lipids increased
Common: Depression, insomnia
Common: Dizziness, paraesthesia, headache
Uncommon: Demyelinating disorders (central and peripheral), balance
disorders, dysguesia
Uncommon: Visual disorders (such as blurred vision and decreased visual
acuity), conjunctivitis, eye allergy (such as pruritis and
irritation)
Uncommon: Congestive heart failure (new onset or worsening), arrhythmia,
ischemic coronary artery disorders
Common: Hypertension
Uncommon: Thrombosis (such as deep venous and aortic), Raynaud’s
phenomenon, flushing
Respiratory, thoracic and
mediastinal disorders
Uncommon: Asthma and related symptoms (such as wheezing and bronchial
hyperactivity)













Rare: Interstitial lung disease
Gastrointestinal disorders
Common: Constipation, dyspepsia, gastrointestinal and abdominal pain
Uncommon: Gastrointestinal inflammatory disorders (such as gastritis and
colitis), gastro-oesophageal reflux disease, stomatitis
Hepatobiliary disorders
Common: Alanine aminotransferase increased, aspartate aminotransferase
increased
Uncommon: Cholelithiasis, hepatic disorders
Skin and subcutaneous tissue
disorders
Common: Alopecia, dermatitis, pruritus, rash
Uncommon: Psoriasis (new onset
or worsening of pre-existing psoriasis,
palmar/plantar and pustular), urticaria, vasculitis (cutaneous)
Musculoskeletal and connective
tissue disorders
Rare: Lupus-like syndrome
Renal and urinary disorders
Uncommon: Bladder disorders
Rare: Renal disorders
Reproductive system and breast
disorders
Uncommon: Breast disorders, menstrual disorders
General disorders and administration
site conditions
Common: Pyrexia, asthenia, injection site reaction (such as injection site
erythema, urticaria, induration, pain, bruising, pruritus, irritation
and paraesthesia), impaired healing, chest discomfort
Injury, poisoning and procedural
complications
Uncommon: Bone fractures
*: Observed with other TNF-blocking agents, but not observed in clinical studies with
golimumab.
Description of selected adverse drug reactions
Infections
Upper respiratory tract infection was the most common adverse reaction reported in the combined
Phase III RA, PsA, and AS studies through week 16, occurring in 7.2% of golimumab-treated
patients (incidence per patient-year: 0.26; 95% CI: 0.22, 0.31) as compared with 5.8% of control
patients (incidence per patient-year: 0.23; 95% CI: 0.17, 0.31). The incidence per patient year (95%
confidence interval; CI) of upper respiratory tract infections through 1 year of follow up was 0.23
events (0.21, 0.25) for golimumab-treated patients and 0.25 events (0.20, 0.31) for control patients.
In controlled Phase III trials through week 16 in RA, PsA, and AS, infections were observed in
28.3% of golimumab-treated patients (incidence per patient-year: 1.28; 95% CI: 1.18, 1.38)
compared with 24.7% of control patients (incidence per patient-year: 1.17; 95% CI: 1.02, 1.33). The
incidence per patient year (95% CI) of infections through 1 year of follow up was 1.32 events (1.27,
1.38) for golimumab-treated patients and 1.31 events (1.18, 1.44) for control patients.
In controlled Phase III trials through week 16 in RA, PsA, and AS, serious infections were observed
in 1.4% of golimumab-treated patients (incidence per patient-year: 0.06; 95% CI: 0.04, 0.08) and
1.3% of control patients (incidence per patient-year: 0.04; 95% CI: 0.02, 0.08). Serious infections
observed in golimumab-treated patients included tuberculosis, bacterial infections including sepsis
and pneumonia, invasive fungal infections and other opportunistic infections. Some of these
infections have been fatal. The incidence per patient year (95% CI) of serious infections through 1
year of follow up was 0.05 events (0.04, 0.06) for golimumab-treated patients and 0.06 events (0.04,











0.09) for control patients (see section 4.4). In the controlled and uncontrolled portions of the Phase II
RA and the Phase III RA, PsA, and AS trials with a mean follow-up of 1.6 years, a greater incidence
of TB was observed in the golimumab 100 mg treatment group compared with the golimumab 50 mg
group.
Lymphoma
The incidence of lymphoma in Simponi treated patients with RA, PsA and AS during the controlled
portions of phase IIb and III clinical trials and through 1 year of follow up was higher than expected
in the general population. Lymphoma was diagnosed in 2 subjects (both in golimumab 100 mg
treatment groups) with an incidence (95%, CI) per 100 subject-years of follow up of 0.10 (0.01, 0.37)
events for golimumab and 0.00 (0.00, 0.90) events for placebo. In the controlled and uncontrolled
portions of these trials through a median follow-up of 2.5 years, a greater incidence of lymphoma
was observed in the golimumab 100 mg treatment group compared with the golimumab 50 mg group.
The majority of lymphomas occurred in study GO-AFTER, which enrolled patients previously
exposed to anti-TNF agents who had longer disease duration and more refractory disease. See section
4.4.
Malignancies other than lymphoma
In the controlled portions of the Simponi Phase IIb and Phase III clinical trials in RA, PsA, and AS,
and through 1 year of follow up, the incidence of non-lymphoma malignancies (excluding
non-melanoma skin cancer) was similar between the Simponi and the control groups.
Through 1 year of follow up,of the Phase IIb and Phase III studies in rheumatologic indications,
nonmelanoma skin cancer was diagnosed in 19 subjects (5 in placebo, 6 in golimumab 50 mg and 8
in golimumab 100 mg treatment groups) with an incidence (95% CI) per 100 subject-years of follow
up of 0.72 (0.39, 1.20) events for golimumab and 1.51 (0.49, 3.52) events for placebo.
Through 1 year of follow up, of the Phase IIb and Phase III studies in rheumatologic indications,
malignancies besides nonmelanoma skin cancer and lymphoma were diagnosed in 12 subjects (2 in
placebo, 6 in golimumab 50 mg and 4 in golimumab 100 mg treatment groups) with an incidence
(95%CI) per 100 subject-years of follow up of 0.51 (0.24, 0.94) events for golimumab and 0.60
(0.07, 2.17) events for placebo. See section 4.4.
Cases reported in clinical studies in asthma
In an exploratory clinical study, patients with severe persistent asthma received a golimumab loading
dose (150% of the assigned treatment dose) subcutaneously at week 0 followed by golimumab 200
mg, golimumab 100 mg or golimumab 50 mg every 4 weeks subcutaneously through week 52. Eight
malignancies in the combined golimumab treatment group (n=230) and none in the placebo treatment
group (n=79). Lymphoma was reported in 1 patient, non-melanoma skin cancer in 2 patients, and
other malignancies in 5 patients. There was no specific clustering of any type of malignancy.
During the placebo-controlled portion of the study, the incidence (95% CI) of all malignancies per
100 subject-years of follow-up was 3.19 (1.38, 6.28) in the golimumab group. In this study, the
incidence (95% CI) per 100 subject-years of follow-up in golimumab-treated subjects was 0.40 (0.01,
2.20) for lymphoma, 0.79 (0.10, 2.86) for non-melanoma skin cancers, and 1.99 (0.64, 4.63) for other
malignancies. For placebo subjects, the incidence (95% CI) per 100 subject-years of follow-up of
these malignancies was 0.00 (0.00, 2.94). The significance of this finding is unknown.
Liver enzyme elevations
In controlled Phase III trials through week 16, mild ALT elevations (> 1 and < 3 x upper limit of
normal (ULN)) occurred in similar proportions of golimumab and control patients in the RA and PsA
studies (22.1% to 27.4% of patients); in the AS study, more golimumab-treated patients (25.6%) than
control patients (3.9 %) had mild ALT elevations. Through 1 year of follow-up the incidence of mild
ALT elevations was similar in golimumab-treated and control patients in RA and PsA studies. In the
AS population, the incidence of mild ALT elevations was higher in golimumab-treated patients than
in control patients.
In the RA and AS studies through week 16, ALT elevations ≥ 5 x ULN were uncommon and seen in
more golimumab-treated patients (0.4% to 0.9%) than control patients (0.0%). This trend was not
observed in the PsA population. Through 1 year of follow-up, the incidence of ALT elevations ≥5 x
ULN was similar in both golimumab-treated and control patients in the Phase III RA, PsA and AS
studies. In general these elevations were asymptomatic and the abnormalities decreased or resolved
with either continuation or discontinuation of golimumab or modification of concomitant
medications.
Within the Phase II and Phase III programme in RA, PsA and AS, one patient with pre-existing liver
abnormalities and confounding medication treated with golimumab developed non-infectious fatal
hepatitis with jaundice. The role of golimumab as a contributing or aggravation factor cannot be
excluded.
Injection site reactions
In controlled Phase III trials through week 16 in RA, PsA and AS, 5.8% of golimumab-treated
patients had injection site reactions compared with 2.2% in control patients. The presence of
antibodies to golimumab may increase the risk of injection site reactions. The majority of the
injection site reactions were mild and moderate and the most frequent manifestation was injection
site erythema. Injection site reactions generally did not necessitate discontinuation of the medicinal
product.
In controlled phase IIb and III trials in RA, PsA, AS and severe persistent asthma, no patients treated
with golimumab developed anaphylactic reactions.
Autoimmune antibodies
In Phase III trials in RA, PsA, and AS through 1 year of follow up, 4.0% of golimumab-treated
patients and 2.6% of control patients were newly ANA-positive (at titres of 1:160 or greater). The
frequency of anti-dsDNA antibodies at 1 year of follow up in patients anti-dsDNA negative at
baseline was uncommon.
Single doses up to 10 mg/kg intravenously have been administered in a clinical study without
dose-limiting toxicity. In case of an overdose, it is recommended that the patient be monitored for
any signs or symptoms of adverse effects and appropriate symptomatic treatment be instituted
immediately.
PHARMACOLOGICAL PROPERTIES
5.1 Pharmacodynamic properties
Pharmacotherapeutic group: Tumour necrosis factor alpha (TNF-α) inhibitors, ATC code:
L04AB06
Mechanism of action
Golimumab is a human monoclonal antibody that forms high affinity, stable complexes with both the
soluble and transmembrane bioactive forms of human TNF-α, which prevents the binding of TNF-α
to its receptors.
Pharmacodynamic effects
The binding of human TNF by golimumab was shown to neutralise TNF-α -induced cell-surface
expression of the adhesion molecules E-selectin, vascular cell adhesion molecule (VCAM)-1 and
intercellular adhesion molecule (ICAM)-1 by human endothelial cells.
In vitro
, TNF-induced
secretion of interleukin (IL)-6, IL-8 and granulocyte-macrophage colony stimulating factor
(GM-CSF) by human endothelial cells was also inhibited by golimumab.
Improvement in C-reactive protein (CRP) levels were observed relative to placebo groups and
treatment with Simponi resulted in significant reductions from baseline in serum levels of IL-6,
ICAM-1, matrix-metalloproteinase (MMP)-3 and vascular endothelial growth factor (VEGF)
compared to control treatment. In addition, levels of TNF-α were reduced in RA and AS patients and
levels of IL-8 were reduced in PsA patients. These changes were observed at the first assessment
(week 4) after the initial Simponi administration and were generally maintained through week 24.
Rheumatoid arthritis
The efficacy of Simponi was demonstrated in three multi-centre, randomised, double-blind,
placebo-controlled studies in over 1500 patients ≥18 years of age with moderately to severely active
RA diagnosed according to American College of Rheumatology (ACR) criteria for at least 3 months
prior to screening. Patients had at least 4 swollen and 4 tender joints. Simponi or placebo were
subcutaneously administered every 4 weeks.
GO-FORWARD evaluated 444 patients who had active RA despite a stable dose of at least
15 mg/week of MTX and who had not been previously treated with an anti-TNF agent. Patients were
randomised to receive placebo + MTX, Simponi 50 mg + MTX, Simponi 100 mg + MTX or Simponi
100 mg + placebo. Patients receiving placebo + MTX were switched to Simponi 50 mg + MTX after
week 24. At week 52, patients entered an open label long-term extension.
GO-AFTER evaluated 445 patients who were previously treated with one or more of the anti-TNF
agents adalimumab, etanercept, or infliximab. Patients were randomised to receive placebo, Simponi
50 mg, or Simponi 100 mg. Patients were allowed to continue concomitant DMARD therapy with
MTX, sulfasalazine (SSZ), and/or hydroxychloroquine (HCQ) during the study. The stated reasons
for discontinuation of prior anti TNF therapies were lack of efficacy (58%), intolerance (13%),
and/or reasons other than safety or efficacy (29%, mostly for financial reasons).
GO-BEFORE evaluated 637 patients with active RA who were MTX-naïve and had not previously
been treated with an anti-TNF agent. Patients were randomised to receive placebo + MTX, Simponi
50 mg + MTX, Simponi 100 mg + MTX or Simponi 100 mg + placebo. At week 52, patients entered
an open label long-term extension in which patients receiving placebo + MTX who had at least 1
tender or swollen joint were switched to Simponi 50 mg + MTX.
In GO-FORWARD, the (co-)primary endpoints were the percentage of patients achieving an ACR 20
response at week 14 and the improvement from baseline in Health Assessment Questionnaire (HAQ)
at week 24. In GO-AFTER, the primary endpoint was the percentage of patients achieving an ACR
20 response at week 14. In GO-BEFORE, the co-primary endpoints were the percentage of patients
achieving ACR 50 response at week 24 and the change from baseline in the van der Heijde-modified
Sharp (vdH-S) score at week 52. In addition to the primary endpoint(s), additional assessments of the
impact of Simponi treatment on the signs and symptoms of arthritis, physical function and
health-related quality of life were performed.
In general, no clinically meaningful differences in measures of efficacy were observed between the
Simponi 50 mg and 100 mg dosing regimens with concomitant MTX.
Signs and symptoms
Key ACR results for the Simponi 50 mg dose at weeks 14,24 and 52 for GO-FORWARD, GO-
AFTER and GO-BEFORE are shown in Table 2 and are described below. Responses were observed
at the first assessment (week 4) after the initial Simponi administration.
In GO-FORWARD, among 89 subjects randomised to Simponi 50 mg + MTX, 48 were still on this
treatment at week 104. Among those, 40, 33 and 24 patients had ACR 20/50/70 response,
respectively at week 104.
In GO-AFTER, the percentage of patients achieving an ACR 20 response was greater for patients
receiving Simponi than for patients receiving placebo regardless of the reason reported for
discontinuation of one or more prior anti-TNF therapies.
Table 2
Key efficacy outcomes from the controlled portions of GO-FORWARD, GO-AFTER and
GO-BEFORE.
GO-FORWARD
Active RA despite MTX
GO-AFTER
Active RA, previously
treated with one or more
anti-TNF agent(s)
GO-BEFORE
Active RA, MTX Naïve
Responders, % of patients
ACR 20
Week 14
ACR 70
Week 14 4% 14% p=0.008 2% 10% p=0.005 NA NA
Week 24 5% 20%* 2% 9% p=0.009 16% 24%
Week 52
NA NA NA NA 22% 28%
a nreflects randomised patients; actual number of patients evaluable for each endpoint may vary
by timepoint.
* p ≤ 0.001
NA: Not Applicable
In GO-BEFORE the primary analysis in patients with moderate to severe rheumatoid arthritis
(combined Simponi 50 and 100 mg + MTX groups vs MTX alone for ACR50) was not statistically
significant at week 24 (p=0.053). At week 52 in the overall population, the percentage of patients in
the Simponi 50 mg + MTX group who achieved an ACR response was generally higher but not
significantly different when compared with MTX alone (see Table 2). Additional analyses were
performed in subsets representative of the indicated population of patients with severe, active and
progressive RA. A generally greater effect of Simponi 50 mg + MTX versus MTX alone was
demonstrated in the indicated population compared with the overall population.
In GO-FORWARD and GO-AFTER, clinically meaningful and statistically significant responses in
Disease Activity Scale (DAS)28 were observed at each prespecified time point, at week 14 and at
week 24 ( p ≤ 0.001). Among patients who remained on the Simponi treatment to which they were
randomised at study start, DAS28 responses were maintained through week 104.
In GO-BEFORE, major clinical response, defined as the maintenance of an ACR 70 response over a
continuous 6-month period, was measured. At week 52, 15% of patients in the Simponi 50 mg +
MTX group achieved a major clinical response compared with 7% of patients in the placebo + MTX
group (p = 0.018). Among 159 subjects randomised to Simponi 50 mg + MTX, 96 were still on this

























treatment at week 104. Among those, 85, 66 and 53 patients had ACR 20/50/70 response,
respectively, at week 104.
Radiographic Response:
In GO-BEFORE the change from baseline in the vdH-S score, a composite score of structural
damage that radiographically measures the number and size of joint erosions and the degree of joint
space narrowing in hands/wrists and feet, was used to assess the degree of structural damage. Key
results for the Simponi 50 mg dose at week 52 are presented in Table 3.
The number of patients with no new erosions or a change from baseline in total vdH-S Score ≤ 0 was
significantly higher in the Simponi treatment group than in the control group (p = 0.003). The
radiographic effects observed at week 52 were maintained through week 104.
Table 3
Radiographic Mean (SD) Changes from Baseline in Total vdH-S Score at week 52 in the overall
population of GO-BEFORE
a
n reflects randomized patients
*
p = 0.015
**
p = 0.044
Physical function and health-related quality of life
Physical function and disability were assessed as a separate endpoint in GO-FORWARD and
GO-AFTER using the disability index of the HAQ. In these studies, Simponi demonstrated clinically
meaningful and statistically significant improvement in HAQ from baseline versus control at week
24. Among patients who remained on the Simponi treatment to which they were randomised at study
start, improvement in HAQ was maintained through week 104.
In GO-FORWARD clinically meaningful and statistically significant improvements were
demonstrated in health-related quality of life as measured by the physical component score of the
SF-36 in patients treated with Simponi versus placebo at week 24. Among patients who remained on
the Simponi treatment to which they were randomised at study start, improvement of the SF-36
physical component was maintained through week 104. In GO-FORWARD and GO-AFTER,
statistically significant improvements were observed in fatigue as measured by functional assessment
of chronic illness therapy-fatigue scale (FACIT-F).
Psoriatic arthritis
The safety and efficacy of Simponi were evaluated in a multi-centre, randomised, double-blind,
placebo-controlled study (GO-REVEAL) in 405 adult patients with active PsA (≥ 3 swollen joints
and ≥ 3 tender joints) despite non-steroidal anti-inflammatory (NSAID) or DMARD therapy. Patients
in this study had a diagnosis of PsA for at least 6 months and had at least mild psoriatic disease.
Patients with each sub-type of psoriatic arthritis were enrolled, including polyarticular arthritis with
no rheumatoid nodules (43%), asymmetric peripheral arthritis (30%), distal interphalangeal (DIP)
joint arthritis (15%), spondylitis with peripheral arthritis (11%), and arthritis mutilans (1%). Previous
















treatment with an anti-TNF agent was not allowed. Simponi or placebo were administered
subcutaneously every 4 weeks. Patients were randomly assigned to placebo, Simponi 50 mg, or
Simponi 100 mg.
Approximately forty-eight percent of patients continued on stable doses of methotrexate
(≤25 mg/week). The primary endpoint was the percentage of patients achieving ACR 20 response at
week 14. Placebo-controlled efficacy data were collected and analysed through week 24.
Key results for the 50 mg dose are shown in Table 4 and described below. In general, no clinically
meaningful differences in measures of efficacy were observed between the Simponi 50 mg and
100 mg dosing regimens.
Table 4
Key efficacy outcomes from GO-REVEAL
Responders, % of patients
ACR 20
ACR 70
Week 14 1 % 12 %
Week 24 1 % 19 %
PASI
b
75
c
Week 14
3 %
40 %
Week 24
1 %
56 %
* p < 0.05 for all comparisons; p-value calculations are based on comparisons
of median values for continuous variables
a
n reflects randomised patients; actual number of patients evaluable for each
endpoint may vary by timepoint
b Psoriasis Area and Severity Index
c
Based on the subset of patients with ≥ 3% BSA involvement at baseline, 79
patients (69.9%) in the placebo group and 109 (74.3%) in the Simponi 50 mg
group.
Statistically significant responses in DAS28 were also observed at weeks 14 and 24 (p < 0.05).
Improvements in key measures of disease activity were observed at the first assessment (week 4)
after the initial Simponi administration and were maintained through week 24. Similar ACR 20
responses at week 14 were observed in patients with polyarticular arthritis with no rheumatoid
nodules and asymmetric peripheral arthritis PsA subtypes. The number of patients with other PsA
subtypes was too small to allow meaningful assessment. Responses observed in the Simponi treated
groups were similar in patients receiving and not receiving concomitant MTX.
Improvements in parameters of peripheral activity characteristic of psoriatic arthritis (e.g. number of
swollen joints, number of painful/tender joints, dactylitis and enthesitis) were seen in the
Simponi-treated patients.
Simponi treatment resulted in significant improvement in physical function as assessed by HAQ, as
well as significant improvements in health-related quality of life as measured by the physical and
mental component summary scores of the SF-36.




















The safety and efficacy of Simponi were evaluated in a multi-centre, randomised, double-blind,
placebo-controlled study (GO-RAISE) in 356 adult patients with active ankylosing spondylitis
(defined as a Bath Ankylosing Spondylitis Disease Activity Index (BASDAI) ≥4 and a VAS for total
back pain of ≥4, on a scale of 0 to 10 cm). Patients enrolled in this study had active disease despite
current or previous NSAID or DMARD therapy and had not previously been treated with anti-TNF
therapy. Simponi or placebo were administered subcutaneously every 4 weeks. Patients were
randomly assigned to placebo, Simponi 50 mg and Simponi 100 mg and were allowed to continue
concomitant DMARD therapy (MTX, SSZ and/or HCQ). The primary endpoint was the percentage
of patients achieving Ankylosing Spondylitis Assessment Study Group (ASAS) 20 response at week
14. Placebo-controlled efficacy data were collected and analysed through week 24.
Key results for the 50 mg dose are shown in Table 5 and described below. In general, no clinically
meaningful differences in measures of efficacy were observed between the Simponi 50 mg and
100 mg dosing regimens.
Table 5
Key efficacy outcomes from GO-RAISE.
Responders, % of patients
ASAS 20
* p ≤ 0.001 for all comparisons
a
n reflects randomised patients; actual number of patients evaluable for each endpoint
may vary by timepoint
Statistically significant responses in BASDAI 50, 70 and 90 (p ≤ 0.017) were also seen at weeks 14
and 24. Improvements in key measures of disease activity were observed at the first assessment
(week 4) after the initial Simponi administration and were maintained through week 24. Consistent
efficacy was seen in patients regardless of use of DMARDs (MTX, sulfasalazine and/or
hydroxychloroquine), HLA-B27 antigen status or baseline CRP levels as assessed by ASAS 20
responses at week 14.
Simponi treatment resulted in significant improvements in physical function as assessed by changes
from baseline in BASFI at weeks 14 and 24. Health-related quality of life as measured by the
physical component score of the SF-36 was also improved significantly at weeks 14 and 24.
Immunogenicity
Across the Phase III RA, PsA and AS studies through week 52, antibodies to golimumab were
detected in 5% (105/2115) of golimumab treated patients and, where tested, nearly all antibodies
were neutralising
in vitro
. Similar rates were shown across rheumatologic indications. Treatment
with concomitant MTX resulted in a lower proportion of patients with antibodies to golimumab than
patients receiving golimumab without MTX (approximately 3% [41/1262] versus 8% [64/853],
respectively).
The presence of antibodies to golimumab may increase the risk of injection site reactions (see section
4.4).
The small number of patients positive for antibodies to golimumab limits the ability to draw






























definitive conclusions regarding the relationship between antibodies to golimumab and clinical
efficacy or safety measures.
Because immunogenicity analyses are product- and assay-specific, comparison of antibody rates with
those from other products is not appropriate.
Paediatric population
The European Medicines Agency has waived the obligation to conduct studies with Simponi in all
subsets of the paediatric population in ankylosing spondylitis and rheumatoid arthritis
(see section
4.2 for information on paediatric use).
The European Medicines Agency has deferred the obligation to submit the results of studies with
Simponi in one or more subsets of the paediatric population in juvenile idiopathic arthritis and
psoriatic arthritis (see section 4.2 for information on paediatric use).
Pharmacokinetic properties
Following a single subcutaneous administration of golimumab
to healthy subjects or patients with
RA, the median time to reach maximum serum concentrations (T
max
) ranged from 2 to 6 days. A
subcutaneous injection of 50 mg golimumab to healthy subjects produced a mean ± standard
deviation maximum serum concentration (C
max
) of 3.1 ± 1.4 μg/ml.
Golimumab exhibited approximately dose-proportional pharmacokinetics in patients with RA over
the dose range of 0.1 to 10.0 mg/kg following a single intravenous dose. The systemic clearance of
golimumab was estimated to be 6.9 ± 2.0 ml/day/kg, and mean volume of distribution was
115 ± 19 ml/kg. Terminal half-life value was estimated to be approximately 12 ± 3 days in healthy
subjects and similar values were observed in patients with RA, PsA or AS.
Following a single subcutaneous injection of 100 mg, the absorption of golimumab was similar in the
upper arm, abdomen, and thigh, with a mean absolute bioavailability of 51%. Since golimumab
exhibited approximately dose proportional PK following a subcutaneous administration, the absolute
bioavailability of the golimumab 50 mg dose is expected to be similar.
When 50 mg golimumab was administered subcutaneously to patients with RA, PsA or AS every 4
weeks, serum concentrations reached steady state by week 12. With concomitant use of MTX,
treatment with 50 mg golimumab subcutaneous every 4 weeks resulted in a mean (± standard
deviation) steady-state trough serum concentration of approximately 0.6 ± 0.4 μg/ml in RA patients
with active RA despite MTX therapy, and approximately 0.5 ± 0.4 μg/ml
in patients with active PsA
and approximately 0.8 ± 0.4 μg/ml in patients with AS.
Patients with RA, PsA or AS who did not receive concomitant MTX had approximately 30% lower
steady-state trough concentrations of golimumab than those who received golimumab with MTX. In
a limited number of RA patients treated with subcutaneous golimumab over a 6-month period,
concomitant use of MTX reduced the apparent clearance of golimumab by approximately 36%.
However, population pharmacokinetic analysis indicated that concomitant use of NSAIDs, oral
corticosteroids or sulfasalazine did not influence the apparent clearance of golimumab.
There was a trend toward higher apparent clearance of golimumab with increasing weight (see
section 4.2).
Patients who developed anti-golimumab antibodies generally had low trough steady-state serum
concentrations of golimumab (see section 5.1).
5.3 Preclinical safety data
Non-clinical data reveal no special hazard for humans based on conventional studies of safety
pharmacology, repeated dose toxicity, toxicity to reproduction and development.
No mutagenicity studies, animal fertility studies nor long-term carcinogenic studies have been
conducted with golimumab.
In a fertility and general reproductive function study in mouse, using an analogous antibody that
selectively inhibits the functional activity of mouse TNFα, the number of pregnant mice was reduced.
It is not known whether this finding was due to effects on the males and/or the females. In a
developmental toxicity study conducted in mice following administration of the same analogous
antibody, and in cynomolgus monkeys using golimumab, there was no indication of maternal
toxicity, embryotoxicity or teratogenicity.
PHARMACEUTICAL PARTICULARS
Sorbitol(E420)
L-histidine
L-histidine monohydrochloride monohydrate
Polysorbate 80
Water for injections.
In the absence of compatibility studies, this medicinal product must not be mixed with other
medicinal products.
6.4 Special precautions for storage
Store in a refrigerator (2°C – 8°C).
Do not freeze.
Keep the pre-filled syringe in the outer carton in order to protect it from light.
6.5
Nature and contents of container
0.5 ml solution in a pre-filled syringe (1.0 ml Type 1 glass) with a fixed needle (stainless steel) and a
needle cover (rubber containing latex) in a pre-filled syringe. Simponi is available in packs
containing 1 pre-filled syringe and multipacks containing 3 (3 packs of 1) pre-filled syringes.
Not all pack sizes may be marketed.
Simponi should not be used if the solution is discoloured, cloudy or containing visible foreign
particles.
6.6 Special precautions for disposal and other handling
Simponi is supplied in a single use pre-filled syringe. Each Simponi pack is provided with
instructions for use that fully describes the use of the syringe. After removing the pre-filled syringe
from the refrigerator the pre-filled syringe should be allowed to reach room temperature by waiting
30 minutes, before injecting Simponi. The syringe should not be shaken.
The solution is clear to slightly opalescent, colourless to light yellow and may contain a few small
translucent or white particles of protein. This appearance is not unusual for solutions containing
protein.
Comprehensive instructions for the preparation and administration of Simponi in a pre filled syringe
are given in the package leaflet.
Any unused product or waste material should be disposed of in accordance with local requirements.
MARKETING AUTHORISATION HOLDER
Janssen Biologics B.V.
Einsteinweg 101
2333 CB Leiden
The Netherlands
MARKETING AUTHORISATION NUMBER(S)
EU/1/09/546/003 1 pre-filled syringe
EU/1/09/546/004 3 pre-filled syringes
DATE OF FIRST AUTHORISATION
10. DATE OF REVISION OF THE TEXT
Detailed information on this medicinal product is available on the website of the European Medicines
Agency
http://www.ema.europa.eu
.
MANUFACTURER OF THE BIOLOGICAL ACTIVE
SUBSTANCE AND MANUFACTURING
AUTHORISATION HOLDER RESPONSIBLE FOR
BATCH RELEASE
CONDITIONS OF THE MARKETING AUTHORISATION
A. MANUFACTURERS OF THE BIOLOGICAL ACTIVE SUBSTANCE AND
MANUFACTURING AUTHORISATION HOLDER RESPONSIBLE FOR BATCH
RELEASE
Name and address of the manufacturers of the biological active substance
Janssen Biologics B.V.
Einsteinweg 101
NL-2333 CB Leiden
The Netherlands
Name and address of the manufacturer responsible for batch release
Janssen Biologics B.V.
Einsteinweg 101
NL-2333 CB Leiden
The Netherlands
B. CONDITIONS OF THE MARKETING AUTHORISATION
•
CONDITIONS OR RESTRICTIONS REGARDING SUPPLY AND USE IMPOSED ON
THE MARKETING AUTHORISATION HOLDER
Medicinal product subject to restricted medical prescription (See Annex I: Summary of Product
Characteristics, section 4.2).
•
CONDITIONS OR RESTRICTIONS WITH REGARD TO THE SAFE AND
EFFECTIVE USE OF THE MEDICINAL PRODUCT
The Marketing Authorisation Holder (MAH) shall ensure that, prior to launch, all physicians who are
expected to prescribe/use Simponi are provided with a physician information pack containing the
following:
•
The Summary of Product Characteristics
•
Physician information
•
Patient Alert Card
The physician information should contain the following key messages:
•
The risk of serious infections, including opportunistic bacterial, viral and fungal infections in
patients treated with Simponi,
•
The need to evaluate patients for both active and inactive tuberculosis prior to starting the
treatment, including use of appropriate screening tests,
•
The contraindication of Simponi in patients with history of moderate to severe heart failure
(NYHA III/IV), and potential risk of congestive heart failure being worsened by Simponi,
•
The risk of acute injection-related reactions and delayed serious systemic hypersensitivity
reactions, the need for instructing patients on techniques for administration, and guidance for
Health Care Professionals on how to report administration errors,
•
The role and use of patient alert card.
Pharmacovigilance system
The MAH must ensure that the system of pharmacovigilance, presented in Module 1.8.1. of the
Marketing Authorisation, is in place and functioning before and whilst the product is on the market.
The MAH commits to performing the studies and additional pharmacovigilance activities detailed in
the Pharmacovigilance Plan, as agreed in version 3.1 (dated 25 May 2010) of the Risk Management
Plan (RMP) presented in Module 1.8.2. of the Marketing Authorisation Application and any
subsequent updates of the RMP agreed by the CHMP.
As per the CHMP Guideline on Risk Management Systems for medicinal products for human use,
the updated RMP should be submitted at the same time as the next Periodic Safety Update Report
(PSUR).
In addition, an updated RMP should be submitted
•
When new information is received that may impact on the current Safety Specification,
Pharmacovigilance Plan or risk minimisation activities
•
Within 60 days of an important (pharmacovigilance or risk minimisation) milestone being
reached
•
At the request of the EMEA
ANNEX III
LABELLING AND PACKAGE LEAFLET
PARTICULARS TO APPEAR ON THE OUTER PACKAGING
NAME OF THE MEDICINAL PRODUCT
Simponi 50 mg solution for injection in pre-filled pen
golimumab
STATEMENT OF ACTIVE SUBSTANCE(S)
One 0.5 ml pre-filled pen contains 50 mg golimumab
Excipients: sorbitol (E420), L-histidine, L-histidine monohydrochloride monohydrate, polysorbate
80, water for injections. See package leaflet for further information.
PHARMACEUTICAL FORM AND CONTENTS
Solution for injection in pre-filled pen (SmartJect)
1 pre-filled pen
METHOD AND ROUTE(S) OF ADMINISTRATION
Subcutaneous use
Do not shake
Read the package leaflet before use
SPECIAL WARNING THAT THE MEDICINAL PRODUCT MUST BE STORED OUT
OF THE REACH AND SIGHT OF CHILDREN
Keep out of the reach and sight of children
OTHER SPECIAL WARNING(S), IF NECESSARY
The needle cover contains latex rubber. See the package leaflet for further information.
Allow the pen to sit at room temperature outside the box for 30 minutes before use.



















SPECIAL STORAGE CONDITIONS
Store in a refrigerator
Do not freeze
Keep the pre-filled pen in the outer carton in order to protect from light
10. SPECIAL PRECAUTIONS FOR DISPOSAL OF UNUSED MEDICINAL PRODUCTS
OR WASTE MATERIALS DERIVED FROM SUCH MEDICINAL PRODUCTS, IF
APPROPRIATE
11. NAME AND ADDRESS OF THE MARKETING AUTHORISATION HOLDER
Janssen Biologics B.V.
Einsteinweg 101
2333 CB Leiden
The Netherlands
12. MARKETING AUTHORISATION NUMBER(S)
14. GENERAL CLASSIFICATION FOR SUPPLY
Medicinal product subject to medical prescription
16. INFORMATION IN BRAILLE




















PARTICULARS TO APPEAR ON THE OUTER PACKAGING
CARTON FOR 1 PRE-FILLED PEN AS INTERMEDIATE PACK / COMPONENT OF A
MULTIPACK (WITHOUT BLUE BOX)
NAME OF THE MEDICINAL PRODUCT
Simponi 50 mg solution for injection in pre-filled pen
golimumab
STATEMENT OF ACTIVE SUBSTANCE(S)
One 0.5 ml pre-filled pen contains 50 mg golimumab
Excipients: sorbitol (E420), L-histidine, L-histidine monohydrochloride monohydrate, polysorbate
80, water for injections. See package leaflet for further information.
PHARMACEUTICAL FORM AND CONTENTS
Solution for injection in pre-filled pen (Smartject)
Component of a multipack comprising 3 packs, each containing 1 pre-filled pen
METHOD AND ROUTE(S) OF ADMINISTRATION
Subcutaneous use
Do not shake
Read the package leaflet before use
SPECIAL WARNING THAT THE MEDICINAL PRODUCT MUST BE STORED OUT
OF THE REACH AND SIGHT OF CHILDREN
Keep out of the reach and sight of children
OTHER SPECIAL WARNING(S), IF NECESSARY
The needle cover contains latex rubber. See the package leaflet for further information.
Allow the pen to sit at room temperature outside the box for 30 minutes before use.



















SPECIAL STORAGE CONDITIONS
Store in a refrigerator
Do not freeze
Keep the pre-filled pen in the outer carton in order to protect from light
10. SPECIAL PRECAUTIONS FOR DISPOSAL OF UNUSED MEDICINAL PRODUCTS
OR WASTE MATERIALS DERIVED FROM SUCH MEDICINAL PRODUCTS, IF
APPROPRIATE
NAME AND ADDRESS OF THE MARKETING AUTHORISATION HOLDER
Janssen Biologics B.V.
Einsteinweg 101
2333 CB Leiden
The Netherlands
MARKETING AUTHORISATION NUMBER(S)
GENERAL CLASSIFICATION FOR SUPPLY
Medicinal product subject to medical prescription



















PARTICULARS TO APPEAR ON THE OUTER PACKAGING
CARTON FOR MULTIPACK COMPRISING 3 PACKS (INCLUDING BLUE BOX)
NAME OF THE MEDICINAL PRODUCT
Simponi 50 mg
solution for injection in pre-filled pen
golimumab
STATEMENT OF ACTIVE SUBSTANCE(S)
One 0.5 ml pre-filled pen contains 50 mg golimumab
Excipients: sorbitol (E420), L-histidine, L-histidine monohydrochloride monohydrate, polysorbate
80, water for injections. See package leaflet for further information.
PHARMACEUTICAL FORM AND CONTENTS
Solution for injection in pre-filled pen (SmartJect)
Multipack comprising 3 packs, each containing 1 pre-filled pen
METHOD AND ROUTE(S) OF ADMINISTRATION
Subcutaneous use
Do not shake
Read the package leaflet before use
SPECIAL WARNING THAT THE MEDICINAL PRODUCT MUST BE STORED OUT
OF THE REACH AND SIGHT OF CHILDREN
Keep out of the reach and sight of children
OTHER SPECIAL WARNING(S), IF NECESSARY
The needle cover contains latex rubber. See the package leaflet for further information.
Allow the pen to sit at room temperature outside the box for 30 minutes before use.





















SPECIAL STORAGE CONDITIONS
Store in a refrigerator
Do not freeze
Keep the pre-filled pen in the outer carton in order to protect from light
10. SPECIAL PRECAUTIONS FOR DISPOSAL OF UNUSED MEDICINAL PRODUCTS
OR WASTE MATERIALS DERIVED FROM SUCH MEDICINAL PRODUCTS, IF
APPROPRIATE
NAME AND ADDRESS OF THE MARKETING AUTHORISATION HOLDER
Janssen Biologics B.V.
Einsteinweg 101
2333 CB Leiden
The Netherlands
MARKETING AUTHORISATION NUMBER(S)
GENERAL CLASSIFICATION FOR SUPPLY
Medicinal product subject to medical prescription



















PARTICULARS TO APPEAR ON THE OUTER PACKAGING
Before you start using Simponi:
•
Please read the enclosed package leaflet
•
Do not shake the product
•
Check the expiration date and the security seal
•
Wait 30 minutes to allow the product to reach room temperature



PARTICULARS TO APPEAR ON THE OUTER PACKAGING
PRE-FILLED SYRINGE CARTON
NAME OF THE MEDICINAL PRODUCT
Simponi 50 mg solution for injection in pre-filled syringe
golimumab
STATEMENT OF ACTIVE SUBSTANCE(S)
One 0.5 ml pre-filled syringe contains 50 mg golimumab
Excipients: sorbitol (E420), L-histidine, L-histidine monohydrochloride monohydrate, polysorbate
80, water for injections. See package leaflet for further information.
PHARMACEUTICAL FORM AND CONTENTS
Solution for injection in pre-filled syringe
1 pre-filled syringe
METHOD AND ROUTE(S) OF ADMINISTRATION
Subcutaneous use
Do not shake
Read the package leaflet before use
SPECIAL WARNING THAT THE MEDICINAL PRODUCT MUST BE STORED OUT
OF THE REACH AND SIGHT OF CHILDREN
Keep out of the reach and sight of children
OTHER SPECIAL WARNING(S), IF NECESSARY
The needle cover contains latex rubber. See the package leaflet for further information.
Allow the syringe to sit at room temperature outside the box for 30 minutes before use.



















SPECIAL STORAGE CONDITIONS
Store in a refrigerator
Do not freeze
Keep the pre-filled syringe in the outer carton in order to protect from light
10. SPECIAL PRECAUTIONS FOR DISPOSAL OF UNUSED MEDICINAL PRODUCTS
OR WASTE MATERIALS DERIVED FROM SUCH MEDICINAL PRODUCTS, IF
APPROPRIATE
11. NAME AND ADDRESS OF THE MARKETING AUTHORISATION HOLDER
Janssen Biologics B.V.
Einsteinweg 101
2333 CB Leiden
The Netherlands
12. MARKETING AUTHORISATION NUMBER(S)
14. GENERAL CLASSIFICATION FOR SUPPLY
Medicinal product subject to medical prescription
16. INFORMATION IN BRAILLE




















PARTICULARS TO APPEAR ON THE OUTER PACKAGING
CARTON FOR 1 PRE-FILLED SYRINGE AS INTERMEDIATE PACK / COMPONENT OF
A MULTIPACK (WITHOUT BLUE BOX)
NAME OF THE MEDICINAL PRODUCT
Simponi 50 mg solution for injection in pre-filled syringe
golimumab
STATEMENT OF ACTIVE SUBSTANCE(S)
One 0.5 ml pre-filled syringe contains 50 mg golimumab
Excipients: sorbitol (E420), L-histidine, L-histidine monohydrochloride monohydrate, polysorbate
80, water for injections. See package leaflet for further information.
PHARMACEUTICAL FORM AND CONTENTS
Solution for injection in pre-filled syringe
Component of a multipack comprising 3 packs, each containing 1 pre-filled syringe
METHOD AND ROUTE(S) OF ADMINISTRATION
Subcutaneous use
Do not shake
Read the package leaflet before use
SPECIAL WARNING THAT THE MEDICINAL PRODUCT MUST BE STORED OUT
OF THE REACH AND SIGHT OF CHILDREN
Keep out of the reach and sight of children
OTHER SPECIAL WARNING(S), IF NECESSARY
The needle cover contains latex rubber. See the package leaflet for further information.
Allow the syringe to sit at room temperature outside the box for 30 minutes before use.



















SPECIAL STORAGE CONDITIONS
Store in a refrigerator
Do not freeze
Keep the pre-filled syringe in the outer carton in order to protect from light
10. SPECIAL PRECAUTIONS FOR DISPOSAL OF UNUSED MEDICINAL PRODUCTS
OR WASTE MATERIALS DERIVED FROM SUCH MEDICINAL PRODUCTS, IF
APPROPRIATE
NAME AND ADDRESS OF THE MARKETING AUTHORISATION HOLDER
Janssen Biologics B.V.
Einsteinweg 101
2333 CB Leiden
The Netherlands
MARKETING AUTHORISATION NUMBER(S)
GENERAL CLASSIFICATION FOR SUPPLY
Medicinal product subject to medical prescription



















PARTICULARS TO APPEAR ON THE OUTER PACKAGING
CARTON FOR MULTIPACK COMPRISING 3 PACKS (INCLUDING BLUE BOX)
NAME OF THE MEDICINAL PRODUCT
Simponi 50 mg
solution for injection in pre-filled syringe
golimumab
STATEMENT OF ACTIVE SUBSTANCE(S)
One 0.5 ml pre-filled syringe contains 50 mg golimumab
Excipients: sorbitol (E420), L-histidine, L-histidine monohydrochloride monohydrate, polysorbate
80, water for injections. See package leaflet for further information.
PHARMACEUTICAL FORM AND CONTENTS
Solution for injection in pre-filled syringe
Multipack comprising 3 packs, each containing 1 pre-filled syringe
METHOD AND ROUTE(S) OF ADMINISTRATION
Subcutaneous use
Do not shake
Read the package leaflet before use
SPECIAL WARNING THAT THE MEDICINAL PRODUCT MUST BE STORED OUT
OF THE REACH AND SIGHT OF CHILDREN
Keep out of the reach and sight of children
OTHER SPECIAL WARNING(S), IF NECESSARY
The needle cover contains latex rubber. See the package leaflet for further information.
Allow the syringe to sit at room temperature outside the box for 30 minutes before use.





















SPECIAL STORAGE CONDITIONS
Store in a refrigerator
Do not freeze
Keep the pre-filled syringe in the outer carton in order to protect from light
10. SPECIAL PRECAUTIONS FOR DISPOSAL OF UNUSED MEDICINAL PRODUCTS
OR WASTE MATERIALS DERIVED FROM SUCH MEDICINAL PRODUCTS, IF
APPROPRIATE
NAME AND ADDRESS OF THE MARKETING AUTHORISATION HOLDER
Janssen Biologics B.V.
Einsteinweg 101
2333 CB Leiden
The Netherlands
MARKETING AUTHORISATION NUMBER(S)
GENERAL CLASSIFICATION FOR SUPPLY
Medicinal product subject to medical prescription



















PARTICULARS TO APPEAR ON THE OUTER PACKAGING
Before you start using Simponi:
•
Please read the enclosed package leaflet
•
Do not shake the product
•
Check the expiration date and the security seal
•
Wait 30 minutes to allow the product to reach room temperature



Simponi Patient Alert Card
This patient alert card contains important safety information that you need to be aware of before and
during treatment with Simponi.
Show this card to any doctor involved in your treatment.
1.
Infections
When you are treated with Simponi, you might get infections more easily. Infections may progress
more rapidly and may be more severe. In addition, some previous infections may reappear.
1.1.
Prior to Simponi treatment:
•
Tell your doctor if you have an infection. You must not be treated with Simponi if you have
tuberculosis (TB) or any other severe infection.
•
You should be screened for TB. It is very important that you tell your doctor if you have ever had
TB, or if you have been in close contact with someone who has had TB.
Ask your doctor to
record the type and date of the last screening(s) for TB below:
Test _______________ Date _______________
Test _______________ Date _______________
•
Tell your doctor if you know or suspect you are a carrier of the hepatitis B virus.
1.2.
During and after Simponi treatment:
•
Seek medical attention immediately if you develop symptoms of an infection, such as fever,
tiredness, (persistent) cough, shortness of breath, or flu-like signs, weight loss, night sweats,
diarrhoea, wounds, dental problems and a burning feeling when urinating.
2.1.
Prior to Simponi treatment:
•
Tell your doctor if you have a history of heart failure. You must not be treated with Simponi if
you have moderate to severe heart failure.
2.2.
During and after Simponi treatment
•
If you develop symptoms of heart failure (e.g. shortness of breath or swelling of the feet) seek
medical attention immediately.
3.
Dates of Simponi treatment:
1st administration: _______________________
Subsequent administrations: ______________________________________________
______________________________________________
Patient’s Name: _____________________
Doctor’s Name: _____________________
Doctor’s Phone: _____________________
•
Please make sure you also have a list of all other medicines that you are using with you at any
visit to a health care professional.
•
Keep this card with you for 6 months after the last Simponi dose, since side effects may occur a
long time after your last dose of Simponi.
•
Read the Simponi package leaflet carefully before you start using this medicine.
PACKAGE LEAFLET: INFORMATION FOR THE USER
Simponi 50 mg solution for injection in a pre-filled pen
golimumab
Read all of this leaflet carefully before you start using this medicine.
-
Keep this leaflet. You may need to read it again.
-
If you have any further questions, ask your doctor or pharmacist.
-
This medicine has been prescribed for you. Do not pass it on to others. It may harm them, even if
their symptoms are the same as yours.
-
If any of the side effects gets serious, or if you notice any side effects not listed in this leaflet,
please tell your doctor or pharmacist.
Your doctor will also give you a Patient Alert Card, which contains important safety information you
need to be aware of before and during your treatment with Simponi.
In this leaflet
:
1. What Simponi is and what it is used for
2. Before you use Simponi
3. How to use Simponi
4. Possible side effects
5. How to store Simponi
6. Further information
1.
WHAT SIMPONI IS AND WHAT IT IS USED FOR
Simponi contains the active substance called golimumab.
Simponi belongs to a group of medicines called ‘TNF blockers’. It is used in adults for the treatment
of the following inflammatory diseases:
•
Rheumatoid arthritis
•
Psoriatic arthritis
•
Ankylosing spondylitis
Simponi works by blocking the action of a protein called ‘tumour necrosis factor alpha’ (TNF-α).
This protein is involved in inflammatory processes of the body, and blocking it can reduce the
inflammation in your body.
Rheumatoid arthritis
Rheumatoid arthritis is an inflammatory disease of the joints
.
If you have active rheumatoid arthritis
you will first be given other medicines. If you do not respond well enough to these medicines, you
may be given Simponi which you will take in combination with another medicine called
methotrexate to:
•
Reduce the signs and symptoms of your disease.
•
Slow down the damage to your bones and joints.
•
Improve your physical function.
Psoriatic arthritis
Psoriatic arthritis is an inflammatory disease of the joints, usually accompanied by psoriasis, an
inflammatory disease of the skin. If you have active psoriatic arthritis you will first be given other
medicines. If you do not respond well enough to these medicines, you may be given Simponi to:
•
Reduce the signs and symptoms of your disease.
•
Improve your physical function.
Ankylosing spondylitis
Ankylosing spondylitis is an inflammatory disease of the spine. If you have ankylosing spondylitis
you will first be given other medicines. If you do not respond well enough to these medicines, you
may be given Simponi to:
•
Reduce the signs and symptoms of your disease.
•
Improve your physical function.
2.
BEFORE YOU USE SIMPONI
Do not use Simponi
•
If you are allergic (hypersensitive) to golimumab or any of the other ingredients of Simponi
(listed in Section 6 “What Simponi contains”).
•
If you have tuberculosis (TB) or any other severe infection.
•
If you have moderate or severe heart failure.
If you are not sure if any of the above applies to you, talk to your doctor or pharmacist before using
Simponi.
Take special care with Simponi
Tell your doctor before using Simponi if you have any of the following:
Infections
Tell your doctor straight away if you already have or get any symptoms of infection, during or after
your treatment with Simponi. Symptoms of infection include fever, cough, shortness of breath, flu-
like symptoms, diarrhoea, wounds, dental problems or a burning feeling when urinating.
•
You may get infections more easily while using Simponi.
•
Infections may progress more rapidly and may be more severe. In addition, some previous
infections may reappear.
Tuberculosis (TB)
Tell your doctor straight away if symptoms of TB appear during or after your treatment.
Symptoms of TB include persistent cough, weight loss, tiredness, fever or night sweats.
•
Cases of TB have been reported in patients treated with Simponi. Your doctor will test you
to see if you have TB. Your doctor will record these tests on your Patient Alert Card.
•
It is very important that you tell your doctor if you have ever had TB, or if you have been in
close contact with someone who has had or has TB.
•
If your doctor feels that you are at risk of TB, you may be treated with medicines for TB
before you begin using Simponi.
Hepatitis B virus (HBV)
•
Tell your doctor if you are a carrier or if you have or have had HBV before you are given
Simponi.
•
Tell your doctor if you think you might be at risk of contracting HBV
•
Your doctor should test you for HBV
•
Treatment with TNF blockers such as Simponi may result in reactivation of HBV in patients
who carry this virus, which can be life-threatening in some cases.
Invasive fungal infections
If you have lived in or travelled to an area where infections caused by specific type of fungi that
can affect the lungs or other parts of the body (called histoplasmosis, coccidioidomycosis, or
blastomycosis), are common, tell your doctor straight away. Ask your doctor if you don’t know if
these fungal infections are common in the area in which you have lived or travelled
Talk to your doctor if you have had, or are due to have a vaccine.
•
You should not receive certain (live) vaccines while using Simponi.
Operations or dental procedures
•
Talk to your doctor if you are going to have any operations or dental procedures.
•
Tell your surgeon or dentist performing the procedure that you are having treatment with
Simponi by showing them your Patient Alert Card.
Cancer and lymphoma
Tell your doctor if you have ever been diagnosed with lymphoma (a type of blood cancer) or any
other cancer before you use Simponi.
•
If you use Simponi or other TNF blockers, your risk for developing lymphoma or another cancer
may increase.
•
Patients with severe rheumatoid arthritis and other inflammatory diseases, who have had the
disease for a long time, may be at higher than average risk of developing lymphoma.
•
There have been cases of cancers, including unusual types, in children and teenage patients
taking TNF-blocking agents, which sometimes resulted in death.
•
Patients with severe persistent asthma, chronic obstructive pulmonary disease (COPD), or are
heavy smokers may be at increased risk for cancer with Simponi treatment. If you have severe
persistent asthma, COPD or are a heavy smoker, you should discuss with your doctor whether
treatment with a TNF blocker is appropriate for you.
Heart failure
Tell your doctor straight away if you get new or worsening symptoms of heart failure. Symptoms of
heart failure include shortness of breath or swelling of your feet.
•
New and worsening congestive heart failure has been reported with TNF blockers.
•
If you have mild heart failure and you are being treated with Simponi, you must be closely
monitored by your doctor.
Nervous system disease
Tell your doctor straight away if you have ever been diagnosed with or develop symptoms of a
demyelinating disease such as multiple sclerosis. Symptoms may include changes in your vision,
weakness in your arms or legs or numbness or tingling in any part of your body. Your doctor will
decide if you should receive Simponi.
Autoimmune disease
Tell your doctor if you develop symptoms of a disease called lupus. Symptoms include persistent
rash, fever, joint pain and tiredness.
•
On rare occasions, people treated with TNF blockers have developed lupus.
Blood disease
In some patients the body may fail to produce enough of the blood cells that help your body fight
infections or help you to stop bleeding. If you develop a fever that does not go away, bruise or bleed
very easily or look very pale, call your doctor right away. Your doctor may decide to stop treatment.
If you are not sure if any of the above applies to you, talk to your doctor or pharmacist before using
Simponi.
Use in children
Simponi is not recommended for children and adolescents (younger than 18 years) because it has not
been studied in this age group.
Using other medicines
•
You should not take Simponi with medicines containing the active substance anakinra or
abatacept. These medicines are used for the treatment of rheumatic diseases.
•
Tell your doctor or pharmacist if you are taking any other medicines that affect your immune
system.
•
You should not receive certain (live) vaccines while using Simponi.
•
Tell your doctor or pharmacist if you are using or have recently used any other medicines,
including medicines obtained without a prescription.
If you are not sure if any of the above applies to you, talk to your doctor or pharmacist before using
Simponi.
Pregnancy and breast-feeding
Talk to your doctor before using Simponi if:
•
You are pregnant or are planning to become pregnant while using Simponi. The effects of
this medicine in pregnant women are not known. The use of Simponi in pregnant women is
not recommended. If you are being treated with Simponi, you must avoid becoming pregnant
by using adequate contraception during your treatment and for at least 6 months after the last
Simponi injection.
•
You are a (potential) nursing mother. Before starting breast-feeding, your last treatment with
Simponi must be at least 6 months ago. You must stop breast-feeding if you are to be given
Simponi.
Ask your doctor or pharmacist for advice before taking any medicine, if you are pregnant or breast-
feeding.
Driving and using machines
Simponi may have a minor influence on your ability to drive and use tools or machines. Dizziness
may occur after you take Simponi. If this happens, do not drive or use any tools or machines.
Important information about some of the ingredients of Simponi
Latex sensitivity
A part of the pre-filled pen, the needle cover, contains latex. Because latex may cause severe allergic
reactions, talk to your doctor before using Simponi if you or your carer are allergic to latex.
Sorbitol intolerance
Simponi contains sorbitol (E420). If you have been told by your doctor that you have an intolerance
to some sugars, contact your doctor before taking this medicine.
Always use Simponi exactly as your doctor has told you. You should check with your doctor if you
are not sure.
How much Simponi is given
•
The recommended dose is 50 mg (the content of 1 pre-filled pen) given once a month, on the
same date each month.
•
Talk to your doctor before taking your fourth dose. Your doctor will determine if you should
continue Simponi treatment.
o
If you weigh more than 100 kg, the dose might be increased to 100 mg (the content
of 2 pre-filled pens) given once a month, on the same date each month.
How Simponi is given
•
Simponi is given by injection under the skin (subcutaneously).
•
At the start, your doctor or nurse may inject Simponi. However, you and your doctor may
decide that you may inject Simponi yourself. In this case you will get training on how to
inject Simponi yourself.
Talk to your doctor if you have any questions about giving yourself an injection. You will find
detailed “Instructions for administration” at the end of this leaflet.
If you use more Simponi than you should
If you have used or been given too much Simponi (either by injecting too much on a single occasion,
or by using it too often), talk to your doctor or pharmacist straight away. Always take the outer carton
with you, even if it is empty.
If you forget to use Simponi
If you forget to use Simponi on your planned date, inject the forgotten dose as soon as you
remember.
Do not use a double dose to make up for a forgotten dose.
When to inject your next dose:
•
If you were less than 2 weeks late, inject the forgotten dose as soon as you remember and
stay on your original schedule.
•
If you were more than 2 weeks late, inject the forgotten dose as soon as you remember and
talk to your doctor or pharmacist to ask when you need to take the next dose.
If you are not sure what to do, talk your doctor or pharmacist.
If you stop using Simponi
If you are considering stopping Simponi, talk to your doctor or pharmacist first.
If you have any further questions on the use of this medicine, ask your doctor or pharmacist.
Like all medicines, Simponi can cause side effects, although not everybody gets them. Some patients
may experience serious side effects and may require treatment. The risk of certain side effects is
greater with the 100 mg dose compared with the 50 mg dose. Side effects may appear up to several
months after the last injection.
Tell your doctor straight away if you notice any of the following:
•
Symptoms of an allergic reaction
such as swelling of the face, lips, mouth or throat which may
cause difficulty in swallowing or breathing, skin rash, hives, swelling of the hands, feet or ankles.
•
Symptoms of an infection (including TB)
such as fever, tiredness, (persistent) cough, shortness
of breath, flu-like symptoms, weight loss, night sweats, diarrhoea, wounds, dental problems and a
burning feeling when urinating.
•
Symptoms of liver problems
such as yellowing of the skin and eyes, dark brown-coloured
urine, right-sided abdominal pain, fever, feeling sick, being sick, and feeling very tired .
Side effects may occur with certain frequencies, which are defined as follows:
Very common (affects more than 1 user in 10)
Common (affects 1 to 10 users in 100)
Uncommon (affects 1 to 10 users in 1,000)
Rare (affects 1 to 10 users in 10,000)
Very rare (affects less than 1 user in 10,000)
Not known (frequency cannot be estimated from the available data)
The following side effects have been observed with Simponi:
Very common side effects:
•
Upper respiratory tract infections, sore throat or hoarseness, runny nose
Common side effects:
•
Abnormal liver tests (increased liver enzymes) found during blood tests done by your doctor
•
Feeling dizzy
•
Headache
•
Superficial fungal infections
•
Bacterial infections (such as cellulitis)
•
Low red blood cell counts
•
Positive blood lupus test
•
Difficulty sleeping
•
Depression
•
Constipation
•
Hair loss
•
Allergic reactions
•
Rash and itching of the skin
•
Indigestion
•
Stomach pain
•
Feeling numb or having a tingling feeling
•
Flu
•
Bronchitis
•
Sinus infection
•
Cold sores
•
High blood pressure
•
Fever
•
Injection site reactions (including redness, hardness, pain, bruising, itching, tingling and
irritation)
•
Feeling weak
•
Impaired healing
•
Chest discomfort
Uncommon side effects:
•
Serious blood infections
•
TB
•
lower respiratory tract infection (including pneumonia)
•
Infection of the joints or the tissue around them
•
Kidney infection
•
Abscess
•
Infections which may occur in people with weakened immune system, such as fungal
infections affecting lungs or other body parts
•
cancers, including skin cancer and non-cancerous growths or lumps, including skin moles
•
Psoriasis (including on the palms of your hand and/or the soles of your feet and/or in the
form of skin blisters)
•
Nervous system disease (symptoms may include changes in vision, weak arms or legs)
•
Low platelet count
•
Low white blood cell counts
•
Combined low platelet, red, and white blood cell count
•
Thyroid disorders
•
Increase in blood sugar levels
•
Increase in blood cholesterol levels
•
Balance disorders
•
Taste disturbances
•
Vision disturbances
•
New onset or worsening congestive heart failure
•
Sensation of heart beating irregularly
•
Narrowing of the blood vessels in the heart
•
Blood clots
•
Pain and discoloration in the fingers or toes
•
Flushing
•
Asthma, shortness of breath, wheezing
•
Stomach and bowel disorders which include inflammation of the stomach lining and colon
which may cause fever
•
Acid reflux
•
Pain and ulcers in the mouth
•
Gall stones
•
Liver disorders
•
Bladder disorders
•
Breast disorders
•
Menstrual disorders
•
Bone fractures
•
Inflammation of the blood vessels in your skin which results in rash
Rare side effects:
•
Cancer of the lymph system
•
Hepatitis B reactivation
•
Chronic inflammatory condition of the lungs
•
Lupus-like syndrome
•
Kidney disorders
•
Serious systemic allergic reactions (including anaphylactic reactions)
•
Inflammation of blood vessels in internal organs
Side effects of which the frequency is not known:
•
Failure of the bone marrow to produce blood cells
•
Leukaemia
If any of the side effects gets serious or if you notice any side effects not listed in this leaflet, please
tell your doctor or pharmacist.
Keep out of the reach and sight of children.
Do not use Simponi after the expiry date which is stated on the label and the carton after “EXP”. The
expiry date refers to the last day of that month.
Store in a refrigerator (2°C-8°C). Do not freeze.
Keep the pre-filled pen in the outer carton in order to protect it from light.
Medicines should not be disposed of via wastewater or household waste. Ask your doctor or
pharmacist how to dispose of medicines no longer required. These measures will help to protect the
environment.
What Simponi contains
The active substance is golimumab. One 0.5 ml pre-filled pen contains 50 mg of golimumab.
The other ingredients are sorbitol (E420),
L-histidine, L-histidine monohydrochloride monohydrate,
polysorbate 80 and water for injections.
What Simponi looks like and contents of the pack
Simponi is supplied as solution for injection in a single-use pre-filled pen.
Simponi is available in
packs containing 1 pre-filled pen and multipacks containing 3 (3 packs of 1) pre-filled pens. Not all
pack sizes may be marketed.
The solution is clear to slightly opalescent (having a pearl-like shine), colourless to light yellow and
may contain a few small translucent or white particles of protein. Do not use Simponi if the solution
is discoloured, cloudy or you can see foreign particles in it.
Marketing Authorisation Holder and Manufacturer
Janssen Biologics B.V.
Einsteinweg 101
2333 CB Leiden
The Netherlands
For any information about this medicine, please contact the local representative of the Marketing
Authorisation Holder:
België/Belgique/Belgien
N.V. Schering-Plough S.A.
Rue de Stalle/Stallestraat 73
B-1180 Bruxelles/Brussel/Brüssel
Tél/Tel: + 32-(0)2 370 92 11
Luxembourg/Luxemburg
N.V. Schering-Plough S.A.
Rue de Stalle 73
B-1180 Bruxelles/Brüssel
Belgique/Belgien
Tél/Tel: + 32-(0)2 370 92 11
България
Шеринг-Плау Сентрал Ийст - клон България
Ийст Парк Трейд Център
Бул. „Н.Й.Вапцаров” 53А, ет. 2
BG-София 1407
Тел.: +359 2 806 3030
Magyarország
Schering-Plough Hungary Kft.
Alkotás u. 53.
H-1123 Budapest
Tel.:+36 1 457-8500
Česká republika
Schering-Plough, s.r.o.
Ke Štvanici 3,
186 00 Praha 8
Tel: +420 221771250
Malta
Associated Drug Co. Ltd
Triq l-Esportaturi
Mriehel
Birkirkara BKR 3000
Tel.: +35622778000
Danmark
Schering-Plough A/S
Lautrupbjerg 2
DK-2750 Ballerup
Tlf: + 45-44 39 50 00
Nederland
Schering-Plough Nederland B.V.
Walmolen 1
NL-3994 DL Houten
Tel: + 31-(0)800 999 90 00
Deutschland
Essex Pharma GmbH
Thomas-Dehler-Straße 27
D-81737 München
Tel: + 49-(0)89 627 31-0
Norge
Schering-Plough AS
Pb. 398
N-1326 Lysaker
Tlf: + 47 67 16 64 50
Eesti
Schering-Plough CE AG Estonian Branch
Järvevana tee 9
EE-11314 Tallinn
Tel: + 372 654 96 86
Österreich
Merck Sharp & Dohme Ges.m.b.H.
Am Euro Platz 2
A-1120 Wien
Tel: +43 (0) 1 26 044
Ελλάδα
MSD Α.Β.Φ.Ε.Ε.
Αγίου Δημητρίου 63
GR-174 55 Άλιμος
Τηλ: + 30-210 98 97 300
Polska
Schering-Plough Polska Sp. z o.o.
ul. Taśmowa 7
PL-02-677 Warszawa
Tel.: + 48-(0)22 478 41 50
España
Merck Sharp & Dohme de España, S.A.
Josefa Valcárcel, 38
E-28027 Madrid
Tel: + 34 91 3210600
Portugal
Schering-Plough Farma, Lda.
Rua Agualva dos Açores 16
P-2735-557 Agualva-Cacém
Tel: + 351-21 433 93 00
France
Schering-Plough
34 avenue Léonard de Vinci
F-92400 Courbevoie
Tél: + 33-(0)1 80 46 40 40
România
Schering Plough Central East AG
Şos. Bucureşti-Ploieşti, nr. 17-21, Băneasa Center,
et. 8, sector 1
Bucureşti, 013682-RO
Tel. + 40 21 233 35 30
Ireland
Schering-Plough Ltd
Shire Park
Welwyn Garden City
Hertfordshire AL7 1TW
United KingdomTel: + 44-(0)1 707 363 636
Slovenija
Schering-Plough CE AG
Dunajska 22
SI-1000 Ljubljana
Tel. + 386 01 3001070
Ísland
Vistor hf
Hörgatún 2
IS-210 Garðabær
Sími: + 354 535 70 00
Slovenská republika
Schering-Plough, s.r.o.
Strakova 5
SK-811 01 Bratislava
Tel.: + 421 (2) 5920 2712
Italia
Schering-Plough S.p.A.
Via fratelli Cervi snc
Centro Direzionale Milano Due
Palazzo Borromini
I-20090 Segrate (Milano)
Tel: + 39-02 21018.1
Suomi/Finland
Schering-Plough Oy
PL 86/PB 86
FIN-02151 Espoo/Esbo
Puh/Tel: + 358-(0)20-7570 300
Κύπρος
Μ.Σ. Ιακωβίδης & Σία Λτδ
Οδός Αγίου Νικολάου, 8
CY-1055 Λευκωσία
Τηλ: +357-22 757188
Sverige
Merck Sharp & Dohme (Sweden) AB Box 7125
S-192 07 Sollentuna
Tel: + 46-(0)8 626 14 00
Latvija
Schering Plough CE AG filiāle Latvijā
Bauskas 58a -401
Rīga LV-1004
Tel: + 371-7 21 38 25
United Kingdom
Schering-Plough Ltd
Shire Park
Welwyn Garden City
Hertfordshire AL7 1TW
Tel: + 44-(0)1707 363 636
Lietuva
UAB Schering-Plough
Kęstučio g. 65/40
LT 08124 Vilnius
Tel. + 370 52 101 868
This leaflet was last approved in.
Detailed information on this medicine is available on the European Medicines Agency web site:
http://www.ema.europa.eu/.
INSTRUCTIONS FOR ADMINISTRATION
If you would like to self inject Simponi, you must be trained by a healthcare professional to
prepare an injection and give it to yourself. If you have not been trained, please contact your
doctor, nurse or pharmacist to schedule a training session.
The diagram below (see figure 1) shows what the ”SmartJect” pre-filled pen looks like. In this leaflet
“SmartJect” pre-filled pen may sometimes be shortened to “pen”.
Preparing for use of the pen
•
Do not shake the pen at any time.
•
Do not remove the cap from the pen until instructed to do so.
1. Check expiry date
•
Check the expiration date (as indicated as “EXP”) on the pen.
•
You can also check the expiration date printed on the carton.
Do not use the pen if the expiration date has passed. Please contact your doctor or pharmacist for
assistance.
2. Check security seal
•
Check the security seal around the cap of the pen.
Do not use the pen if the seal is broken. Please contact your doctor or pharmacist.
3. Wait 30 minutes
•
To ensure proper injection, allow the pen to sit at room temperature outside the box for 30
minutes out of the reach of children.
Do not
warm the pen in any other way (for example, do not warm it in a microwave or in hot water).
Do not remove the pen’s cap while allowing it to reach room temperature.
4. Get the rest of your equipment ready
While you are waiting you can get the rest of your equipment ready, including an alcohol swab, a
cotton ball or gauze and a sharps container.
5. Check the liquid in the pen
•
Look through the viewing window to make sure that the liquid in the pen is clear to slightly
opalescent (having a pearl-like shine) and colourless to light yellow. The solution may
contain a few small translucent or white particles of protein.
•
You will also notice an air bubble, which is normal.
Do not use the pen if the liquid is the wrong colour, cloudy, or contains foreign particles.
If this
happens, talk to your doctor or pharmacist.

Choosing and preparing the injection site
1. Choose your injection site (see figure 2)
•
You usually inject the medicine into the front of the middle thighs.
•
You can also use the stomach (abdomen) below the belly button, except for the 5 cm area
directly underneath the belly button.
•
You can use both sites, regardless of your body type or size.
Do not inject into areas where the skin is tender, bruised, red, scaly, hard or has scars or stretch
marks.
2. Injection site selection for caregivers (see figure 3)
•
If a caregiver is giving you the injection, they can also use the outer area of the upper arms
•
Again, all sites mentioned can be used regardless of your body type or size.
3. Preparing injection site
•
Wash your hands thoroughly with soap and warm water.
•
Wipe the injection site with an alcohol swab.
•
Allow the skin to dry before injecting. Do not fan or blow on the clean area.
Do not touch this area again before giving the injection.
Injecting the medicine
The cap should not
be removed until you are ready to inject the medication. The medication should
be injected within 5 minutes after the cap has been removed.




1. Remove the cap (figure 4)
•
When you are ready to inject, twist the cap slightly to break the security seal.
•
Pull the cap off and throw it away.
Do not put the cap back on because it may damage the needle inside the pen. Do not use the pen if it
is dropped without the cap in place. If this happens please contact your doctor or pharmacist.
2. Push the pen against the skin (see figures 5 and 6)
•
Hold the pen comfortably in your hand.
•
Without pressing the button, hold the open end of the pen at a right angle (90-degree) against
your skin and push the pen down firmly (see figure 5).



Some people find that using their free hand to pinch and hold the skin at the injection site during
administration makes injecting easier. This makes the injection site a firmer, more stable surface
for injection (see figure 6).
3. Press button to inject (see figure 7)
•
Continue to push the pen firmly against your skin and press the raised part of the button.
Once the button is pressed, it will remain pressed in so you do not need to keep pressure on
it.
•
The first “click” means that the needle has been inserted and the injection has started. You
may or may not feel a needle prick at this time.
Do not lift the pen away from your skin. If you pull the pen away from your skin, you may not get
your full dose of medicine.
You will not be able to push in the button unless the pen is pushed firmly against the skin.
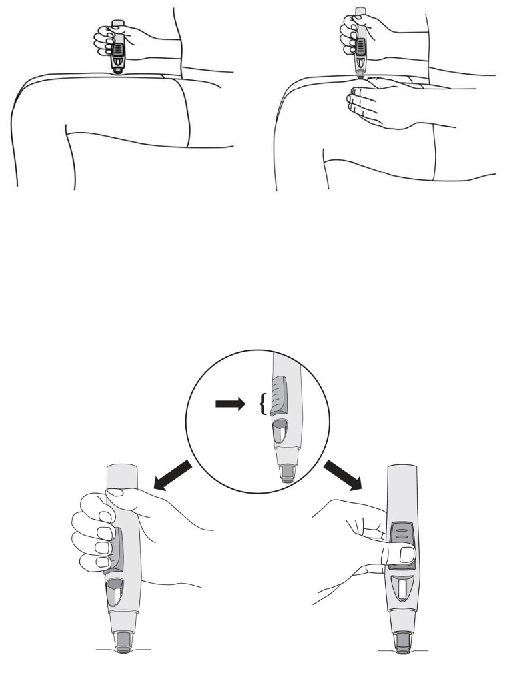
4. Continue to hold until the
second “click” (see figure 8)
•
Continue to hold the pen down firmly against your skin until you hear a second “click’. This
usually takes about 3-6 seconds, but may take up to 15 seconds.
•
The second “click” means that the injection is finished and the needle has gone back into the
pen.
•
Lift the pen from the injection site.
If you have hearing problem, count 15 seconds from the time you first press the button and then lift
the pen from the injection site.
1. Use cotton ball or gauze
•
There may be a small amount of blood or liquid at the injection site. This is normal.
•
You can press a cotton ball or gauze over the injection site for 10 seconds.
•
You may cover the injection site with a small adhesive bandage, if necessary.
Do not rub your skin.
2. Check the window (see figure 9)
•
After injecting, check the viewing window to make sure that a yellow indicator is visible.
•
This shows that the pen has worked properly.
•
If you do not think you received your injection, check the yellow indicator again to confirm
that the dose was delivered. Do not administer a second dose without speaking to your
doctor.
Talk to your doctor or pharmacist if the yellow indicator is not visible in the window.



3. Throw the pen away (see figure 10)
•
Place your pen in a sharps container straight away. Make sure you dispose of the bin as
instructed by your doctor or nurse.
If you feel that something has gone wrong with the injection or if you are not sure, talk to your doctor
or pharmacist.

PACKAGE LEAFLET: INFORMATION FOR THE USER
Simponi 50 mg solution for injection in a pre-filled syringe
golimumab
Read all of this leaflet carefully before you start using this medicine.
-
Keep this leaflet. You may need to read it again.
-
If you have any further questions, ask your doctor or pharmacist.
-
This medicine has been prescribed for you. Do not pass it on to others. It may harm them, even if
their symptoms are the same as yours.
-
If any of the side effects gets serious, or if you notice any side effects not listed in this leaflet,
please tell your doctor or pharmacist.
Your doctor will also give you a Patient Alert Card, which contains important safety information you
need to be aware of before and during your treatment with Simponi.
In this leaflet
:
1. What Simponi is and what it is used for
2. Before you use Simponi
3. How to use Simponi
4. Possible side effects
5.
How to store Simponi
6.
Further information
1.
WHAT SIMPONI IS AND WHAT IT IS USED FOR
Simponi contains the active substance called golimumab.
Simponi belongs to a group of medicines called ‘TNF blockers’. It is used in adults for the treatment
of the following inflammatory diseases:
•
Rheumatoid arthritis
•
Psoriatic arthritis
•
Ankylosing spondylitis.
Simponi works by blocking the action of a protein called ‘tumour necrosis factor alpha’ (TNF-α).
This protein is involved in inflammatory processes of the body, and blocking it can reduce the
inflammation in your body.
Rheumatoid arthritis
Rheumatoid arthritis is an inflammatory disease of the joints
.
If you have active rheumatoid arthritis
you will first be given other medicines. If you do not respond well enough to these medicines, you
may be given Simponi which you will take in combination with another medicine called
methotrexate to:
•
Reduce the signs and symptoms of your disease.
•
Slow down the damage to your bones and joints.
•
Improve your physical function.
Psoriatic arthritis:
Psoriatic arthritis is an inflammatory disease of the joints, usually accompanied by psoriasis, an
inflammatory disease of the skin. If you have active psoriatic arthritis you will first be given other
medicines. If you do not respond well enough to these medicines, you may be given Simponi to:
•
Reduce the signs and symptoms of your disease.
•
Improve your physical function.
Ankylosing spondylitis:
Ankylosing spondylitis is an inflammatory disease of the spine. If you have ankylosing spondylitis
you will first be given other medicines. If you do not respond well enough to these medicines, you
may be given Simponi to:
•
Reduce the signs and symptoms of your disease.
•
Improve your physical function.
2.
BEFORE YOU USE SIMPONI
Do not use Simponi:
•
If you are allergic (hypersensitive) to golimumab or any of the other ingredients of Simponi
(listed in Section 6 “What Simponi contains”).
•
If you have tuberculosis (TB) or any other severe infection.
•
If you have moderate or severe heart failure.
If you are not sure if any of the above applies to you, talk to your doctor or pharmacist before using
Simponi.
Take special care with Simponi
Tell your doctor before using Simponi if you have any of the following:
Infections:
Tell your doctor straight away if you already have or get any symptoms of infection, during or after
your treatment with Simponi. Symptoms of infection include fever, cough, shortness of breath, flu-
like symptoms, diarrhoea, wounds, dental problems or a burning feeling when urinating.
•
You may get infections more easily while using Simponi.
•
Infections may progress more rapidly and may be more severe. In addition, some previous
infections may reappear.
Tuberculosis (TB)
Tell your doctor straight away if symptoms of TB appear during or after your treatment.
Symptoms of TB include persistent cough, weight loss, tiredness, fever or night sweats.
•
Cases of TB have been reported in patients treated with Simponi. Your doctor will test you
to see if you have TB. Your doctor will record these tests on your Patient Alert Card.
•
It is very important that you tell your doctor if you have ever had TB, or if you have been in
close contact with someone who has had or has TB.
•
If your doctor feels that you are at risk of TB, you may be treated with medicines for TB
before you begin using Simponi.
Hepatitis B virus (HBV)
•
Tell your doctor if you are a carrier or if you have or have had HBV before you are given
Simponi.
•
Tell your doctor if you think you might be at risk of contracting HBV
•
Your doctor should test you for HBV
•
Treatment with TNF blockers such as Simponi may result in reactivation of HBV in patients
who carry this virus, which can be life-threatening in some cases.
Invasive fungal infections
If you have lived in or travelled to an area where infections caused by specific type of fungi that
can affect the lungs or other parts of the body (called histoplasmosis, coccidioidomycosis, or
blastomycosis), are common, tell your doctor straight away. Ask your doctor if you don’t know
if these fungal infections are common in the area in which you have lived or travelled.
Talk to your doctor if you have had, or are due to have a vaccine.
•
You should not receive certain (live) vaccines while using Simponi.
Operations or dental procedures:
•
Talk to your doctor if you are going to have any operations or dental procedures.
•
Tell your surgeon or dentist performing the procedure that you are having treatment with
Simponi by showing them your Patient Alert Card.
Cancer and lymphoma:
Tell your doctor if you have ever been diagnosed with lymphoma (a type of blood cancer) or any
other cancer before you use Simponi.
•
If you use Simponi or other TNF blockers, your risk for developing lymphoma or another cancer
may increase.
•
Patients with severe rheumatoid arthritis and other inflammatory diseases, who have had the
disease for a long time, may be at higher than average risk of developing lymphoma.
•
There have been cases of cancers, including unusual types, in children and teenage patients
taking TNF-blocking agents, which sometimes resulted in death.
•
Patients with severe persistent asthma, chronic obstructive pulmonary disease (COPD), or are
heavy smokers may be at increased risk for cancer with Simponi treatment. If you have severe
persistent asthma, COPD or are a heavy smoker, you should discuss with your doctor whether
treatment with a TNF blocker is appropriate for you.
Heart failure:
Tell your doctor straight away if you get new or worsening symptoms of heart failure. Symptoms of
heart failure include shortness of breath or swelling of your feet.
•
New and worsening congestive heart failure has been reported with TNF blockers.
•
If you have mild heart failure and you are being treated with Simponi, you must be closely
monitored by your doctor.
Nervous system disease:
Tell your doctor straight away if you have ever been diagnosed with or develop symptoms of a
demyelinating disease such as multiple sclerosis. Symptoms may include changes in your vision,
weakness in your arms or legs or numbness or tingling in any part of your body. Your doctor will
decide if you should receive Simponi.
Autoimmune disease:
Tell your doctor if you develop symptoms of a disease called lupus. Symptoms include persistent
rash, fever, joint pain and tiredness.
•
On rare occasions, people treated with TNF blockers have developed lupus.
Blood disease:
In some patients the body may fail to produce enough of the blood cells that help your body fight
infections or help you to stop bleeding. If you develop a fever that does not go away, bruise or bleed
very easily or look very pale, call your doctor right away. Your doctor may decide to stop treatment.
If you are not sure if any of the above applies to you, talk to your doctor or pharmacist before using
Simponi.
Use in children
Simponi is not recommended for children and adolescents (younger than 18 years) because it has not
been studied in this age group.
Using other medicines
•
You should not take Simponi with medicines containing the active substance anakinra or
abatacept. These medicines are used for the treatment of rheumatic diseases.
•
Tell your doctor or pharmacist if you are taking any other medicines that affect your immune
system.
•
You should not receive certain (live) vaccines while using Simponi.
•
Tell your doctor or pharmacist if you are using or have recently used any other medicines,
including medicines obtained without a prescription.
If you are not sure if any of the above applies to you, talk to your doctor or pharmacist before using
Simponi.
Pregnancy and breast-feeding
Talk to your doctor before using Simponi if:
•
You are pregnant or are planning to become pregnant while using Simponi. The effects of
this medicine in pregnant women are not known. The use of Simponi in pregnant women is
not recommended. If you are being treated with Simponi, you must avoid becoming pregnant
by using adequate contraception during your treatment and for at least 6 months after the last
Simponi injection.
•
You are a (potential) nursing mother. Before starting breast-feeding, your last treatment with
Simponi must be at least 6 months ago. You must stop breast-feeding if you are to be given
Simponi.
Ask your doctor or pharmacist for advice before taking any medicine, if you are pregnant or breast-
feeding.
Driving and using machines
Simponi may have a minor influence on your ability to drive and use tools or machines. Dizziness
may occur after you take Simponi. If this happens, do not drive or use any tools or machines.
Important information about some of the ingredients of Simponi
Latex sensitivity:
A part of the pre-filled syringe, the needle cover, contains latex. Because latex may cause severe
allergic reactions, talk to your doctor before using Simponi if you or your carer are allergic to latex.
Sorbitol intolerance:
Simponi contains sorbitol (E420). If you have been told by your doctor that you have an intolerance
to some sugars, contact your doctor before taking this medicine.
Always use Simponi exactly as your doctor has told you. You should check with your doctor if you
are not sure.
How much Simponi is given
•
The recommended dose is 50 mg (the content of 1 pre-filled syringe) given once a month, on
the same date each month.
•
Talk to your doctor before taking your fourth dose. Your doctor will determine if you should
continue Simponi treatment.
o
If you weigh more than 100 kg, the dose might be increased to 100 mg (the content
of 2 pre-filled syringes) given once a month, on the same date each month.
How Simponi is given
•
Simponi is given by injection under the skin (subcutaneously).
•
At the start, your doctor or nurse may inject Simponi. However, you and your doctor may
decide that you may inject Simponi yourself. In this case you will get training on how to
inject Simponi yourself.
Talk to your doctor if you have any questions about giving yourself an injection. You will find
detailed “Instructions for administration” at the end of this leaflet.
If you use more Simponi than you should
If you have used or been given too much Simponi (either by injecting too much on a single occasion,
or by using it too often), talk to your doctor or pharmacist straight away. Always take the outer carton
with you, even if it is empty.
If you forget to use Simponi
If you forget to use Simponi on your planned date, inject the forgotten dose as soon as you
remember.
Do not use a double dose to make up for a forgotten dose.
When to inject your next dose:
•
If you were less than 2 weeks late, inject the forgotten dose as soon as you remember and
stay on your original schedule.
•
If you were more than 2 weeks late, inject the forgotten dose as soon as you remember and
talk to your doctor or pharmacist to ask when you need to take the next dose.
If you are not sure what to do, talk your doctor or pharmacist.
If you stop using Simponi
If you are considering stopping Simponi, talk to your doctor or pharmacist first.
If you have any further questions on the use of this medicine, ask your doctor or pharmacist.
Like all medicines, Simponi can cause side effects, although not everybody gets them. Some patients
may experience serious side effects and may require treatment. The risk of certain side effects is
greater with the 100 mg dose compared with the 50 mg dose. Side effects may appear up to several
months after the last injection.
Tell your doctor straight away if you notice any of the following:
•
Symptoms of an allergic reaction
such as swelling of the face, lips, mouth or throat which may
cause difficulty in swallowing or breathing, skin rash, hives, swelling of the hands, feet or ankles.
•
Symptoms of an infection (including TB)
such as fever, tiredness, (persistent) cough, shortness
of breath, flu-like symptoms, weight loss, night sweats, diarrhoea, wounds, dental problems and a
burning feeling when urinating.
•
Symptoms of liver problems
such as yellowing of the skin and eyes, dark brown-coloured
urine, right-sided abdominal pain, fever, feeling sick, being sick, and feeling very tired.
Side effects may occur with certain frequencies, which are defined as follows:
Very common (affects more than 1 user in 10)
Common (affects 1 to 10 users in 100)
Uncommon (affects 1 to 10 users in 1,000)
Rare (affects 1 to 10 users in 10,000)
Very rare (affects less than 1 user in 10,000)
Not known (frequency cannot be estimated from the available data)
The following side effects have been observed with Simponi:
Very common side effects:
•
Upper respiratory tract infections, sore throat or hoarseness, runny nose
Common side effects:
•
Abnormal liver tests (increased liver enzymes) found during blood tests done by your doctor
•
Feeling dizzy
•
Headache
•
Superficial fungal infections
•
Bacterial infections (such as cellulitis)
•
Low red blood cell counts
•
Positive blood lupus test
•
Difficulty sleeping
•
Depression
•
Constipation
•
Hair loss
•
Allergic reactions
•
Rash and itching of the skin
•
Indigestion
•
Stomach pain
•
Feeling numb or having a tingling feeling
•
Flu
•
Bronchitis
•
Sinus infection
•
Cold sores
•
High blood pressure
•
Fever
•
Injection site reactions (including redness, hardness, pain, bruising, itching, tingling and
irritation)
•
Feeling weak
•
Impaired healing
•
Chest discomfort
Uncommon side effects:
•
Serious blood infections
•
TB
•
lower respiratory tract infection (including pneumonia)
•
Infection of the joints or the tissue around them
•
Kidney infection
•
Abscess
•
Infections which may occur in people with weakened immune system, such as fungal
infections affecting lungs or other body parts
•
cancers, including skin cancer and non-cancerous growths or lumps, including skin moles
•
Psoriasis (including on the palms of your hand and/or the soles of your feet and/or in the
form of skin blisters)
•
Nervous system disease (symptoms may include changes in vision, weak arms or legs)
•
Low platelet count
•
Low white blood cell counts
•
Combined low platelet, red, and white blood cell count
•
Thyroid disorders
•
Increase in blood sugar levels
•
Increase in blood cholesterol levels
•
Balance disorders
•
Taste disturbances
•
Vision disturbances
•
New onset or worsening congestive heart failure
•
Sensation of heart beating irregularly
•
Narrowing of the blood vessels in the heart
•
Blood clots
•
Pain and discoloration in the fingers or toes
•
Flushing
•
Asthma, shortness of breath, wheezing
•
Stomach and bowel disorders which include inflammation of the stomach lining and colon
which may cause fever
•
Acid reflux
•
Pain and ulcers in the mouth
•
Gall stones
•
Liver disorders
•
Bladder disorders
•
Breast disorders
•
Menstrual disorders
•
Bone fractures
•
Inflammation of the blood vessels in your skin which results in rash
Rare side effects:
•
Cancer of the lymph system
•
Hepatitis B reactivation
•
Chronic inflammatory condition of the lungs
•
Lupus-like syndrome
•
Kidney disorders
•
Serious systemic allergic reactions (including anaphylactic reactions)
•
Inflammation of the blood vessels in internal organs
Side effects of which the frequency is not known:
•
Failure of the bone marrow to produce blood cells
•
Leukaemia
If any of the side effects gets serious or if you notice any side effects not listed in this leaflet, please
tell your doctor or pharmacist.
Keep out of the reach and sight of children.
Do not use Simponi after the expiry date which is stated on the label and the carton after “EXP”. The
expiry date refers to the last day of that month.
Store in a refrigerator (2°C-8°C). Do not freeze.
Keep the pre-filled syringe in the outer carton in order to protect it from light.
Medicines should not be disposed of via wastewater or household waste. Ask your doctor or
pharmacist how to dispose of medicines no longer required. These measures will help to protect the
environment.
What Simponi contains
The active substance is golimumab. One 0.5 ml pre-filled syringe contains 50 mg of golimumab.
The other ingredients are sorbitol (E420),
L-histidine, L-histidine monohydrochloride monohydrate,
polysorbate 80 and water for injections.
What Simponi looks like and contents of the pack
Simponi is supplied as solution for injection in a single-use pre-filled syringe Simponi is available in
packs containing 1 pre filled syringe and multipacks containing 3 (3 packs of 1) pre-filled syringes.
Not all pack sizes may be marketed.
The solution is clear to slightly opalescent (having a pearl-like shine), colourless to light yellow and
may contain a few small translucent or white particles of protein. Do not use Simponi if the solution
is discoloured, cloudy or you can see foreign particles in it.
Marketing Authorisation Holder and Manufacturer
Janssen Biologics B.V.
Einsteinweg 101
2333 CB Leiden
The Netherlands
For any information about this medicine, please contact the local representative of the Marketing
Authorisation Holder:
België/Belgique/Belgien
N.V. Schering-Plough S.A.
Rue de Stalle/Stallestraat 73
B-1180 Bruxelles/Brussel/Brüssel
Tél/Tel: + 32-(0)2 370 92 11
Luxembourg/Luxemburg
N.V. Schering-Plough S.A.
Rue de Stalle 73
B-1180 Bruxelles/Brüssel
Belgique/Belgien
Tél/Tel: + 32-(0)2 370 92 11
България
Шеринг-Плау Сентрал Ийст - клон България
Ийст Парк Трейд Център
Бул. „Н.Й.Вапцаров” 53А, ет. 2
BG-София 1407
Тел.: +359 2 806 3030
Magyarország
Schering-Plough Hungary Kft.
Alkotás u. 53.
H-1123 Budapest
Tel.:+36 1 457-8500
Česká republika
Schering-Plough, s.r.o.
Ke Štvanici 3,
186 00 Praha 8
Tel: +420 221771250
Malta
Associated Drug Co. Ltd
Triq l-Esportaturi
Mriehel
Birkirkara BKR 3000
Tel.: +35622778000
Danmark
Schering-Plough A/S
Lautrupbjerg 2
DK-2750 Ballerup
Tlf: + 45-44 39 50 00
Nederland
Schering-Plough Nederland B.V.
Walmolen 1
NL-3994 DL Houten
Tel: + 31-(0)800 999 90 00
Deutschland
Essex Pharma GmbH
Thomas-Dehler-Straße 27
D-81737 München
Tel: + 49-(0)89 627 31-0
Norge
Schering-Plough AS
Pb. 398
N-1326 Lysaker
Tlf: + 47 67 16 64 50
Eesti
Schering-Plough CE AG Estonian Branch
Järvevana tee 9
EE-11314 Tallinn
Tel: + 372 654 96 86
Österreich
Merck Sharp & Dohme Ges.m.b.H.
Am Euro Platz 2
A-1120 Wien
Tel: +43 (0) 1 26 044
Ελλάδα
MSD Α.Β.Φ.Ε.Ε.
Αγίου Δημητρίου 63
GR-174 55 Άλιμος
Τηλ: + 30-210 98 97 300
Polska
Schering-Plough Polska Sp. z o.o.
ul. Taśmowa 7
PL-02-677 Warszawa
Tel.: + 48-(0)22 478 41 50
España
Merck Sharp & Dohme de España, S.A.
Josefa Valcárcel, 38
E-28027 Madrid
Tel: + 34 91 3210600
Portugal
Schering-Plough Farma, Lda.
Rua Agualva dos Açores 16
P-2735-557 Agualva-Cacém
Tel: + 351-21 433 93 00
France
Schering-Plough
34 avenue Léonard de Vinci
F-92400 Courbevoie
Tél: + 33-(0)1 80 46 40 40
România
Schering Plough Central East AG
Şos. Bucureşti-Ploieşti, nr. 17-21, Băneasa Center,
et. 8, sector 1
Bucureşti, 013682-RO
Tel. + 40 21 233 35 30
Ireland
Schering-Plough Ltd
Shire Park
Welwyn Garden City
Hertfordshire AL7 1TW
United KingdomTel: + 44-(0)1 707 363 636
Slovenija
Schering-Plough CE AG
Dunajska 22
SI-1000 Ljubljana
Tel. + 386 01 3001070
Ísland
Vistor hf
Hörgatún 2
IS-210 Garðabær
Sími: + 354 535 70 00
Slovenská republika
Schering-Plough, s.r.o.
Strakova 5
SK-811 01 Bratislava
Tel.: + 421 (2) 5920 2712
Italia
Schering-Plough S.p.A.
Via fratelli Cervi snc
Centro Direzionale Milano Due
Palazzo Borromini
I-20090 Segrate (Milano)
Tel: + 39-02 21018.1
Suomi/Finland
Schering-Plough Oy
PL 86/PB 86
FIN-02151 Espoo/Esbo
Puh/Tel: + 358-(0)20-7570 300
Κύπρος
Μ.Σ. Ιακωβίδης & Σία Λτδ
Οδός Αγίου Νικολάου, 8
CY-1055 Λευκωσία
Τηλ: +357-22 757188
Sverige
Merck Sharp & Dohme (Sweden) AB Box 7125
S-192 07 Sollentuna
Tel: + 46-(0)8 626 14 00
Latvija
Schering Plough CE AG filiāle Latvijā
Bauskas 58a -401
Rīga LV-1004
Tel: + 371-7 21 38 25
United Kingdom
Schering-Plough Ltd
Shire Park
Welwyn Garden City
Hertfordshire AL7 1TW
Tel: + 44-(0)1707 363 636
Lietuva
UAB Schering-Plough
Kęstučio g. 65/40
LT 08124 Vilnius
Tel. + 370 52 101 868
This leaflet was last approved in.
Detailed information on this medicine is available on the European Medicines Agency web site:
http://www.ema.europa.eu/.
INSTRUCTIONS FOR ADMINISTRATION
If you would like to self inject Simponi, you must be trained by a healthcare professional to
prepare an injection and give it to yourself. If you have not been trained, please contact your
doctor, nurse or pharmacist to schedule a training session.
The diagram below (see figure 1) shows what the pre-filled syringe looks like. In this leaflet “pre-
filled syringe” may sometimes be shortened to ”syringe”.
Preparing for use of the syringe
Hold the pre-filled syringe by the body of the syringe
•
Do not hold by the plunger head, plunger, needle guard wings, or needle cover.
•
Do not pull back on the plunger at any time.
•
Do not shake the pre-filled syringe at any time.
•
Do not remove the needle cover from the pre-filled syringe until instructed to do so.
•
Do not touch the needle guard activation clips (as indicated by asterisks * in figure 1) to
prevent prematurely covering the needle with the needle guard.
1. Check expiry date (see figure 2)
•
Check the expiration date (as indicated by “EXP”) on the label by looking through the
viewing window located within the body of the pre-filled syringe.
•
If you cannot see the expiration date through the viewing window, hold the pre-filled syringe
by its body and rotate the needle cover to line up the expiration date to the viewing window.
•
You can also check the expiration date printed on the carton.
Do not use the pre-filled syringe if the expiration date has passed. Please contact your doctor or
pharmacist for assistance.


2. Wait 30 minutes
•
To ensure proper injection, allow the syringe to sit at room temperature outside the box for
30 minutes, out of the reach of children.
Do not
warm the syringe in any other way (for example, do not warm it in a microwave or in hot
water).
Do not remove the syringe’s needle cover while allowing it to reach room temperature.
3. Get the rest of your equipment ready
While you are waiting you can get the rest of your equipment ready, including an alcohol swab, a
cotton ball or gauze and a sharps container.
4. Check the liquid in the syringe
•
Hold the pre-filled syringe by its body with the covered needle pointing downward.
•
Look at the liquid through the viewing window of the syringe and make sure that it is clear to
slightly opalescent (having a pearl-like shine) and colourless to light yellow. The solution
may contain a few small translucent or white particles of protein.
•
If you cannot see the liquid through the viewing window, hold the pre-filled syringe by its
body and rotate the needle cover to line up the liquid to the viewing window (see figure 2).
Do not
use the syringe if the liquid is the wrong colour, cloudy, or contains foreign particles.
If this
happens, talk to your doctor or pharmacist.
Choosing and preparing the injection site
1. Choose your injection site (see figure 3)
•
You usually inject the medicine into the front of the middle thighs.
•
You can also use the lower stomach (abdomen) below the belly button, except for the 5 cm
area directly underneath the belly button.
•
You can use both sites, regardless of your body type or size.
Do not inject into areas where the skin is tender, bruised, red, scaly, hard or has scars or stretch
marks.
2. Injection site selection for caregivers (see figure 4)
•
If a caregiver is giving you the injection, they can also use the outer area of the upper arms.
•
Again, all sites mentioned can be used regardless of your body type or size.






3. Preparing injection site
•
Wash your hands thoroughly with soap and warm water.
•
Wipe the injection site with an alcohol swab.
•
Allow the skin to dry before injecting. Do not fan or blow on the clean area.
Do not touch this area again before giving the injection.
Injecting the medicine
The needle cover should not
be removed until you are ready to inject the medication. The medication
should be injected within 5 minutes after the needle cover has been removed.
Do not
touch the plunger during needle cover removal.
1. Remove the needle cover (see figure 5)
•
When you are ready to inject, hold the body of the syringe with one hand.
•
Pull the needle cover straight off and throw it away. Do not
touch the plunger while you do
this.
•
You may notice an air bubble in the pre-filled syringe or a drop of liquid at the end of the
needle. These are both normal and do not need to be removed.
•
Inject the dose promptly after removing the needle cover.
Do not touch the needle or allow it to touch any surface.
Do not use the syringe if it is dropped without the needle cover in place. If this happens, please
contact your doctor or
pharmacist.
2. Position the syringe to inject
•
Hold the body of the syringe with one hand between the middle and index fingers and place
the thumb on top of the plunger head and use the other hand to gently pinch the area of skin
that you previously cleaned. Hold firmly.
Do not pull back on the plunger at any time.
3. Inject the medicine
•
Place the needle at approximately a 45-degree angle to the pinched skin. In a single and swift
motion, insert the needle through the skin as far as it will go (see figure 6).

•
Inject all of the medication by pushing in the plunger until the plunger head is completely
between the needle guard wings (see figure 7).
•
When the plunger is pushed as far as it will go, continue to keep the pressure on the plunger
head, take out the needle and let go of the skin (see figure 8).
Slowly take your thumb off the plunger head to allow the empty syringe to move up until the
entire needle is covered by the needle guard, as shown by the figure 9:







1. Use cotton ball or gauze
•
There may be a small amount of blood or liquid at the injection site. This is normal.
•
You can press a cotton ball or gauze over the injection site and hold for 10 seconds.
•
You may cover the injection site with a small adhesive bandage, if necessary.
Do not rub your skin.
2. Throw the syringe away (see figure 10)
•
Place your syringe in a sharps container straight away. Make sure you dispose of the bin as
instructed by your doctor or nurse.
Do not attempt to recap the needle.
Do not ever re-use a syringe, for your safety and health and for the safety of others.
If you feel that something has gone wrong with the injection or if you are not sure, talk to your doctor
or pharmacist.

Source: European Medicines Agency
 - Please bookmark this page (add it to your favorites).
- Please bookmark this page (add it to your favorites).
- If you wish to link to this page, you can do so by referring to the URL address below this line.
https://theodora.com/drugs/eu/simponi.html
Copyright © 1995-2021 ITA all rights reserved.
|
























































































































































































































































































































































































































































































































































 - Please bookmark this page (add it to your favorites).
- Please bookmark this page (add it to your favorites).










































