Product Characteristics
ANNEX I
SUMMARY OF PRODUCT CHARACTERISTICS
NAME OF THE MEDICINAL PRODUCT
Tamiflu 30 mg hard capsule.
QUALITATIVE AND QUANTITATIVE COMPOSITION
Each hard capsule contains oseltamivir phosphate equivalent to 30 mg of oseltamivir.
For a full list of excipients, see section 6.1.
Hard capsule
The hard capsule consists of a light yellow opaque body bearing the imprint “ROCHE” and a light
yellow opaque cap bearing the imprint “30 mg”. Imprints are blue.
4.1 Therapeutic indications
Treatment of influenza
In patients one year of age and older who present with symptoms typical of influenza, when influenza
virus is circulating in the community. Efficacy has been demonstrated when treatment is initiated
within two days of first onset of symptoms. This indication is based on clinical studies of naturally
occurring influenza in which the predominant infection was influenza A (see section 5.1).
Tamiflu is indicated for the treatment of infants below 12 months of age during a pandemic influenza
outbreak (see section 5.2).
Prevention of influenza
-
Post-exposure prevention in individuals one year of age or older following contact with a
clinically diagnosed influenza case when influenza virus is circulating in the community.
The appropriate use of Tamiflu for prevention of influenza should be determined on a case by
case basis by the circumstances and the population requiring protection. In exceptional
situations (e.g., in case of a mismatch between the circulating and vaccine virus strains, and a
pandemic situation) seasonal prevention could be considered in individuals one year of age or
older.
Tamiflu is indicated for post-exposure prevention of influenza in infants below 12 months of
age during a pandemic influenza outbreak (see section 5.2).
Tamiflu is not a substitute for influenza vaccination.
The use of antivirals for the treatment and prevention of influenza should be determined on the basis
of official recommendations. Decisions regarding the use of oseltamivir for treatment and prophylaxis
should take into consideration what is known about the characteristics of the circulating influenza
viruses, available information on influenza drug susceptibility patterns for each season and the impact
of the disease in different geographical areas and patient populations (see section 5.1).
Based on limited pharmacokinetic and safety data, Tamiflu can be used in infants below 12 months of
age for treatment during a pandemic influenza outbreak. The treating physician should take into
account the pathogenicity of the circulating strain and the underlying condition of the patient to ensure
there is a potential benefit to the child.
4.2 Posology and method of administration
Tamiflu capsules and Tamiflu suspension are bioequivalent formulations. 75 mg doses can be
administered as either
-
one 30 mg capsule plus one 45 mg capsule or
by administering one 30 mg dose plus one 45 mg dose of suspension.
Adults, adolescents or children ( 1 year of age or older) who are unable to swallow capsules may
receive appropriate doses of Tamiflu suspension.
For infants below 1 year of age
: In the absence of a suitable formulation, a pharmacy compounded
preparation should preferentially be used as the syringe provided in the Tamiflu 12 mg/ml powder for
oral suspension pack (with mg markings) does not allow for appropriate dose adjustments and
commercially available syringes (with ml markings) may lead to unacceptable dosing inaccuracies
(see below 4.2).
Treatment of influenza
Treatment should be initiated as soon as possible within the first two days of onset of symptoms of
influenza.
¾
For adolescents (13 to 17 years of age) and adults
:
The recommended oral dose is 75 mg
oseltamivir twice daily for 5 days.
¾
For infants older than 1 year of age and for children 2 to 12 years of age
: Tamiflu 30 mg and 45 mg
capsules and oral suspension are available.
The following weight-adjusted dosing regimens are recommended for children 1 year of
age and older:
Body Weight
Recommended dose for 5 days
Children who are able to swallow capsules may receive treatment with Tamiflu capsules (30 mg,
45 mg, 75 mg) twice daily for 5 days as an alternative to the recommended dose of Tamiflu
suspension.
¾
For infants below 12 months of age
: The recommended treatment dose for infants less than 12
months is between 2 mg/kg twice daily and 3 mg/kg twice daily during a pandemic influenza
outbreak. This is based upon limited pharmacokinetic data indicating that these doses provide plasma
drug exposures in the majority of patients similar to those shown to be clinically efficacious in older
children and adults (see section 5.2). The following weight-adjusted dosing regimens are
recommended for treatment of infants below 1 year of age:
Age Recommended dose for 5 days
> 3 months to 12 months 3 mg/kg twice daily
> 1 month to 3 months 2.5 mg/kg twice daily
0 to 1 month* 2 mg/kg twice daily
* There is no data available regarding the administration of Tamiflu to infants less than one month of
age.
Administration of Tamiflu to infants less than one year of age should be based upon the judgment of
the physician after considering the potential benefit of treatment versus any potential risk to the infant.


















These age-based dosing recommendations are not intended for premature infants, i.e. those with a
postmenstrual age less than 37 weeks. Insufficient data are available for these patients, in whom
different dosing may be required due to the immaturity of physiological functions
Prevention of influenza
Post-exposure prevention
¾
For adolescents (13 to 17 years of age) and adults
: The recommended dose for prevention of
influenza following close contact with an infected individual is 75 mg oseltamivir once daily for 10
days. Therapy should begin as soon as possible within two days of exposure to an infected individual.
¾
For infants older than 1 year of age and for children 2 to 12 years of age
: Tamiflu 30 mg and 45 mg
capsules and oral suspension are available.
The recommended post-exposure prevention dose of Tamiflu is:
Body Weight
Recommended dose for 10 days
Children who are able to swallow capsules may receive prevention Tamiflu capsules (30 mg, 45 mg,
75 mg) once daily for 10 days as an alternative to the recommended dose of Tamiflu suspension.
¾
For infants below 12 months of age
: The recommended prophylaxis dose for infants less than
12 months during a pandemic influenza outbreak is half of the daily treatment dose. This is based upon
clinical data in children > 1 year of age and adults showing that a prophylaxis dose equivalent to half
the daily treatment dose is clinically efficacious for the prevention of influenza. The following
weight-adjusted dosing prophylaxis regimens are recommended for infants below 1 year of age:
Age Recommended dose for 10 days
> 3 months to 12 months 3 mg/kg once daily
> 1 month to 3 months 2.5 mg/kg once daily
0 to 1 month* 2 mg/kg once daily
* There is no data available regarding the administration of Tamiflu to infants less than one month of
age.
Administration of Tamiflu to infants less than one year of age should be based upon the judgment of
the physician after considering the potential benefit of prophylaxis versus any potential risk to the
infant.
These age-based dosing recommendations are not intended for premature infants, i.e. those with a
postmenstrual age less than 37 weeks. Insufficient data are available for these patients, in whom
different dosing may be required due to the immaturity of physiological functions
Prevention during an influenza epidemic in the community
The recommended dose for prevention of influenza during a community outbreak is 75 mg oseltamivir
once daily for up to 6 weeks.
Extemporaneous formulation
When Tamiflu powder for oral suspension is not available
When commercially manufactured Tamiflu powder for oral suspension is not available, patients who
are unable to swallow capsules may receive appropriate doses of Tamiflu prepared in a pharmacy or
prepared at home.
For infants below 12 months, the pharmacy preparation should be preferred to home preparation.
Detailed information on the home preparation can be found in section 3 of the package leaflet of
Tamiflu capsules.




















Pharmacy compounding
¾
Adults and children greater than 1 year who are unable to swallow intact capsules
This procedure describes the preparation of a 15 mg/ml solution that will provide one patient with
enough medication for a 5-day course of treatment or a 10-day course of prophylaxis.
The pharmacist may compound a suspension (15 mg/ml) from Tamiflu 30 mg, 45 mg or 75 mg
capsules using water containing 0.1% w/v sodium benzoate added as a preservative.
First, calculate the Total Volume needed to be compounded and dispensed to provide a 5-day course
of treatment or a 10=day course of prophylaxis for the patient. The Total Volume required is
determined by the weight of the patient according to the recommendation in the table below:
Volume of Compounded Suspension (15 mg/ml) Prepared Based Upon th
e Patient’s Weight
Body Weight
(kg)
Total Volume to Compound per Patient Weight
(ml)
Second, determine the number of capsules and the amount of vehicle (water containing 0.1% w/v
sodium benzoate added as a preservative) that is needed to prepare the Total Volume (calculated from
the table above: 30 ml, 40 ml, 50 ml or 60 ml) of compounded suspension (15 mg/ml) as shown in the
table below:
Number of Capsules and Amount of Vehicle Needed to Prepare the Total Volume of a
Compounded Suspension (15 mg/ml)
Required Number of Tamiflu Capsules
(mg of oseltamivir)
Total Volume
of Compounded
Suspension
to be Prepared
Required Volume
of Vehicle
Please use
alternative capsule
strength*
Please use
alternative capsule
strength*
* No integral number of capsules can be used to achieve the target concentration; therefore, please use
either the 30 mg or 75 mg capsules.
Third, follow the procedure below for compounding the suspension (15 mg/ml) from Tamiflu
capsules:
1.
Carefully separate the capsule body and cap and transfer the contents of the required number of
Tamiflu capsules into a clean mortar.
Add one-third (1/3) of the specified amount of vehicle (water containing 0.1% w/v sodium
benzoate added as a preservative) and triturate the powder until a uniform suspension is
achieved.
Transfer the suspension to an amber glass or amber polyethyleneterephthalate (PET) bottle.
A funnel may be used to eliminate any spillage.
Add another one-third (1/3) of the vehicle to the mortar, rinse the pestle and mortar by a
triturating motion and transfer the vehicle into the bottle.
Triturate the granules to a fine powder.



















6. Repeat the rinsing (Step 5) with the remainder of the vehicle.
7. Close the bottle using a child-resistant cap.
8. Shake well to completely dissolve the active drug and to ensure homogeneous distribution of
the dissolved drug in the resulting suspension.
(Note: Undissolved residue may be visible but is comprised of inert ingredients of Tamiflu
capsules, which are insoluble. However, the active drug, oseltamivir phosphate, readily
dissolves in the specified vehicle and therefore forms a uniform solution.)
9. Put an ancillary label on the bottle indicating
“
Shake Gently Before Use
”
.
10. Instruct the parent or caregiver that after the patient has completed the full course of therapy any
remaining solution must be discarded. It is recommended that this information be provided by
affixing an ancillary label to the bottle or adding a statement to the pharmacy label instructions.
11. Place an appropriate expiration date label according to storage condition (see below).
Storage of the pharmacy-compounded suspension (15 mg/ml)
Room temperature storage conditions: Stable for 3 weeks (21 days) when stored at room temperature
“do not store above 25 °C”.
Refrigerated storage conditions: Stable for 6 weeks when stored at 2 °C - 8 °C.
Place a pharmacy label on the bottle that includes the patient’s name, dosing instructions, use by date,
drug name and any other required information to be in compliance with local pharmacy regulations.
Refer to the table below for the proper dosing instructions.
Prophylaxis Dose
(for 10 days)
10 kg to 15 kg 30 mg 2 ml 2 ml twice daily 2 ml once daily
> 15 to 23 kg 45 mg 3 ml 3 ml twice daily 3 ml once daily
> 23 to 40 kg 60 mg 4 ml 4 ml twice daily 4 ml once daily
> 40 kg 75 mg 5 ml 5 ml twice daily 5 ml once daily
Note: This compounding procedure results in a 15 mg/ml suspension, which is different from the
commercially available Tamiflu powder for oral suspension.
Treatment Dose
(for 5 days)
Dispense the suspension with a graduated oral syringe for measuring small amounts of suspension. If
possible, mark or highlight the graduation corresponding to the appropriate dose (2 ml, 3 ml, 4 ml or
5 ml) on the oral syringe for each patient.
The appropriate dose must be mixed by the caregiver with an equal quantity of sweet liquid food, such
as sugar water, chocolate syrup, cherry syrup, dessert toppings (like caramel or fudge sauce) to mask
the bitter taste.
¾
Infants less than 1 year of age
This procedure describes the preparation of a 10 mg/ml solution that will provide one patient with
enough medication for a 5-day course of treatment or a 10-day course of prophylaxis.
The pharmacist may compound a suspension (10 mg/ml) from Tamiflu 30 mg, 45 mg or 75 mg
capsules using water containing 0.1% w/v sodium benzoate added as a preservative.
First, calculate the Total Volume needed to be compounded and dispensed for each patient. The Total
Volume required is determined by the weight of the patient according to the recommendation in the
table below:
Volume of Compounded Suspension (10 mg/ml) Prepared Based Upon th
e Patient’s Weight
Body Weight
(kg)
Total Volume to Compound per Patient Weight
(ml)
Dosing Chart for Pharmacy-Compounded Suspension from Tamiflu Capsules for Children One
Year of Age or Older
Body Weight
(kg)













Second, determine the number of capsules and the amount of vehicle (water containing 0.1% w/v
sodium benzoate added as a preservative) that is needed to prepare the Total Volume (calculated from
the table above: 30 ml, 45 ml) of compounded suspension (10 mg/ml) as shown in the table below:
Number of Capsules and Amount of Vehicle Needed to Prepare the Total Volume of a
Compounded Suspension (10 mg/ml)
Required Number of Tamiflu Capsules
(mg of oseltamivir)
Required Volume
of Vehicle
Please use
alternative capsule
strength*
* No integral number of capsules can be used to achieve the target concentration; therefore, please use
either the 30 mg or 75 mg capsules.
Third, follow the procedure below for compounding the suspension (10 mg/ml) from Tamiflu
capsules:
1. Carefully separate the capsule body and cap and transfer the contents of the required number of
Tamiflu capsules into a clean mortar.
2. Triturate the granules to a fine powder.
3. Add one-third (1/3) of the specified amount of vehicle and triturate the powder until a uniform
suspension is achieved.
4. Transfer the suspension to an amber glass or amber polyethyleneterephthalate (PET) bottle.
A funnel may be used to eliminate any spillage.
5. Add another one-third (1/3) of the vehicle to the mortar, rinse the pestle and mortar by a
triturating motion and transfer the vehicle into the bottle.
6. Repeat the rinsing (Step 5) with the remainder of the vehicle.
7. Close the bottle using a child-resistant cap.
8. Shake well to completely dissolve the active drug and to ensure homogeneous distribution of
the dissolved drug in the resulting suspension.
(Note: Undissolved residue may be visible but is comprised of inert ingredients of Tamiflu
capsules, which are insoluble. However, the active drug, oseltamivir phosphate, readily
dissolves in the specified vehicle and therefore forms a uniform solution.)
9. Put an ancillary label on the bottle indicating “Shake Gently Before Use”.
10. Instruct the parent or caregiver that after the patient has completed the full course of therapy any
remaining solution must be discarded. It is recommended that this information be provided by
affixing an ancillary label to the bottle or adding a statement to the pharmacy label instructions.
11. Place an appropriate expiration date label according to storage condition (see below).
Storage of the pharmacy-compounded suspension (10 mg/ml)
Room temperature storage conditions: Stable for 3 weeks (21 days) when stored at room temperature
“do not store above 25 °C”.
Refrigerated storage conditions: Stable for 6 weeks when stored at 2 °C - 8 °C.
Place a pharmacy label on the bottle that includes the patient’s name, dosing instructions, use by date,
drug name and any other required information to be in compliance with local pharmacy regulations.
Refer to the table below for the proper dosing instructions.
Total Volume
of Compounded
Suspension
to be Prepared










Dosing Chart for Pharmacy-Compounded Suspension (10 mg/ml) from Tamiflu Capsules for
Infants Less Than One Month of Age
Body Weight
(rounded to the
nearest 0.5 kg)
Treatment Dose
(for 5 days)
Prophylaxis Dose
(for 10 days)
Dosing Chart for Pharmacy-Compounded Suspension (10 mg/ml) from Tamiflu Capsules for
Infants One to Twelve Months of Age
Body Weight
(rounded to the
nearest 0.5 kg)
Treatment Dose
(for 5 days)
Prophylaxis Dose
(for 10 days)
Note: This compounding procedure results in a 10 mg/ml suspension, which is different from the
commercially available Tamiflu powder for oral suspension.
Dispense the suspension with a graduated oral syringe for measuring small amounts of suspension. If
possible, mark or highlight the graduation corresponding to the appropriate dose on the oral syringe
for each patient.
The appropriate dose must be mixed by the caregiver with an equal quantity of sweet liquid food, such
as sugar water, chocolate syrup, cherry syrup, dessert toppings (like caramel or fudge sauce) to mask
the bitter taste.
Home preparation
When commercially manufactured Tamiflu oral suspension is not available, a pharmacy preparation
from Tamiflu capsules can be used (detailed instructions above in section 4.2). If the pharmacy
preparation is not available either, Tamiflu doses may be prepared at home. The pharmacy preparation
is the preferred option in infants below 12 months of age.
When appropriate capsule strengths are available, the dose is given by opening the capsule and mixing
its contents with no more than one teaspoon of a suitable sweetened food product. The bitter taste can
be masked by products such as sugar water, chocolate syrup, cherry syrup, dessert toppings (like
caramel or fudge sauce). The mixture should be stirred and given entirely to the patient. The mixture
must be swallowed immediately after its preparation.
When only 75 mg capsules are available, and doses of 30 mg or 45 mg are needed, the preparation
involves additional steps. Detailed instructions can be found in section 3 in the package leaflet of
Tamiflu capsules.
Special populations
Hepatic impairment
No dose adjustment is required either for treatment or for prevention in patients with hepatic
dysfunction. No studies have been carried out in paediatric patients with hepatic disorder.




















Renal impairment
Treatment of influenza
: Dose adjustment is recommended for adults with severe renal impairment.
Recommended doses are detailed in the table below.
Creatinine clearance
Recommended dose for treatment
75 mg once daily,
or 30 mg suspension twice daily,
or 30 mg capsules twice daily
Prevention of influenza
: Dose adjustment is recommended for adults with severe renal impairment as
detailed in the table below.
Creatinine clearance
Recommended dose for prevention
75 mg every second day,
or 30 mg suspension once daily,
or 30 mg capsules once daily
Elderly
No dose adjustment is required, unless there is evidence of severe renal impairment.
Children
There is insufficient clinical data available in children with renal impairment to be able to make any
dosing recommendation.
Immunocompromised patients
Longer duration of seasonal prophylaxis up to 12 weeks has been evaluated in immunocompromised
subjects (see sections 4.4, 4.8 and 5.1).
Hypersensitivity to the active substance or to any of the excipients.
4.4 Special warnings and precautions for use
Oseltamivir is effective only against illness caused by influenza viruses. There is no evidence for
efficacy of oseltamivir in any illness caused by agents other than influenza viruses.
No information is available regarding the safety and efficacy of oseltamivir in patients with any
medical condition sufficiently severe or unstable to be considered at imminent risk of requiring
hospitalisation.
The efficacy of oseltamivir in either treatment or prophylaxis of influenza in immunocompromised
patients has not been firmly established (see section 5.1).
Efficacy of oseltamivir in the treatment of subjects with chronic cardiac disease and/or respiratory
disease has not been established. No difference in the incidence of complications was observed
between the treatment and placebo groups in this population (see section 5.1).
No data allowing a dose recommendation for premature children (< 37 weeks post-menstrual age*) are
currently available.














* Time between first day of last normal menstrual period and day of assessment, gestational age plus
post-natal age.
Tamiflu is not a substitute for influenza vaccination. Use of Tamiflu must not affect the evaluation of
individuals for annual influenza vaccination. The protection against influenza lasts only as long as
Tamiflu is administered. Tamiflu should be used for the treatment and prevention of influenza only
when reliable epidemiological data indicate that influenza virus is circulating in the community.
Susceptibility of circulating influenza virus strains to oseltamivir has been shown to be highly variable
(see section 5.1). Therefore, prescribers should take into account the most recent information available
on oseltamivir susceptibility patterns of the currently circulating viruses when deciding whether to use
Tamiflu.
Severe renal impairment
Dose adjustment is recommended for both treatment and prevention in adults with severe renal
insufficiency. There is insufficient clinical data available in children with renal impairment to be able
to make any dosing recommendation.(see sections 4.2 and 5.2).
Neuropsychiatric events have been reported during administration of Tamiflu in patients with
influenza, especially in children and adolescents. These events are also experienced by patients with
influenza without oseltamivir administration. Patients should be closely monitored for behavioural
changes, and the benefits and risks of continuing treatment should be carefully evaluated for each
patient (see section 4.8).
4.5 Interaction with other medicinal products and other forms of interaction
Pharmacokinetic properties of oseltamivir, such as low protein binding and metabolism independent of
the CYP450 and glucuronidase systems (see section 5.2), suggest that clinically significant drug
interactions via these mechanisms are unlikely.
No dose adjustment is required when co-administering with probenecid in patients with normal renal
function. Co-administration of probenecid, a potent inhibitor of the anionic pathway of renal tubular
secretion, results in an approximate 2-fold increase in exposure to the active metabolite of oseltamivir.
Oseltamivir has no kinetic interaction with amoxicillin, which is eliminated via the same pathway,
suggesting that oseltamivir interaction with this pathway is weak.
Clinically important drug interactions involving competition for renal tubular secretion are unlikely,
due to the known safety margin for most of these substances, the elimination characteristics of the
active metabolite (glomerular filtration and anionic tubular secretion) and the excretion capacity of
these pathways. However, care should be taken when prescribing oseltamivir in subjects when taking
co-excreted agents with a narrow therapeutic margin (e.g., chlorpropamide, methotrexate,
phenylbutazone).
No pharmacokinetic interactions between oseltamivir or its major metabolite have been observed
when co-administering oseltamivir with paracetamol, acetyl-salicylic acid, cimetidine or with antacids
(magnesium and aluminium hydroxides and calcium carbonates).
4.6 Pregnancy and lactation
While no controlled clinical trials have been conducted on the use of oseltamivir in pregnant women,
there is limited data available from post-marketing and retrospective observational surveillance
reports. These data in conjunction with animal studies do not indicate direct or indirect harmful effects
with respect to pregnancy, embryonal/foetal or postnatal development (see section 5.3). Pregnant
women may receive Tamiflu, after considering the available safety information, the pathogenicity of
the circulating influenza virus strain and the underlying condition of the pregnant woman.
In lactating rats, oseltamivir and the active metabolite are excreted in milk. Very limited information is
available on children breast-fed by mothers taking oseltamivir and on excretion of oseltamivir in
breast milk. Limited data demonstrated that oseltamivir and the active metabolite were detected in
breast milk, however the levels were low, which would result in a subtherapeutic dose to the infant.
Considering this information, the pathogenicity of the circulating influenza virus strain and the
underlying condition of the lactating woman, administration of oseltamivir may be considered, where
there are clear potential benefits to lactating mothers.
4.7 Effects on ability to drive and use machines
Tamiflu has no influence on the ability to drive and use machines.
The overall safety profile of Tamiflu is based on data from 2107 adult and 1032 paediatric patients
treated with Tamiflu or placebo for influenza, and on data from 2914 adult and 148 paediatric patients
receiving Tamiflu or placebo for the prophylaxis of influenza in clinical trials. In addition, 475
immunocompromised patients (including 18 children) received Tamiflu or placebo for the prophylaxis
of influenza.
In adults, the most commonly reported adverse drug reactions (ADRs) were vomiting and nausea in
the treatment studies, and nausea and headache in the prevention studies. The majority of these ADRs
were reported on a single occasion on either the first or second treatment day and resolved
spontaneously within 1-2 days. In children, the most commonly reported adverse drug reaction was
vomiting.
The ADRs listed in the tables below fall into the following categories: Very Common (≥
1/10
),
Common (≥
1/100
to <
1/10
), Uncommon (≥
1/1,000
to <
1/100
), Rare (≥
1/10,000
to <
1/1,000
), Very
rare (<
1/10,000
) and not known (cannot be estimated from the available data). ADRs are added to the
appropriate category in the tables according to the pooled analysis from clinical trials. Within each
frequency grouping ADRs are presented in the order of decreasing seriousness.
Treatment and prevention of influenza in adults and adolescents
:
Most Frequent Adverse Drug Reactions (
≥
1 % in the oseltamivir group) in Studies Investigating
Tamiflu for Treatment and Prevention of Influenza in Adults and Adolescents or Through Post-
Marketing Surveillance
System Organ Class (SOC)
Frequency Category
Adverse Drug Reaction
Percentage of Patients Experiencing the ADR
Treatment
Oseltamivir
75 mg bid
(n = 1057)
Oseltamivir
75 mg od
(n = 1480)
Infections and infestations
Upper respiratory tract infections
Disorders of the ear and labyrinth











System Organ Class (SOC)
Frequency Category
Adverse Drug Reaction
Percentage of Patients Experiencing the ADR
Treatment
Oseltamivir
75 mg bid
(n = 1057)
Oseltamivir
75 mg od
(n = 1480)
Respiratory, thoracic and
mediastinal disorders
Gastrointestinal disorders
Skin and subcutaneous tissue
disorders
Uncommon
:
Dermatitis
a
< 1 % < 1 % 1 % 1 %
Rash
a
< 1 % < 1 % < 1 % < 1 %
Urticaria
a
< 1 % < 1 % < 1 % < 1 %
Eczema
a
< 1 % 0 % < 1 % < 1 %
General disorders
Common
:
Dizziness 2 % 3 % 2 % 2 %
Fatigue 1 % 1 % 8 % 8 %
Pain < 1 % < 1 % 4 % 3 %
a
These are events identified during post-marketing surveillance. They were also reported in the
pooled clinical studies at the incidence presented in the table above.
b
Subjects who experienced nausea alone; excludes subjects who experienced nausea in association
with vomiting.
c
The difference between the placebo and oseltamivir groups was statistically significant.
Treatment and prevention of influenza in children
:
The table below shows the most frequently reported ADRs from paediatric clinical trials.
Most Frequent Adverse Drug Reactions (
≥
1 % in the oseltamivir group in the treatment studies
and
≥
10 % in the oseltamivir group in the prophylaxis study) in Children
Percentage of Patients Experiencing the ADR
Treatment
Oseltamivir
2 mg/kg bid
(n = 515)
Oseltamivir
30 to 75 mg
b
(n = 158)
Oseltamivir
30 to 75 mg
b
(n = 148)
Infections and infestations
System Organ Class (SOC)
Frequency Category
Adverse Drug Reaction

















System Organ Class (SOC)
Frequency Category
Adverse Drug Reaction
Percentage of Patients Experiencing the ADR
Treatment
Oseltamivir
2 mg/kg bid
(n = 515)
Oseltamivir
30 to 75 mg
b
(n = 158)
Oseltamivir
30 to 75 mg
b
(n = 148)
Disorders of the blood and
lymphatic system
Respiratory, thoracic and
mediastinal disorders
Asthma (incl. aggravated)
Gastrointestinal disorder
Disorders of the ear and labyrinth
Tympanic membrane disorder
Skin and subcutaneous tissue
disorders
a
The prevention study did not contain a placebo arm, i.e. was an uncontrolled study.
b
Unit dose = weight-based dosing (see section 4.2).
c
Patients experienced ear ache and ear pain.
In general, the adverse event profile in children with pre-existing bronchial asthma was qualitatively
similar to that of otherwise healthy children.
Further post marketing surveillance data on selected serious adverse drug reactions
:
Immune system disorders
Frequency not known: hypersensitivity reactions, including anaphylactic/anaphylactoid reactions.
Psychiatric disorders and nervous system disorders
Frequency not known: influenza can be associated with a variety of neurologic and behavioural
symptoms which can include events such as hallucinations, delirium, and abnormal behaviour, in some
cases resulting in fatal outcomes. These events may occur in the setting of encephalitis or
encephalopathy but can occur without obvious severe disease.
In patients with influenza who were receiving Tamiflu, there have been postmarketing reports of
convulsions and delirium (including symptoms such as altered level of consciousness, confusion,
abnormal behaviour, delusions, hallucinations, agitation, anxiety, nightmares), in a very few cases
resulting in self-injury or fatal outcomes. These events were reported primarily among pediatric and
adolescent patients and often had an abrupt onset and rapid resolution. The contribution of Tamiflu to














those events is unknown. Such neuropsychiatric events have also been reported in patients with
influenza who were not taking Tamiflu.
Eye disorders
Frequency not known: visual disturbance.
Cardiac disorders
Frequency not known: cardiac arrhythmia.
Gastrointestinal disorders
Frequency not known
:
gastrointestinal bleedings and hemorrhagic colitis.
Hepato-biliary disorders
Frequency not known: hepato-biliary system disorders, including hepatitis and elevated liver enzymes
in patients with influenza-like illness. These cases include fatal fulminant hepatitis/hepatic failure.
Skin and subcutaneous tissue disorders
Frequency not known: severe skin reactions, including Stevens-Johnson syndrome, toxic epidermal
necrolysis, erythema multiforme and angioneurotic oedema.
Additional information on special populations
:
Infants less than one year of age
Safety information available on oseltamivir administered for treatment of influenza in infants less than
one year of age from prospective and retrospective observational trials (comprising together more than
2400 infants of that age class), epidemiological databases research and postmarketing reports suggest
that the safety profile in infants less than one year of age is similar to the established safety profile of
children aged one year and older.
Elderly patients
There were no clinically relevant differences in the safety population of the elderly subjects who
received oseltamivir or placebo compared with the adult population aged up to 65 years.
Patients with chronic cardiac and/or respiratory disease
The adverse event profile in adolescents and patients with chronic cardiac and/or respiratory disease
was qualitatively similar to those of healthy young adults.
Immunocompromised patients
The adverse reactions noted in immunocompromised subjects 13 years of age and older who received
oseltamivir during 12 weeks for the seasonal prophylaxis of influenza were consistent with those
previously observed in Tamiflu clinical trials.
There is no experience with overdose. However, the anticipated manifestations of acute overdose
would be nausea, with or without accompanying vomiting, and dizziness. Patients should discontinue
the treatment in the event of overdose. No specific antidote is known.
PHARMACOLOGICAL PROPERTIES
5.1 Pharmacodynamic properties
Pharmacotherapeutic group: Antiviral ATC code: J05AH02
Oseltamivir phosphate is a pro-drug of the active metabolite (oseltamivir carboxylate). The active
metabolite is a selective inhibitor of influenza virus neuraminidase enzymes, which are glycoproteins
found on the virion surface. Viral neuraminidase enzyme activity is important both for viral entry into
uninfected cells and for the release of recently formed virus particles from infected cells, and for the
further spread of infectious virus in the body.
Oseltamivir carboxylate inhibits influenza A and B neuraminidases
in vitro
. Oseltamivir phosphate
inhibits influenza virus infection and replication
in vitro
. Oseltamivir given orally inhibits influenza A
and B virus replication and pathogenicity
in vivo
in animal models of influenza infection at antiviral
exposures similar to that achieved in man with 75 mg twice daily
.
Antiviral activity of oseltamivir was supported for influenza A and B by experimental challenge
studies in healthy volunteers.
Neuraminidase enzyme IC50 values for oseltamivir for clinically isolated influenza A ranged from
0.1 nM to 1.3 nM, and for influenza B was 2.6 nM. Higher IC50 values for influenza B, up to a
median of 8.5 nM, have been observed in published trials.
Reduced sensitivity of viral neuraminidase
Clinical studies
: The risk of emergence of influenza viruses with reduced susceptibility or frank
resistance to oseltamivir has been examined during Roche-sponsored clinical studies. All patients who
were found to carry oseltamivir-resistant virus did so transiently, cleared the virus normally and
showed no clinical deterioration.
Patients with Resistance Mutations (%)
* Full genotyping was not performed in all studies.
There has been no evidence for emergence of drug resistance associated with the use of Tamiflu in
clinical studies conducted to date in post-exposure (7 days), post-exposure within household groups
(10 days) and seasonal (42 days) prevention of influenza in immunocompetent subjects. There was no
resistance observed during a 12-week prophylaxis study in immunocompromised subjects.
Clinical and surveillance data
: Natural mutations associated with reduced susceptibility to oseltamivir
in vitro
have been detected in influenza A and B viruses isolated from patients without exposure to
oseltamivir. Resistant strains selected during oseltamivir treatment have been isolated from both
immunocompetent and immunocompromised patients. Immunocompromised patients and young
children are at a higher risk of developing oseltamivir-resistant virus during treatment.
Oseltamivir-resistant viruses isolated from oseltamivir-treated patients and oseltamivir-resistant
laboratory strains of influenza viruses have been found to contain mutations in N1 and N2
neuraminidases. Resistance mutations tend to be viral sub-type specific. Since 2007 resistance
associated H275Y mutation in seasonal H1N1 strains has become widespread. The susceptibility to
oseltamivir and the prevalence of such viruses appear to vary seasonally and geographically. In 2008,
H275Y was found in > 99 % of circulating H1N1 influenza isolates in Europe. The 2009 H1N1
influenza (“swine flu”) was almost uniformly susceptible to oseltamivir, with only sporadic reports of
resistance in connection with both therapeutic and prophylactic regimens.
Treatment of influenza infection
Oseltamivir is effective only against illnesses caused by influenza virus. Statistical analyses are
therefore presented only for influenza-infected subjects. In the pooled treatment study population,
which included both influenza-positive and -negative subjects (ITT), primary efficacy was reduced
proportional to the number of influenza-negative individuals. In the overall treatment population,
influenza infection was confirmed in 67 % (range 46 % to 74 %) of the recruited patients. Of the
elderly subjects, 64 % were influenza-positive and of those with chronic cardiac and/or respiratory
disease 62 % were influenza-positive. In all phase III treatment studies, patients were recruited only
during the period in which influenza was circulating in the local community.








Adults and adolescents 13 years of age and older
: Patients were eligible if they reported within
36 hours of onset of symptoms, had fever ≥ 37.8 °C, accompanied by at least one respiratory symptom
(cough, nasal symptoms or sore throat) and at least one systemic symptom (myalgia, chills/sweats,
malaise, fatigue or headache). In a pooled analysis of all influenza-positive adults and adolescents
(N = 2413) enrolled into treatment studies, oseltamivir 75 mg twice daily for 5 days reduced the
median duration of influenza illness by approximately one day from 5.2 days (95 % CI 4.9 – 5.5 days)
in the placebo group to 4.2 days (95 % CI 4.0 – 4.4 days; p
≤
0.0001).
The proportion of subjects who developed specified lower respiratory tract complications (mainly
bronchitis) treated with antibiotics was reduced from 12.7 % (135/1063) in the placebo group to 8.6 %
(116/1350) in the oseltamivir treated population (p = 0.0012).
Treatment of influenza in high risk populations
: The median duration of influenza illness in elderly
subjects (≥ 65 years) and in subjects with chronic cardiac and/or respiratory disease receiving
oseltamivir 75 mg twice daily for 5 days was
not
reduced significantly. The total duration of fever was
reduced by one day in the groups treated with oseltamivir. In the influenza-positive elderly,
oseltamivir significantly reduced the incidence of specified lower respiratory tract complications
(mainly bronchitis) treated with antibiotics from 19 % (52/268) in the placebo group to 12 % (29/250)
in the oseltamivir treated population (p = 0.0156).
In influenza-positive patients with chronic cardiac and/or respiratory disease, the combined incidence
of lower respiratory tract complications (mainly bronchitis) treated with antibiotics was 17 % (22/133)
in the placebo group and 14 % (16/118) in the oseltamivir treated population (p = 0.5976).
Treatment of influenza in children
: In a study of otherwise healthy children (65 % influenza-positive)
aged 1 to 12 years (mean age 5.3 years) who had fever (≥ 37.8 °C) plus either cough or coryza, 67 %
of
influenza-positive patients were infected with influenza A and 33 % with influenza B. Oseltamivir
treatment, started within 48 hours of onset of symptoms, significantly reduced the time to freedom
from illness (defined as the simultaneous return to normal health and activity and alleviation of fever,
cough and coryza) by 1.5 days (95 % CI 0.6 – 2.2 days; p < 0.0001) compared to placebo. Oseltamivir
reduced the incidence of acute otitis media from 26.5 % (53/200) in the placebo group to 16 %
(29/183) in the oseltamivir treated children (p = 0.013).
A second study was completed in 334 asthmatic children aged 6 to 12 years old of which 53.6 % were
influenza-positive. In the oseltamivir treated group, the median duration of illness was
not
reduced
significantly. By day 6 (the last day of treatment) FEV
1
had increased by 10.8 % in the oseltamivir
treated group compared to 4.7 % on placebo (p = 0.0148) in this population.
The European Medicines Agency has deferred the obligation to submit the results of studies with
Tamiflu in one or more subsets of the paediatric population in influenza. See section 4.2 for
information on paediatric use.
Treatment of influenza B infection
:
Overall, 15 % of the influenza-positive population were infected
by influenza B, proportions ranging from 1 to 33 % in individual studies. The median duration of
illness in influenza B infected subjects did not differ significantly between the treatment groups in
individual studies. Data from 504 influenza B infected subjects were pooled across all studies for
analysis. Oseltamivir reduced the time to alleviation of all symptoms by 0.7 days (95 % CI 0.1 – 1.6
days; p = 0.022) and the duration of fever (≥ 37.8 °C), cough and coryza by one day (95 % CI 0.4 –
1.7 days; p < 0.001) compared to placebo.
Prevention of influenza
The efficacy of oseltamivir in preventing naturally occurring influenza illness has been demonstrated
in a post-exposure prevention study in households and two seasonal prevention studies. The primary
efficacy parameter for all of these studies was the incidence of laboratory-confirmed influenza. The
virulence of influenza epidemics is not predictable and varies within a region and from season to
season, therefore the number needed to treat (NNT) in order to prevent one case of influenza illness
varies.
Post-exposure prevention
: In a study in contacts (12.6 % vaccinated against influenza) of an index
case of influenza, oseltamivir 75 mg once daily was started within 2 days of onset of symptoms in the
index case and continued for seven days. Influenza was confirmed in 163 out of 377 index cases.
Oseltamivir significantly reduced the incidence of clinical influenza illness occurring in the contacts of
confirmed influenza cases from 24/200 (12 %) in the placebo group to 2/205 (1 %) in the oseltamivir
group (92 % reduction [95 % CI 6 – 16; p
≤
0.0001]). The number needed to treat (NNT) in contacts
of true influenza cases was 10 (95 % CI 9 – 12) and was 16 (95 % CI 15 – 19) in the whole population
(ITT) regardless of infection status in the index case.
The efficacy of oseltamivir in preventing naturally occurring influenza illness has been demonstrated
in a post-exposure prevention study in households that included adults, adolescents, and children aged
1 to 12 years, both as index cases and as family contacts. The primary efficacy parameter for this study
was the incidence of laboratory-confirmed clinical influenza in the households. Oseltamivir
prophylaxis lasted for 10 days. In the total population, there was a reduction in the incidence of
laboratory-confirmed clinical influenza in households from 20 % (27/136) in the group not receiving
prevention to 7 % (10/135) in the group receiving prevention (62.7 % reduction [95 % CI 26.0 – 81.2;
p = 0.0042]). In households of influenza-infected index cases, there was a reduction in the incidence of
influenza from 26 % (23/89) in the group not receiving prevention to 11 % (9/84) in the group
receiving prevention (58.5 % reduction [95 % CI 15.6 – 79.6; p = 0.0114]).
According to subgroup analysis in children at 1 to 12 years of age, the incidence of laboratory-
confirmed clinical influenza among children was significantly reduced from 19 % (21/111) in the
group not receiving prevention to 7 % (7/104) in the group receiving prevention (64.4 % reduction
[95 % CI 15.8 – 85.0; p = 0.0188]). Among children who were not already shedding virus at baseline,
the incidence of laboratory-confirmed clinical influenza was reduced from 21 % (15/70) in the group
not receiving prevention to 4 % (2/47) in the group receiving prevention (80.1 % reduction [95 % CI
22.0 – 94.9; p = 0.0206]). The NNT for the total paediatric population was 9 (95 % CI 7 – 24) and
8 (95 % CI 6, upper limit not estimable) in the whole population (ITT) and in paediatric contacts of
infected index cases (ITTII), respectively.
Prevention during an influenza epidemic in the community
: In a pooled analysis of two other studies
conducted in unvaccinated otherwise healthy adults, oseltamivir 75 mg once daily given for 6 weeks
significantly reduced the incidence of clinical influenza illness from 25/519 (4.8 %) in the placebo
group to 6/520 (1.2 %) in the oseltamivir group (76 % reduction [95 % CI 1.6 – 5.7; p = 0.0006])
during a community outbreak of influenza. The NNT in this study was 28 (95 % CI 24 – 50).
A study in elderly residents of nursing homes, where 80 % of participants received vaccine in the
season of the study, oseltamivir 75 mg once daily given for 6 weeks significantly reduced the
incidence of clinical influenza illness from 12/272 (4.4 %) in the placebo group to 1/276 (0.4 %) in the
oseltamivir group (92 % reduction [95 % CI 1.5 – 6.6; p = 0.0015]). The NNT in this study was
25 (95 % CI 23 – 62).
Prophylaxis of influenza in immunocompromised patients
:
A double-blind, placebo-controlled,
randomised study was conducted for seasonal prophylaxis of influenza in 475 immunocompromised
subjects (388 subjects with solid organ transplantation [195 placebo; 193 oseltamivir], 87 subjects
with hemopoetic stem cell transplantation [43 placebo; 44 oseltamivir], no subject with other
immunosuppressant conditions), including 18 children 1 to 12 years of age. The primary endpoint in
this study was the incidence of laboratory-confirmed clinical influenza as determined by viral culture
and/or a four-fold rise in HAI antibodies. The incidence of laboratory-confirmed clinical influenza was
2.9 % (7/238) in the placebo group and 2.1 % (5/237) in the oseltamivir group (95 % CI -2.3 % –
4.1 %; p = 0.772).
Specific studies have not been conducted to assess of the reduction in the risk of complications.
5.2 Pharmacokinetic properties
Absorption
Oseltamivir is readily absorbed from the gastrointestinal tract after oral administration of oseltamivir
phosphate (pro-drug) and is extensively converted by predominantly hepatic esterases to the active
metabolite (oseltamivir carboxylate). At least 75 % of an oral dose reaches the systemic circulation as
the active metabolite. Exposure to the pro-drug is less than 5 % relative to the active metabolite.
Plasma concentrations of both pro-drug and active metabolite are proportional to dose and are
unaffected by co-administration with food.
Distribution
The mean volume of distribution at steady state of the oseltamivir carboxylate is approximately
23 litres in humans, a volume equivalent to extracellular body fluid. Since neuraminidase activity is
extracellular, oseltamivir carboxylate distributes to all sites of influenza virus spread.
The binding of the oseltamivir carboxylate to human plasma protein is negligible (approximately 3 %).
Metabolism
Oseltamivir is extensively converted to oseltamivir carboxylate by esterases located predominantly in
the liver.
In vitro
studies demonstrated that neither oseltamivir nor the active metabolite is a substrate
for, or an inhibitor of, the major cytochrome P450 isoforms. No phase 2 conjugates of either
compound have been identified
in vivo
.
Elimination
Absorbed oseltamivir is primarily (> 90 %) eliminated by conversion to oseltamivir carboxylate. It is
not further metabolised and is eliminated in the urine. Peak plasma concentrations of oseltamivir
carboxylate decline with a half-life of 6 to 10 hours in most subjects. The active metabolite is
eliminated entirely by renal excretion. Renal clearance (18.8 l/h) exceeds glomerular filtration rate
(7.5 l/h) indicating that tubular secretion occurs in addition to glomerular filtration. Less than 20 % of
an oral radiolabelled dose is eliminated in faeces.
Renal impairment
Administration of 100 mg oseltamivir phosphate twice daily for 5 days to patients with various
degrees of renal impairment showed that exposure to oseltamivir carboxylate is inversely proportional
to declining renal function. For dosing, see section 4.2.
Hepatic impairment
In vitro
studies have concluded that exposure to oseltamivir is not expected to be increased
significantly nor is exposure to the active metabolite expected to be significantly decreased in patients
with hepatic impairment (see section 4.2).
Elderly
Exposure to the active metabolite at steady state was 25 to 35 % higher in elderly (age 65 to 78 years)
compared to adults less than 65 years of age given comparable doses of oseltamivir. Half-lives
observed in the elderly were similar to those seen in young adults. On the basis of drug exposure and
tolerability, dosage adjustments are not required for elderly patients unless there is evidence of severe
renal impairment (creatinine clearance below 30 ml/min) (see section 4.2).
Children
Children 1 year of age and older
: The pharmacokinetics of oseltamivir have been evaluated in single-
dose pharmacokinetic studies in children aged 1 to 16 years. Multiple-dose pharmacokinetics were
studied in a small number of children enrolled in a clinical efficacy study. Younger children cleared
both the pro-drug and its active metabolite faster than adults, resulting in a lower exposure for a
given mg/kg dose. Doses of 2 mg/kg give oseltamivir carboxylate exposures comparable to those
achieved in adults receiving a single 75 mg dose (approximately 1 mg/kg). The pharmacokinetics of
oseltamivir in children over 12 years of age are similar to those in adults.
Infants below 12 months of age:
Limited pharmacokinetic and safety data are available for infants less
than 2 years of age. Pharmacokinetic modeling was undertaken using these data in addition to data
from studies in adults and children older than 1 year of age. The results demonstrate that doses of
3 mg/kg twice daily for infants aged 3 to 12 months and 2.5 mg/kg twice daily for infants aged
between 1 and 3 months provide exposures similar to those shown to be clinically efficacious in adults
and children > 1 year of age (see sections 4.1 and 4.2). There are currently no data available in infants
less than 1 month of age using Tamiflu.
5.3 Preclinical safety data
Preclinical data reveal no special hazard for humans based on conventional studies of safety
pharmacology, repeated-dose toxicity and genotoxicity. Results of the conventional rodent
carcinogenicity studies showed a trend towards a dose-dependent increase in the incidence of some
tumours that are typical for the rodent strains used. Considering the margins of exposure in relation to
the expected exposure in the human use, these findings do not change the benefit-risk of Tamiflu in its
adopted therapeutic indications.
Teratology studies have been conducted in rats and rabbits at doses of up to 1500 mg/kg/day and
500 mg/kg/day, respectively. No effects on foetal development were observed. A rat fertility study up
to a dose of 1500 mg/kg/day demonstrated no adverse effects on either sex. In pre- and post-natal rat
studies, prolonged parturition was noted at 1500 mg/kg/day: the safety margin between human
exposure and the highest no-effect dose (500 mg/kg/day) in rats is 480-fold for oseltamivir and 44-fold
for the active metabolite, respectively. Foetal exposure in the rats and rabbits was approximately
15 to 20 % of that of the mother.
In lactating rats, oseltamivir and the active metabolite are excreted in the milk. Limited data indicate
that oseltamivir and the active metabolite are excreted in human milk. Extrapolation of the animal data
provides estimates of 0.01 mg/day and 0.3 mg/day for the respective compounds.
A potential for skin sensitisation to oseltamivir was observed in a "maximisation" test in guinea pigs.
Approximately 50 % of the animals treated with the unformulated active ingredient showed erythema
after challenging the induced animals. Reversible irritancy of rabbits' eyes was detected.
Whereas very high oral single doses of oseltamivir phosphate salt, up to the highest dose tested
(1310 mg/kg), had no adverse effects in adult rats, such doses resulted in toxicity in juvenile 7-day-old
rat pups, including death. These effects were seen at doses of 657 mg/kg and higher. At 500 mg/kg, no
adverse effects were seen, including upon chronic treatment (500 mg/kg/day administered from 7 to
21 days post partum).
PHARMACEUTICAL PARTICULARS
Capsule core:
Pregelatinized starch (derived from maize starch)
Talc
Povidone
Croscarmellose sodium
Sodium stearyl fumarate
Capsule shell:
Gelatin
Yellow iron oxide (E172)
Red iron oxide (E172)
Titanium dioxide (E171)
Printing ink
:
Shellac
Titanium dioxide (E171)
FD and C Blue 2 (indigo carmine, E132)
6.4 Special precautions for storage
Do not store above 25 °C.
Storage of the pharmacy-compounded suspension
:
Room temperature storage conditions: Stable for 3 weeks (21 days) when stored at room temperature
“do not store above 25 °C”.
Refrigerated storage conditions: Stable for 6 weeks when stored at 2 °C - 8 °C.
6.5 Nature and contents of container
One box contains 10 capsules in a triplex blister pack (PVC/PE/PVDC, sealed with aluminium foil).
6.6 Special precautions for disposal
No special requirements.
Any unused product or waste material should be disposed of in accordance with local requirements.
Special instructions for use, handling and disposal of extemporaneous formulation prepared for
children less than one year of age
Extemporaneous formulation
When commercially manufactured Tamiflu powder for oral suspension is not available, patients who
are unable to swallow capsules may receive appropriate doses of Tamiflu prepared in a pharmacy or
prepared at home.
For infants below 12 months, the pharmacy preparation should be preferred to home preparation.
Detailed information on the pharmacy preparation can be found in section 4.2 and on the home
preparation can be found in section 3 of the package leaflet of Tamiflu capsules.
Syringes of appropriate volume and grading should be provided for administering the pharmacy
compounded suspension as well as for the procedures involved in the home preparation. In both cases,
the correct volumes should preferably be marked on the syringes.
MARKETING AUTHORISATION HOLDER
Roche Registration Limited
6 Falcon Way
Shire Park
Welwyn Garden City
AL7 1TW
United Kingdom
MARKETING AUTHORISATION NUMBER(S)
DATE OF FIRST AUTHORISATION/RENEWAL OF THE AUTHORISATION
Date of first authorisation: 20 June 2002
Date of last renewal: 20 June 2007
10. DATE OF REVISION OF THE TEXT
Detailed information on this medicinal product is available on the website of the European Medicines
Agency (EMEA) http://www.emea.europa.eu/.
NAME OF THE MEDICINAL PRODUCT
Tamiflu 45 mg hard capsule.
QUALITATIVE AND QUANTITATIVE COMPOSITION
Each hard capsule contains oseltamivir phosphate equivalent to 45 mg of oseltamivir.
For a full list of excipients, see section 6.1.
Hard capsule
The hard capsule consists of a grey opaque body bearing the imprint “ROCHE” and a grey opaque cap
bearing the imprint “45 mg”. Imprints are blue.
4.1 Therapeutic indications
Treatment of influenza
In patients one year of age and older who present with symptoms typical of influenza, when influenza
virus is circulating in the community. Efficacy has been demonstrated when treatment is initiated
within two days of first onset of symptoms. This indication is based on clinical studies of naturally
occurring influenza in which the predominant infection was influenza A (see section 5.1).
Tamiflu is indicated for the treatment of infants below 12 months of age during a pandemic influenza
outbreak (see section 5.2).
Prevention of influenza
-
Post-exposure prevention in individuals one year of age or older following contact with a
clinically diagnosed influenza case when influenza virus is circulating in the community.
The appropriate use of Tamiflu for prevention of influenza should be determined on a case by
case basis by the circumstances and the population requiring protection. In exceptional
situations (e.g., in case of a mismatch between the circulating and vaccine virus strains, and a
pandemic situation) seasonal prevention could be considered in individuals one year of age or
older.
Tamiflu is indicated for post-exposure prevention of influenza in infants below 12 months of
age during a pandemic influenza outbreak (see section 5.2).
Tamiflu is not a substitute for influenza vaccination.
The use of antivirals for the treatment and prevention of influenza should be determined on the basis
of official recommendations. Decisions regarding the use of oseltamivir for treatment and prophylaxis
should take into consideration what is known about the characteristics of the circulating influenza
viruses, available information on influenza drug susceptibility patterns for each season and the impact
of the disease in different geographical areas and patient populations (see section 5.1).
Based on limited pharmacokinetic and safety data, Tamiflu can be used in infants below 12 months of
age for treatment during a pandemic influenza outbreak. The treating physician should take into
account the pathogenicity of the circulating strain and the underlying condition of the patient to ensure
there is a potential benefit to the child.
4.2 Posology and method of administration
Tamiflu capsules and Tamiflu suspension are bioequivalent formulations. 75 mg doses can be
administered as either
-
one 30 mg capsule plus one 45 mg capsule or
by administering one 30 mg dose plus one 45 mg dose of suspension.
Adults, adolescents or children ( 1 year of age or older) who are unable to swallow capsules may
receive appropriate doses of Tamiflu suspension.
For infants below 1 year of age
: In the absence of a suitable formulation, a pharmacy compounded
preparation should preferentially be used as the syringe provided in the Tamiflu 12 mg/ml powder for
oral suspension pack (with mg markings) does not allow for appropriate dose adjustments and
commercially available syringes (with ml markings) may lead to unacceptable dosing inaccuracies
(see below 4.2).
Treatment of influenza
Treatment should be initiated as soon as possible within the first two days of onset of symptoms of
influenza.
¾
For adolescents (13 to 17 years of age) and adults
:
The recommended oral dose is 75 mg
oseltamivir twice daily for 5 days.
¾
For infants older than 1 year of age and for children 2 to 12 years of age
: Tamiflu 30 mg and 45 mg
capsules and oral suspension are available.
The following weight-adjusted dosing regimens are recommended for children 1 year of
age and older:
Body Weight
Recommended dose for 5 days
Children who are able to swallow capsules may receive treatment with Tamiflu capsules (30 mg,
45 mg, 75 mg) twice daily for 5 days as an alternative to the recommended dose of Tamiflu
suspension.
¾
For infants below 12 months of age
: The recommended treatment dose for infants less than 12
months is between 2 mg/kg twice daily and 3 mg/kg twice daily during a pandemic influenza
outbreak. This is based upon limited pharmacokinetic data indicating that these doses provide plasma
drug exposures in the majority of patients similar to those shown to be clinically efficacious in older
children and adults (see section 5.2). The following weight-adjusted dosing regimens are
recommended for treatment of infants below 1 year of age:
Age Recommended dose for 5 days
> 3 months to 12 months 3 mg/kg twice daily
> 1 month to 3 months 2.5 mg/kg twice daily
0 to 1 month* 2 mg/kg twice daily
* There is no data available regarding the administration of Tamiflu to infants less than one month of
age.
Administration of Tamiflu to infants less than one year of age should be based upon the judgment of
the physician after considering the potential benefit of treatment versus any potential risk to the infant.


















These age-based dosing recommendations are not intended for premature infants, i.e. those with a
postmenstrual age less than 37 weeks. Insufficient data are available for these patients, in whom
different dosing may be required due to the immaturity of physiological functions
Prevention of influenza
Post-exposure prevention
¾
For adolescents (13 to 17 years of age) and adults
: The recommended dose for prevention of
influenza following close contact with an infected individual is 75 mg oseltamivir once daily for 10
days. Therapy should begin as soon as possible within two days of exposure to an infected individual.
¾
For infants older than 1 year of age and for children 2 to 12 years of age
: Tamiflu 30 mg and 45 mg
capsules and oral suspension are available.
The recommended post-exposure prevention dose of Tamiflu is:
Body Weight
Recommended dose for 10 days
Children who are able to swallow capsules may receive prevention with Tamiflu capsules (30 mg,
45 mg, 75 mg) once daily for 10 days as an alternative to the recommended dose of Tamiflu
suspension.
¾
For infants below 12 months of age
: The recommended prophylaxis dose for infants less than
12 months during a pandemic influenza outbreak is half of the daily treatment dose. This is based upon
clinical data in children > 1 year of age and adults showing that a prophylaxis dose equivalent to half
the daily treatment dose is clinically efficacious for the prevention of influenza. The following
weight-adjusted dosing prophylaxis regimens are recommended for infants below 1 year of age:
Age Recommended dose for 10 days
> 3 months to 12 months 3 mg/kg once daily
> 1 month to 3 months 2.5 mg/kg once daily
0 to 1 month* 2 mg/kg once daily
* There is no data available regarding the administration of Tamiflu to infants less than one month of
age.
Administration of Tamiflu to infants less than one year of age should be based upon the judgment of
the physician after considering the potential benefit of prophylaxis versus any potential risk to the
infant.
These age-based dosing recommendations are not intended for premature infants, i.e. those with a
postmenstrual age less than 37 weeks. Insufficient data are available for these patients, in whom
different dosing may be required due to the immaturity of physiological functions
Prevention during an influenza epidemic in the community
The recommended dose for prevention of influenza during a community outbreak is 75 mg oseltamivir
once daily for up to 6 weeks.
Extemporaneous formulation
When Tamiflu powder for oral suspension is not available




















When commercially manufactured Tamiflu powder for oral suspension is not available, patients who
are unable to swallow capsules may receive appropriate doses of Tamiflu prepared in a pharmacy or
prepared at home.
For infants below 12 months, the pharmacy preparation should be preferred to home preparation.
Detailed information on the home preparation can be found in section 3 of the package leaflet of
Tamiflu capsules.
Pharmacy compounding
¾
Adults and children greater than 1 year who are unable to swallow intact capsules
This procedure describes the preparation of a 15 mg/ml solution that will provide one patient with
enough medication for a 5-day course of treatment or a 10-day course of prophylaxis.
The pharmacist may compound a suspension (15 mg/ml) from Tamiflu 30 mg, 45 mg or 75 mg
capsules using water containing 0.1% w/v sodium benzoate added as a preservative.
First, calculate the Total Volume needed to be compounded and dispensed to provide a 5-day course
of treatment or a 10=day course of prophylaxis for the patient. The Total Volume required is
determined by the weight of the patient according to the recommendation in the table below:
Volume of Compounded Suspension (15 mg/ml) Prepared Based Upon th
e Patient’s Weight
Body Weight
(kg)
Total Volume to Compound per Patient Weight
(ml)
Second, determine the number of capsules and the amount of vehicle (water containing 0.1% w/v
sodium benzoate added as a preservative) that is needed to prepare the Total Volume (calculated from
the table above: 30 ml, 40 ml, 50 ml or 60 ml) of compounded suspension (15 mg/ml) as shown in the
table below:
Number of Capsules and Amount of Vehicle Needed to Prepare the Total Volume of a
Compounded Suspension (15 mg/ml)
Required Number of Tamiflu Capsules
(mg of oseltamivir)
Total Volume
of Compounded
Suspension
to be Prepared
Required Volume
of Vehicle
Please use
alternative capsule
strength*
Please use
alternative capsule
strength*
* No integral number of capsules can be used to achieve the target concentration; therefore, please use
either the 30 mg or 75 mg capsules.
Third, follow the procedure below for compounding the suspension (15 mg/ml) from Tamiflu
capsules:
1.
Carefully separate the capsule body and cap and transfer the contents of the required number of
Tamiflu capsules into a clean mortar.
Triturate the granules to a fine powder.



















3. Add one-third (1/3) of the specified amount of vehicle (water containing 0.1% w/v sodium
benzoate added as a preservative) and triturate the powder until a uniform suspension is
achieved.
4. Transfer the suspension to an amber glass or amber polyethyleneterephthalate (PET) bottle.
A funnel may be used to eliminate any spillage.
5. Add another one-third (1/3) of the vehicle to the mortar, rinse the pestle and mortar by a
triturating motion and transfer the vehicle into the bottle.
6. Repeat the rinsing (Step 5) with the remainder of the vehicle.
7. Close the bottle using a child-resistant cap.
8. Shake well to completely dissolve the active drug and to ensure homogeneous distribution of
the dissolved drug in the resulting suspension.
(Note: Undissolved residue may be visible but is comprised of inert ingredients of Tamiflu
capsules, which are insoluble. However, the active drug, oseltamivir phosphate, readily
dissolves in the specified vehicle and therefore forms a uniform solution.)
9. Put an ancillary label on the bottle indicating
“
Shake Gently Before Use
”
.
10. Instruct the parent or caregiver that after the patient has completed the full course of therapy any
remaining solution must be discarded. It is recommended that this information be provided by
affixing an ancillary label to the bottle or adding a statement to the pharmacy label instructions.
11. Place an appropriate expiration date label according to storage condition (see below).
Storage of the pharmacy-compounded suspension (15 mg/ml)
Room temperature storage conditions: Stable for 3 weeks (21 days) when stored at room temperature
“do not store above 25 °C”.
Refrigerated storage conditions: Stable for 6 weeks when stored at 2 °C - 8 °C.
Place a pharmacy label on the bottle that includes the patient’s name, dosing instructions, use by date,
drug name and any other required information to be in compliance with local pharmacy regulations.
Refer to the table below for the proper dosing instructions.
Prophylaxis Dose
(for 10 days)
10 kg to 15 kg 30 mg 2 ml 2 ml twice daily 2 ml once daily
> 15 to 23 kg 45 mg 3 ml 3 ml twice daily 3 ml once daily
> 23 to 40 kg 60 mg 4 ml 4 ml twice daily 4 ml once daily
> 40 kg 75 mg 5 ml 5 ml twice daily 5 ml once daily
Note: This compounding procedure results in a 15 mg/ml suspension, which is different from the
commercially available Tamiflu powder for oral suspension.
Treatment Dose
(for 5 days)
Dispense the suspension with a graduated oral syringe for measuring small amounts of suspension. If
possible, mark or highlight the graduation corresponding to the appropriate dose (2 ml, 3 ml, 4 ml or
5 ml) on the oral syringe for each patient.
The appropriate dose must be mixed by the caregiver with an equal quantity of sweet liquid food, such
as sugar water, chocolate syrup, cherry syrup, dessert toppings (like caramel or fudge sauce) to mask
the bitter taste.
¾
Infants less than 1 year of age
This procedure describes the preparation of a 10 mg/ml solution that will provide one patient with
enough medication for a 5-day course of treatment or a 10-day course of prophylaxis.
The pharmacist may compound a suspension (10 mg/ml) from Tamiflu 30 mg, 45 mg or 75 mg
capsules using water containing 0.1% w/v sodium benzoate added as a preservative.
Dosing Chart for Pharmacy-Compounded Suspension from Tamiflu Capsules for Children One
Year of Age or Older
Body Weight
(kg)







First, calculate the Total Volume needed to be compounded and dispensed for each patient. The Total
Volume required is determined by the weight of the patient according to the recommendation in the
table below:
Volume of Compounded Suspension (10 mg/ml) Prepared Based Upon th
e Patient’s Weight
Body Weight
(kg)
Total Volume to Compound per Patient Weight
(ml)
Second, determine the number of capsules and the amount of vehicle (water containing 0.1% w/v
sodium benzoate added as a preservative) that is needed to prepare the Total Volume (calculated from
the table above: 30 ml, 45 ml) of compounded suspension (10 mg/ml) as shown in the table below:
Number of Capsules and Amount of Vehicle Needed to Prepare the Total Volume of a
Compounded Suspension (10 mg/ml)
Required Number of Tamiflu Capsules
(mg of oseltamivir)
Required Volume
of Vehicle
Please use
alternative capsule
strength*
* No integral number of capsules can be used to achieve the target concentration; therefore, please use
either the 30 mg or 75 mg capsules.
Third, follow the procedure below for compounding the suspension (10 mg/ml) from Tamiflu
capsules:
1. Carefully separate the capsule body and cap and transfer the contents of the required number of
Tamiflu capsules into a clean mortar.
2. Triturate the granules to a fine powder.
3. Add one-third (1/3) of the specified amount of vehicle and triturate the powder until a uniform
suspension is achieved.
4. Transfer the suspension to an amber glass or amber polyethyleneterephthalate (PET) bottle.
A funnel may be used to eliminate any spillage.
5. Add another one-third (1/3) of the vehicle to the mortar, rinse the pestle and mortar by a
triturating motion and transfer the vehicle into the bottle.
6. Repeat the rinsing (Step 5) with the remainder of the vehicle.
7. Close the bottle using a child-resistant cap.
8. Shake well to completely dissolve the active drug and to ensure homogeneous distribution of
the dissolved drug in the resulting suspension.
(Note: Undissolved residue may be visible but is comprised of inert ingredients of Tamiflu
capsules, which are insoluble. However, the active drug, oseltamivir phosphate, readily
dissolves in the specified vehicle and therefore forms a uniform solution.)
9. Put an ancillary label on the bottle indicating “Shake Gently Before Use”.
10. Instruct the parent or caregiver that after the patient has completed the full course of therapy any
remaining solution must be discarded. It is recommended that this information be provided by
affixing an ancillary label to the bottle or adding a statement to the pharmacy label instructions.
11. Place an appropriate expiration date label according to storage condition (see below).
Storage of the pharmacy-compounded suspension (10 mg/ml)
Room temperature storage conditions: Stable for 3 weeks (21 days) when stored at room temperature
“do not store above 25 °C”.
Refrigerated storage conditions: Stable for 6 weeks when stored at 2 °C - 8 °C.
Total Volume
of Compounded
Suspension
to be Prepared
















Place a pharmacy label on the bottle that includes the patient’s name, dosing instructions, use by date,
drug name and any other required information to be in compliance with local pharmacy regulations.
Refer to the table below for the proper dosing instructions.
Dosing Chart for Pharmacy-Compounded Suspension (10 mg/ml) from Tamiflu Capsules for
Infants Less Than One Month of Age
Body Weight
(rounded to the
nearest 0.5 kg)
Treatment Dose
(for 5 days)
Prophylaxis Dose
(for 10 days)
Dosing Chart for Pharmacy-Compounded Suspension (10 mg/ml) from Tamiflu Capsules for
Infants One to Twelve Months of Age
Body Weight
(rounded to the
nearest 0.5 kg)
Treatment Dose
(for 5 days)
Prophylaxis Dose
(for 10 days)
Note: This compounding procedure results in a 10 mg/ml suspension, which is different from the
commercially available Tamiflu powder for oral suspension.
Dispense the suspension with a graduated oral syringe for measuring small amounts of suspension. If
possible, mark or highlight the graduation corresponding to the appropriate dose on the oral syringe
for each patient.
The appropriate dose must be mixed by the caregiver with an equal quantity of sweet liquid food, such
as sugar water, chocolate syrup, cherry syrup, dessert toppings (like caramel or fudge sauce) to mask
the bitter taste.
Home preparation
When commercially manufactured Tamiflu oral suspension is not available, a pharmacy preparation
from Tamiflu capsules can be used (detailed instructions above in section 4.2). If the pharmacy
preparation is not available either, Tamiflu doses may be prepared at home. The pharmacy preparation
is the preferred option in infants below 12 months of age.
When appropriate capsule strengths are available, the dose is given by opening the capsule and mixing
its contents with no more than one teaspoon of a suitable sweetened food product. The bitter taste can
be masked by products such as sugar water, chocolate syrup, cherry syrup, dessert toppings (like
caramel or fudge sauce). The mixture should be stirred and given entirely to the patient. The mixture
must be swallowed immediately after its preparation.
When only 75 mg capsules are available, and doses of 30 mg or 45 mg are needed, the preparation
involves additional steps. Detailed instructions can be found in section 3 in the package leaflet of
Tamiflu capsules.




















Special populations
Hepatic impairment
No dose adjustment is required either for treatment or for prevention in patients with hepatic
dysfunction. No studies have been carried out in paediatric patients with hepatic disorder.
Renal impairment
Treatment of influenza
: Dose adjustment is recommended for adults with severe renal impairment.
Recommended doses are detailed in the table below.
Creatinine clearance
Recommended dose for treatment
75 mg once daily,
or 30 mg suspension twice daily,
or 30 mg capsules twice daily
Prevention of influenza
: Dose adjustment is recommended for adults with severe renal impairment as
detailed in the table below.
Creatinine clearance
Recommended dose for prevention
75 mg every second day,
or 30 mg suspension once daily,
or 30 mg capsules once daily
Elderly
No dose adjustment is required, unless there is evidence of severe renal impairment.
Children
There is insufficient clinical data available in children with renal impairment to be able to make any
dosing recommendation.
Immunocompromised patients
TLonger duration of seasonal prophylaxis up to 12 weeks has been evaluated in immunocompromised
subjects (see sections 4.4, 4.8 and 5.1).
Hypersensitivity to the active substance or to any of the excipients.
4.4 Special warnings and precautions for use
Oseltamivir is effective only against illness caused by influenza viruses. There is no evidence for
efficacy of oseltamivir in any illness caused by agents other than influenza viruses.
No information is available regarding the safety and efficacy of oseltamivir in patients with any
medical condition sufficiently severe or unstable to be considered at imminent risk of requiring
hospitalisation.
The efficacy of oseltamivir in either treatment or prophylaxis of influenza in immunocompromised
patients has not been firmly established (see section 5.1).
Efficacy of oseltamivir in the treatment of subjects with chronic cardiac disease and/or respiratory
disease has not been established. No difference in the incidence of complications was observed
between the treatment and placebo groups in this population (see section 5.1).














No data allowing a dose recommendation for premature children (< 37 weeks post-menstrual age*) are
currently available.
* Time between first day of last normal menstrual period and day of assessment, gestational age plus
post-natal age.
Tamiflu is not a substitute for influenza vaccination. Use of Tamiflu must not affect the evaluation of
individuals for annual influenza vaccination. The protection against influenza lasts only as long as
Tamiflu is administered. Tamiflu should be used for the treatment and prevention of influenza only
when reliable epidemiological data indicate that influenza virus is circulating in the community.
Susceptibility of circulating influenza virus strains to oseltamivir has been shown to be highly variable
(see section 5.1). Therefore, prescribers should take into account the most recent information available
on oseltamivir susceptibility patterns of the currently circulating viruses when deciding whether to use
Tamiflu.
Severe renal impairment
Dose adjustment is recommended for both treatment and prevention in adults with severe renal
insufficiency. There is insufficient clinical data available in children with renal impairment to be able
to make any dosing recommendation.(see sections 4.2 and 5.2).
Neuropsychiatric events have been reported during administration of Tamiflu in patients with
influenza, especially in children and adolescents. These events are also experienced by patients with
influenza without oseltamivir administration. Patients should be closely monitored for behavioural
changes, and the benefits and risks of continuing treatment should be carefully evaluated for each
patient (see section 4.8).
4.5 Interaction with other medicinal products and other forms of interaction
Pharmacokinetic properties of oseltamivir, such as low protein binding and metabolism independent of
the CYP450 and glucuronidase systems (see section 5.2), suggest that clinically significant drug
interactions via these mechanisms are unlikely.
No dose adjustment is required when co-administering with probenecid in patients with normal renal
function. Co-administration of probenecid, a potent inhibitor of the anionic pathway of renal tubular
secretion, results in an approximate 2-fold increase in exposure to the active metabolite of oseltamivir.
Oseltamivir has no kinetic interaction with amoxicillin, which is eliminated via the same pathway,
suggesting that oseltamivir interaction with this pathway is weak.
Clinically important drug interactions involving competition for renal tubular secretion are unlikely,
due to the known safety margin for most of these substances, the elimination characteristics of the
active metabolite (glomerular filtration and anionic tubular secretion) and the excretion capacity of
these pathways. However, care should be taken when prescribing oseltamivir in subjects when taking
co-excreted agents with a narrow therapeutic margin (e.g., chlorpropamide, methotrexate,
phenylbutazone).
No pharmacokinetic interactions between oseltamivir or its major metabolite have been observed
when co-administering oseltamivir with paracetamol, acetyl-salicylic acid, cimetidine or with antacids
(magnesium and aluminium hydroxides and calcium carbonates).
4.6 Pregnancy and lactation
While no controlled clinical trials have been conducted on the use of oseltamivir in pregnant women,
there is limited data available from post-marketing and retrospective observational surveillance
reports. These data in conjunction with animal studies do not indicate direct or indirect harmful effects
with respect to pregnancy, embryonal/foetal or postnatal development (see section 5.3). Pregnant
women may receive Tamiflu, after considering the available safety information, the pathogenicity of
the circulating influenza virus strain and the underlying condition of the pregnant woman.
In lactating rats, oseltamivir and the active metabolite are excreted in milk. Very limited information is
available on children breast-fed by mothers taking oseltamivir and on excretion of oseltamivir in
breast milk. Limited data demonstrated that oseltamivir and the active metabolite were detected in
breast milk, however the levels were low, which would result in a subtherapeutic dose to the infant.
Considering this information, the pathogenicity of the circulating influenza virus strain and the
underlying condition of the lactating woman, administration of oseltamivir may be considered, where
there are clear potential benefits to lactating mothers.
4.7 Effects on ability to drive and use machines
Tamiflu has no influence on the ability to drive and use machines.
The overall safety profile of Tamiflu is based on data from 2107 adult and 1032 paediatric patients
treated with Tamiflu or placebo for influenza, and on data from 2914 adult and 148 paediatric patients
receiving Tamiflu or placebo for the prophylaxis of influenza in clinical trials. In addition, 475
immunocompromised patients (including 18 children) received Tamiflu or placebo for the prophylaxis
of influenza.
In adults, the most commonly reported adverse drug reactions (ADRs) were vomiting and nausea in
the treatment studies, and nausea and headache in the prevention studies. The majority of these ADRs
were reported on a single occasion on either the first or second treatment day and resolved
spontaneously within 1-2 days. In children, the most commonly reported adverse drug reaction was
vomiting.
The ADRs listed in the tables below fall into the following categories: Very Common (≥
1/10
),
Common (≥
1/100
to <
1/10
), Uncommon (≥
1/1,000
to <
1/100
), Rare (≥
1/10,000
to <
1/1,000
), Very
rare (<
1/10,000
) and not known (cannot be estimated from the available data). ADRs are added to the
appropriate category in the tables according to the pooled analysis from clinical trials. Within each
frequency grouping ADRs are presented in the order of decreasing seriousness.
Treatment and prevention of influenza in adults and adolescents
:
Most Frequent Adverse Drug Reactions (
≥
1 % in the oseltamivir group) in Studies Investigating
Tamiflu for Treatment and Prevention of Influenza in Adults and Adolescents or Through Post-
Marketing Surveillance
System Organ Class (SOC)
Frequency Category
Adverse
Drug
Reaction
Percentage of Patients Experiencing the ADR
Treatment
Oseltamivir
75 mg bid
(n = 1057)
Oseltamivir
75 mg od
(n = 1480)
Infections and infestations
Upper respiratory tract infections
Disorders of the ear and labyrinth











System Organ Class (SOC)
Frequency Category
Adverse
Drug
Reaction
Percentage of Patients Experiencing the ADR
Treatment
Oseltamivir
75 mg bid
(n = 1057)
Oseltamivir
75 mg od
(n = 1480)
Respiratory, thoracic and
mediastinal disorders
Gastrointestinal disorders
Skin and subcutaneous tissue
disorders
Uncommon
:
Dermatitis
a
< 1 % < 1 % 1 % 1 %
Rash
a
< 1 % < 1 % < 1 % < 1 %
Urticaria
a
< 1 % < 1 % < 1 % < 1 %
Eczema
a
< 1 % 0 % < 1 % < 1 %
General disorders
Common
:
Dizziness 2 % 3 % 2 % 2 %
Fatigue 1 % 1 % 8 % 8 %
Pain < 1 % < 1 % 4 % 3 %
a
These are events identified during post-marketing surveillance. They were also reported in the
pooled clinical studies at the incidence presented in the table above.
b
Subjects who experienced nausea alone; excludes subjects who experienced nausea in association
with vomiting.
c
The difference between the placebo and oseltamivir groups was statistically significant.
Treatment and prevention of influenza in children
:
The table below shows the most frequently reported ADRs from paediatric clinical trials.
Most Frequent Adverse Drug Reactions (
≥
1 % in the oseltamivir group in the treatment studies
and
≥
10 in the oseltamivir group in the prophylaxis study) in Children
Percentage of Patients Experiencing the ADR
Treatment
Oseltamivir
2 mg/kg bid
(n
=
515)
Oseltamivir
30 to 75 mg
b
(n
=
158)
Oseltamivir
30 to 75 mg
b
(n
=
148)
Infections and infestations
System Organ Class (SOC)
Frequency Category
Adverse Drug Reaction


















System Organ Class (SOC)
Frequency Category
Adverse Drug Reaction
Percentage of Patients Experiencing the ADR
Treatment
Oseltamivir
2 mg/kg bid
(n
=
515)
Oseltamivir
30 to 75 mg
b
(n
=
158)
Oseltamivir
30 to 75 mg
b
(n
=
148)
Disorders of the blood and
lymphatic system
Respiratory, thoracic and
mediastinal disorders
Asthma (incl. aggravated)
Gastrointestinal disorder
Disorders of the ear and labyrinth
Tympanic membrane disorder
Skin and subcutaneous tissue
disorders
a
The prevention study did not contain a placebo arm, i.e. was an uncontrolled study.
b
Unit dose = weight-based dosing (see section 4.2).
c
Patients experienced ear ache and ear pain.
In general, the adverse event profile in children with pre-existing bronchial asthma was qualitatively
similar to that of otherwise healthy children.
Further post marketing surveillance data on selected serious adverse drug reactions
:
Immune system disorders
Frequency not known: hypersensitivity reactions, including anaphylactic/anaphylactoid reactions.
Psychiatric disorders and nervous system disorders
Frequency not known: influenza can be associated with a variety of neurologic and behavioural
symptoms which can include events such as hallucinations, delirium, and abnormal behaviour, in some
cases resulting in fatal outcomes. These events may occur in the setting of encephalitis or
encephalopathy but can occur without obvious severe disease.
In patients with influenza who were receiving Tamiflu, there have been postmarketing reports of
convulsions and delirium (including symptoms such as altered level of consciousness, confusion,
abnormal behaviour, delusions, hallucinations, agitation, anxiety, nightmares), in a very few cases
resulting in self-injury or fatal outcomes. These events were reported primarily among pediatric and
adolescent patients and often had an abrupt onset and rapid resolution. The contribution of Tamiflu to















those events is unknown. Such neuropsychiatric events have also been reported in patients with
influenza who were not taking Tamiflu.
Eye disorders
Frequency not known: visual disturbance.
Cardiac disorders
Frequency not known: cardiac arrhythmia.
Gastrointestinal disorders
Frequency not known
:
gastrointestinal bleedings and hemorrhagic colitis.
Hepato-biliary disorders
Frequency not known: hepato-biliary system disorders, including hepatitis and elevated liver enzymes
in patients with influenza-like illness. These cases include fatal fulminant hepatitis/hepatic failure.
Skin and subcutaneous tissue disorders
Frequency not known: severe skin reactions, including Stevens-Johnson syndrome, toxic epidermal
necrolysis, erythema multiforme and angioneurotic oedema.
Additional information on special populations
:
Infants less than one year of age
Safety information available on oseltamivir administered for treatment of influenza in infants less than
one year of age from prospective and retrospective observational trials (comprising together more than
2400 infants of that age class), epidemiological databases research and postmarketing reports suggest
that the safety profile in infants less than one year of age is similar to the established safety profile of
children aged one year and older.
Elderly patients
There were no clinically relevant differences in the safety population of the elderly subjects who
received oseltamivir or placebo compared with the adult population aged up to 65 years.
Patients with chronic cardiac and/or respiratory disease
The adverse event profile in adolescents and patients with chronic cardiac and/or respiratory disease
was qualitatively similar to those of healthy young adults.
Immunocompromised patients
The adverse reactions noted in immunocompromised subjects 13 years of age and older who received
oseltamivir during 12 weeks for the seasonal prophylaxis of influenza were consistent with those
previously observed in Tamiflu clinical trials.
There is no experience with overdose. However, the anticipated manifestations of acute overdose
would be nausea, with or without accompanying vomiting, and dizziness. Patients should discontinue
the treatment in the event of overdose. No specific antidote is known.
PHARMACOLOGICAL PROPERTIES
5.1 Pharmacodynamic properties
Pharmacotherapeutic group: Antiviral ATC code: J05AH02
Oseltamivir phosphate is a pro-drug of the active metabolite (oseltamivir carboxylate). The active
metabolite is a selective inhibitor of influenza virus neuraminidase enzymes, which are glycoproteins
found on the virion surface. Viral neuraminidase enzyme activity is important both for viral entry into
uninfected cells and for the release of recently formed virus particles from infected cells, and for the
further spread of infectious virus in the body.
Oseltamivir carboxylate inhibits influenza A and B neuraminidases
in vitro
. Oseltamivir phosphate
inhibits influenza virus infection and replication
in vitro
. Oseltamivir given orally inhibits influenza A
and B virus replication and pathogenicity
in vivo
in animal models of influenza infection at antiviral
exposures similar to that achieved in man with 75 mg twice daily
.
Antiviral activity of oseltamivir was supported for influenza A and B by experimental challenge
studies in healthy volunteers.
Neuraminidase enzyme IC50 values for oseltamivir for clinically isolated influenza A ranged from
0.1 nM to 1.3 nM, and for influenza B was 2.6 nM. Higher IC50 values for influenza B, up to a
median of 8.5 nM, have been observed in published trials.
Reduced sensitivity of viral neuraminidase
Clinical studies
: The risk of emergence of influenza viruses with reduced susceptibility or frank
resistance to oseltamivir has been examined during Roche-sponsored clinical studies. All patients who
were found to carry oseltamivir-resistant virus did so transiently, cleared the virus normally and
showed no clinical deterioration.
Patients with Resistance Mutations (%)
* Full genotyping was not performed in all studies.
There has been no evidence for emergence of drug resistance associated with the use of Tamiflu in
clinical studies conducted to date in post-exposure (7 days), post-exposure within household groups
(10 days) and seasonal (42 days) prevention of influenza in immunocompetent subjects. There was no
resistance observed during a 12-week prophylaxis study in immunocompromised subjects.
Clinical and surveillance data
: Natural mutations associated with reduced susceptibility to oseltamivir
in vitro
have been detected in influenza A and B viruses isolated from patients without exposure to
oseltamivir. Resistant strains selected during oseltamivir treatment have been isolated from both
immunocompetent and immunocompromised patients. Immunocompromised patients and young
children are at a higher risk of developing oseltamivir-resistant virus during treatment.
Oseltamivir-resistant viruses isolated from oseltamivir-treated patients and oseltamivir-resistant
laboratory strains of influenza viruses have been found to contain mutations in N1 and N2
neuraminidases. Resistance mutations tend to be viral sub-type specific. Since 2007 resistance
associated H275Y mutation in seasonal H1N1 strains has become widespread. The susceptibility to
oseltamivir and the prevalence of such viruses appear to vary seasonally and geographically. In 2008,
H275Y was found in > 99 % of circulating H1N1 influenza isolates in Europe. The 2009 H1N1
influenza (“swine flu”) was almost uniformly susceptible to oseltamivir, with only sporadic reports of
resistance in connection with both therapeutic and prophylactic regimens.
Treatment of influenza infection
Oseltamivir is effective only against illnesses caused by influenza virus. Statistical analyses are
therefore presented only for influenza-infected subjects. In the pooled treatment study population,
which included both influenza-positive and -negative subjects (ITT), primary efficacy was reduced
proportional to the number of influenza-negative individuals. In the overall treatment population,
influenza infection was confirmed in 67 % (range 46 % to 74 %) of the recruited patients. Of the
elderly subjects, 64 % were influenza-positive and of those with chronic cardiac and/or respiratory
disease 62 % were influenza-positive. In all phase III treatment studies, patients were recruited only
during the period in which influenza was circulating in the local community.








Adults and adolescents 13 years of age and older
: Patients were eligible if they reported within
36 hours of onset of symptoms, had fever ≥ 37.8 °C, accompanied by at least one respiratory symptom
(cough, nasal symptoms or sore throat) and at least one systemic symptom (myalgia, chills/sweats,
malaise, fatigue or headache). In a pooled analysis of all influenza-positive adults and adolescents
(N = 2413) enrolled into treatment studies, oseltamivir 75 mg twice daily for 5 days reduced the
median duration of influenza illness by approximately one day from 5.2 days (95 % CI 4.9 – 5.5 days)
in the placebo group to 4.2 days (95 % CI 4.0 – 4.4 days; p
≤
0.0001).
The proportion of subjects who developed specified lower respiratory tract complications (mainly
bronchitis) treated with antibiotics was reduced from 12.7 % (135/1063) in the placebo group to 8.6 %
(116/1350) in the oseltamivir treated population (p = 0.0012).
Treatment of influenza in high risk populations
: The median duration of influenza illness in elderly
subjects (≥ 65 years) and in subjects with chronic cardiac and/or respiratory disease receiving
oseltamivir 75 mg twice daily for 5 days was
not
reduced significantly. The total duration of fever was
reduced by one day in the groups treated with oseltamivir. In the influenza-positive elderly,
oseltamivir significantly reduced the incidence of specified lower respiratory tract complications
(mainly bronchitis) treated with antibiotics from 19 % (52/268) in the placebo group to 12 % (29/250)
in the oseltamivir treated population (p = 0.0156).
In influenza-positive patients with chronic cardiac and/or respiratory disease, the combined incidence
of lower respiratory tract complications (mainly bronchitis) treated with antibiotics was 17 % (22/133)
in the placebo group and 14 % (16/118) in the oseltamivir treated population (p = 0.5976).
Treatment of influenza in children
: In a study of otherwise healthy children (65 % influenza-positive)
aged 1 to 12 years (mean age 5.3 years) who had fever (≥ 37.8 °C) plus either cough or coryza, 67 %
of
influenza-positive patients were infected with influenza A and 33 % with influenza B. Oseltamivir
treatment, started within 48 hours of onset of symptoms, significantly reduced the time to freedom
from illness (defined as the simultaneous return to normal health and activity and alleviation of fever,
cough and coryza) by 1.5 days (95 % CI 0.6 – 2.2 days; p < 0.0001) compared to placebo. Oseltamivir
reduced the incidence of acute otitis media from 26.5 % (53/200) in the placebo group to 16 %
(29/183) in the oseltamivir treated children (p = 0.013).
A second study was completed in 334 asthmatic children aged 6 to 12 years old of which 53.6 % were
influenza-positive. In the oseltamivir treated group, the median duration of illness was
not
reduced
significantly. By day 6 (the last day of treatment) FEV
1
had increased by 10.8 % in the oseltamivir
treated group compared to 4.7 % on placebo (p = 0.0148) in this population.
The European Medicines Agency has deferred the obligation to submit the results of studies with
Tamiflu in one or more subsets of the paediatric population in influenza. See section 4.2 for
information on paediatric use.
Treatment of influenza B infection
:
Overall, 15 % of the influenza-positive population were infected
by influenza B, proportions ranging from 1 to 33 % in individual studies. The median duration of
illness in influenza B infected subjects did not differ significantly between the treatment groups in
individual studies. Data from 504 influenza B infected subjects were pooled across all studies for
analysis. Oseltamivir reduced the time to alleviation of all symptoms by 0.7 days (95 % CI 0.1 – 1.6
days; p = 0.022) and the duration of fever (≥ 37.8 °C), cough and coryza by one day (95 % CI 0.4 –
1.7 days; p < 0.001) compared to placebo.
Prevention of influenza
The efficacy of oseltamivir in preventing naturally occurring influenza illness has been demonstrated
in a post-exposure prevention study in households and two seasonal prevention studies. The primary
efficacy parameter for all of these studies was the incidence of laboratory-confirmed influenza. The
virulence of influenza epidemics is not predictable and varies within a region and from season to
season, therefore the number needed to treat (NNT) in order to prevent one case of influenza illness
varies.
Post-exposure prevention
: In a study in contacts (12.6 % vaccinated against influenza) of an index
case of influenza, oseltamivir 75 mg once daily was started within 2 days of onset of symptoms in the
index case and continued for seven days. Influenza was confirmed in 163 out of 377 index cases.
Oseltamivir significantly reduced the incidence of clinical influenza illness occurring in the contacts of
confirmed influenza cases from 24/200 (12 %) in the placebo group to 2/205 (1 %) in the oseltamivir
group (92 % reduction [95 % CI 6 – 16; p
≤
0.0001]). The number needed to treat (NNT) in contacts
of true influenza cases was 10 (95 % CI 9 – 12) and was 16 (95 % CI 15 – 19) in the whole population
(ITT) regardless of infection status in the index case.
The efficacy of oseltamivir in preventing naturally occurring influenza illness has been demonstrated
in a post-exposure prevention study in households that included adults, adolescents, and children aged
1 to 12 years, both as index cases and as family contacts. The primary efficacy parameter for this study
was the incidence of laboratory-confirmed clinical influenza in the households. Oseltamivir
prophylaxis lasted for 10 days. In the total population, there was a reduction in the incidence of
laboratory-confirmed clinical influenza in households from 20 % (27/136) in the group not receiving
prevention to 7 % (10/135) in the group receiving prevention (62.7 % reduction [95 % CI 26.0 – 81.2;
p = 0.0042]). In households of influenza-infected index cases, there was a reduction in the incidence of
influenza from 26 % (23/89) in the group not receiving prevention to 11 % (9/84) in the group
receiving prevention (58.5 % reduction [95 % CI 15.6 – 79.6; p = 0.0114]).
According to subgroup analysis in children at 1 to 12 years of age, the incidence of laboratory-
confirmed clinical influenza among children was significantly reduced from 19 % (21/111) in the
group not receiving prevention to 7 % (7/104) in the group receiving prevention (64.4 % reduction
[95 % CI 15.8 – 85.0; p = 0.0188]). Among children who were not already shedding virus at baseline,
the incidence of laboratory-confirmed clinical influenza was reduced from 21 % (15/70) in the group
not receiving prevention to 4 % (2/47) in the group receiving prevention (80.1 % reduction [95 % CI
22.0 – 94.9; p = 0.0206]). The NNT for the total paediatric population was 9 (95 % CI 7 – 24) and
8 (95 % CI 6, upper limit not estimable) in the whole population (ITT) and in paediatric contacts of
infected index cases (ITTII), respectively.
Prevention during an influenza epidemic in the community
: In a pooled analysis of two other studies
conducted in unvaccinated otherwise healthy adults, oseltamivir 75 mg once daily given for 6 weeks
significantly reduced the incidence of clinical influenza illness from 25/519 (4.8 %) in the placebo
group to 6/520 (1.2 %) in the oseltamivir group (76 % reduction [95 % CI 1.6 – 5.7; p = 0.0006])
during a community outbreak of influenza. The NNT in this study was 28 (95 % CI 24 – 50).
A study in elderly residents of nursing homes, where 80 % of participants received vaccine in the
season of the study, oseltamivir 75 mg once daily given for 6 weeks significantly reduced the
incidence of clinical influenza illness from 12/272 (4.4 %) in the placebo group to 1/276 (0.4 %) in the
oseltamivir group (92 % reduction [95 % CI 1.5 – 6.6; p = 0.0015]). The NNT in this study was
25 (95 % CI 23 – 62).
Prophylaxis of influenza in immunocompromised patients
:
A double-blind, placebo-controlled,
randomised study was conducted for seasonal prophylaxis of influenza in 475 immunocompromised
subjects (388 subjects with solid organ transplantation [195 placebo; 193 oseltamivir], 87 subjects
with hemopoetic stem cell transplantation [43 placebo; 44 oseltamivir], no subject with other
immunosuppressant conditions), including 18 children 1 to 12 years of age. The primary endpoint in
this study was the incidence of laboratory-confirmed clinical influenza as determined by viral culture
and/or a four-fold rise in HAI antibodies. The incidence of laboratory-confirmed clinical influenza was
2.9 % (7/238) in the placebo group and 2.1 % (5/237) in the oseltamivir group (95 % CI -2.3 % –
4.1 %; p = 0.772).
Specific studies have not been conducted to assess of the reduction in the risk of complications.
5.2 Pharmacokinetic properties
Absorption
Oseltamivir is readily absorbed from the gastrointestinal tract after oral administration of oseltamivir
phosphate (pro-drug) and is extensively converted by predominantly hepatic esterases to the active
metabolite (oseltamivir carboxylate). At least 75 % of an oral dose reaches the systemic circulation as
the active metabolite. Exposure to the pro-drug is less than 5 % relative to the active metabolite.
Plasma concentrations of both pro-drug and active metabolite are proportional to dose and are
unaffected by co-administration with food.
Distribution
The mean volume of distribution at steady state of the oseltamivir carboxylate is approximately
23 litres in humans, a volume equivalent to extracellular body fluid. Since neuraminidase activity is
extracellular, oseltamivir carboxylate distributes to all sites of influenza virus spread.
The binding of the oseltamivir carboxylate to human plasma protein is negligible (approximately 3 %).
Metabolism
Oseltamivir is extensively converted to oseltamivir carboxylate by esterases located predominantly in
the liver.
In vitro
studies demonstrated that neither oseltamivir nor the active metabolite is a substrate
for, or an inhibitor of, the major cytochrome P450 isoforms. No phase 2 conjugates of either
compound have been identified
in vivo
.
Elimination
Absorbed oseltamivir is primarily (> 90 %) eliminated by conversion to oseltamivir carboxylate. It is
not further metabolised and is eliminated in the urine. Peak plasma concentrations of oseltamivir
carboxylate decline with a half-life of 6 to 10 hours in most subjects. The active metabolite is
eliminated entirely by renal excretion. Renal clearance (18.8 l/h) exceeds glomerular filtration rate
(7.5 l/h) indicating that tubular secretion occurs in addition to glomerular filtration. Less than 20 % of
an oral radiolabelled dose is eliminated in faeces.
Renal impairment
Administration of 100 mg oseltamivir phosphate twice daily for 5 days to patients with various
degrees of renal impairment showed that exposure to oseltamivir carboxylate is inversely proportional
to declining renal function. For dosing, see section 4.2.
Hepatic impairment
In vitro
studies have concluded that exposure to oseltamivir is not expected to be increased
significantly nor is exposure to the active metabolite expected to be significantly decreased in patients
with hepatic impairment (see section 4.2).
Elderly
Exposure to the active metabolite at steady state was 25 to 35 % higher in elderly (age 65 to 78 years)
compared to adults less than 65 years of age given comparable doses of oseltamivir. Half-lives
observed in the elderly were similar to those seen in young adults. On the basis of drug exposure and
tolerability, dosage adjustments are not required for elderly patients unless there is evidence of severe
renal impairment (creatinine clearance below 30 ml/min) (see section 4.2).
Children
Children one year of age and older
: The pharmacokinetics of oseltamivir have been evaluated in
single-dose pharmacokinetic studies in children aged 1 to 16 years. Multiple-dose pharmacokinetics
were studied in a small number of children enrolled in a clinical efficacy study. Younger children
cleared both the pro-drug and its active metabolite faster than adults, resulting in a lower exposure for
a given mg/kg dose. Doses of 2 mg/kg give oseltamivir carboxylate exposures comparable to those
achieved in adults receiving a single 75 mg dose (approximately 1 mg/kg). The pharmacokinetics of
oseltamivir in children over 12 years of age are similar to those in adults.
Infants below 12 months of age:
Limited pharmacokinetic and safety data are available for infants less
than 2 years of age. Pharmacokinetic modeling was undertaken using these data in addition to data
from studies in adults and children older than 1 year of age. The results demonstrate that doses of
3 mg/kg twice daily for infants aged 3 to 12 months and 2.5 mg/kg twice daily for infants aged
between 1 and 3 months provide exposures similar to those shown to be clinically efficacious in adults
and children > 1 year of age (see sections 4.1 and 4.2). There are currently no data available in patients
less than 1 month of age using Tamiflu.
5.3 Preclinical safety data
Preclinical data reveal no special hazard for humans based on conventional studies of safety
pharmacology, repeated-dose toxicity and genotoxicity. Results of the conventional rodent
carcinogenicity studies showed a trend towards a dose-dependent increase in the incidence of some
tumours that are typical for the rodent strains used. Considering the margins of exposure in relation to
the expected exposure in the human use, these findings do not change the benefit-risk of Tamiflu in its
adopted therapeutic indications.
Teratology studies have been conducted in rats and rabbits at doses of up to 1500 mg/kg/day and
500 mg/kg/day, respectively. No effects on foetal development were observed. A rat fertility study up
to a dose of 1500 mg/kg/day demonstrated no adverse effects on either sex. In pre- and post-natal rat
studies, prolonged parturition was noted at 1500 mg/kg/day: the safety margin between human
exposure and the highest no-effect dose (500 mg/kg/day) in rats is 480-fold for oseltamivir and 44-fold
for the active metabolite, respectively. Foetal exposure in the rats and rabbits was approximately
15 to 20 % of that of the mother.
In lactating rats, oseltamivir and the active metabolite are excreted in the milk. Limited data indicate
that oseltamivir and the active metabolite are excreted in human milk. Extrapolation of the animal data
provides estimates of 0.01 mg/day and 0.3 mg/day for the respective compounds.
A potential for skin sensitisation to oseltamivir was observed in a "maximisation" test in guinea pigs.
Approximately 50 % of the animals treated with the unformulated active ingredient showed erythema
after challenging the induced animals. Reversible irritancy of rabbits' eyes was detected.
Whereas very high oral single doses of oseltamivir phosphate salt, up to the highest dose tested
(1310 mg/kg), had no adverse effects in adult rats, such doses resulted in toxicity in juvenile 7-day-old
rat pups, including death. These effects were seen at doses of 657 mg/kg and higher. At 500 mg/kg, no
adverse effects were seen, including upon chronic treatment (500 mg/kg/day administered from 7 to
21 days post partum).
PHARMACEUTICAL PARTICULARS
Capsule core:
Pregelatinized starch (derived from maize starch)
Talc
Povidone
Croscarmellose sodium
Sodium stearyl fumarate
Capsule shell:
Gelatin
Black iron oxide (E172)
Titanium dioxide (E171)
Printing ink
:
Shellac
Titanium dioxide (E171)
FD and C Blue 2 (indigo carmine, E132)
6.4 Special precautions for storage
Do not store above 25 °C.
Storage of the pharmacy-compounded suspension
:
Room temperature storage conditions: Stable for 3 weeks (21 days) when stored at room temperature
“do not store above 25 °C”.
Refrigerated storage conditions: Stable for 6 weeks when stored at 2 °C - 8 °C.
6.5 Nature and contents of container
One box contains 10 capsules in a triplex blister pack (PVC/PE/PVDC, sealed with aluminium foil).
6.6 Special precautions for disposal
No special requirements.
Any unused product or waste material should be disposed of in accordance with local requirements.
Special instructions for use, handling and disposal of extemporaneous formulation prepared for
children less than one year of age
Extemporaneous formulation
When commercially manufactured Tamiflu powder for oral suspension is not available, patients who
are unable to swallow capsules may receive appropriate doses of Tamiflu prepared in a pharmacy or
prepared at home.
For infants below 12 months, the pharmacy preparation should be preferred to home preparation.
Detailed information on the pharmacy preparation can be found in section 4.2 and on the home
preparation can be found in section 3 of the package leaflet of Tamiflu capsules.
Syringes of appropriate volume and grading should be provided for administering the pharmacy
compounded suspension as well as for the procedures involved in the home preparation. In both cases,
the correct volumes should preferably be marked on the syringes.
MARKETING AUTHORISATION HOLDER
Roche Registration Limited
6 Falcon Way
Shire Park
Welwyn Garden City
AL7 1TW
United Kingdom
MARKETING AUTHORISATION NUMBER(S)
DATE OF FIRST AUTHORISATION/RENEWAL OF THE AUTHORISATION
Date of first authorisation: 20 June 2002
Date of last renewal: 20 June 2007
10. DATE OF REVISION OF THE TEXT
Detailed information on this medicinal product is available on the website of the European Medicines
Agency (EMEA) http://www.emea.europa.eu/.
NAME OF THE MEDICINAL PRODUCT
Tamiflu 75 mg hard capsule.
QUALITATIVE AND QUANTITATIVE COMPOSITION
Each hard capsule contains oseltamivir phosphate equivalent to 75 mg of oseltamivir.
For a full list of excipients, see section 6.1.
Hard capsule
The hard capsule consists of a grey opaque body bearing the imprint “ROCHE” and a light yellow
opaque cap bearing the imprint “75 mg”. Imprints are blue.
4.1 Therapeutic indications
Treatment of influenza
In patients one year of age and older who present with symptoms typical of influenza, when influenza
virus is circulating in the community. Efficacy has been demonstrated when treatment is initiated
within two days of first onset of symptoms. This indication is based on clinical studies of naturally
occurring influenza in which the predominant infection was influenza A (see section 5.1).
Tamiflu is indicated for the treatment of infants below 12 months of age during a pandemic influenza
outbreak (see section 5.2).
Prevention of influenza
-
Post-exposure prevention in individuals one year of age or older following contact with a
clinically diagnosed influenza case when influenza virus is circulating in the community.
The appropriate use of Tamiflu for prevention of influenza should be determined on a case by
case basis by the circumstances and the population requiring protection. In exceptional
situations (e.g., in case of a mismatch between the circulating and vaccine virus strains, and a
pandemic situation) seasonal prevention could be considered in individuals one year of age or
older.
Tamiflu is indicated for post-exposure prevention of influenza in infants below 12 months of
age during a pandemic influenza outbreak (see section 5.2).
Tamiflu is not a substitute for influenza vaccination.
The use of antivirals for the treatment and prevention of influenza should be determined on the basis
of official recommendations. Decisions regarding the use of oseltamivir for treatment and prophylaxis
should take into consideration what is known about the characteristics of the circulating influenza
viruses, available information on influenza drug susceptibility patterns for each season and the impact
of the disease in different geographical areas and patient populations (see section 5.1).
Based on limited pharmacokinetic and safety data, Tamiflu can be used in infants below 12 months of
age for treatment during a pandemic influenza outbreak. The treating physician should take into
account the pathogenicity of the circulating strain and the underlying condition of the patient to ensure
there is a potential benefit to the child.
4.2 Posology and method of administration
Tamiflu capsules and Tamiflu suspension are bioequivalent formulations. 75 mg doses can be
administered as either
-
by administering one 30 mg dose plus one 45 mg dose of suspension.
Adults, adolescents or children ( 1 year of age or older) who are unable to swallow capsules may
receive appropriate doses of Tamiflu suspension.
For infants below 1 year of age
: In the absence of a suitable formulation, a pharmacy compounded
preparation should preferentially be used as the syringe provided in the Tamiflu 12 mg/ml powder for
oral suspension pack (with mg markings) does not allow for appropriate dose adjustments and
commercially available syringes (with ml markings) may lead to unacceptable dosing inaccuracies
(see below 4.2).
Treatment of influenza
Treatment should be initiated as soon as possible within the first two days of onset of symptoms of
influenza.
¾
For adolescents (13 to 17 years of age) and adults
:
The recommended oral dose is 75 mg
oseltamivir twice daily for 5 days.
¾
For infants older than 1 year of age and for children 2 to 12 years of age
: Tamiflu 30 mg and 45 mg
capsules and oral suspension are available.
For recommended treatment dose of Tamiflu for infants older than 1 year of age and for children 2 to
12 years of age, see SmPC of Tamiflu suspension and Tamiflu 30 and 45 mg capsules.
Children who are able to swallow capsules may receive treatment with Tamiflu capsules (30 mg,
45 mg, 75 mg) twice daily for 5 days as an alternative to the recommended dose of Tamiflu
suspension.
¾
For infants below 12 months of age
: The recommended treatment dose for infants less than 12
months is between 2 mg/kg twice daily and 3 mg/kg twice daily during a pandemic influenza
outbreak. This is based upon limited pharmacokinetic data indicating that these doses provide plasma
drug exposures in the majority of patients similar to those shown to be clinically efficacious in older
children and adults (see section 5.2). The following weight-adjusted dosing regimens are
recommended for treatment of infants below 1 year of age:
Age Recommended dose for 5 days
> 3 months to 12 months 3 mg/kg twice daily
> 1 month to 3 months 2.5 mg/kg twice daily
0 to 1 month* 2 mg/kg twice daily
* There is no data available regarding the administration of Tamiflu to infants less than one month of
age.
Administration of Tamiflu to infants less than one year of age should be based upon the judgment of
the physician after considering the potential benefit of treatment versus any potential risk to the infant.
These age-based dosing recommendations are not intended for premature infants, i.e. those with a
postmenstrual age less than 37 weeks. Insufficient data are available for these patients, in whom
different dosing may be required due to the immaturity of physiological functions
one 30 mg capsule plus one 45 mg capsule or










Prevention of influenza
Post-exposure prevention
¾
For adolescents (13 to 17 years of age) and adults
: The recommended dose for prevention of
influenza following close contact with an infected individual is 75 mg oseltamivir once daily for 10
days. Therapy should begin as soon as possible within two days of exposure to an infected individual.
¾
For infants older than 1 year of age and for children 2 to 12 years of age
: Tamiflu 30 mg and 45 mg
capsules and oral suspension are available.
For recommended post-exposure prevention dose of Tamiflu for infants older than 1 year of age and
for children of 2 to 12 years of age, see SmPC of Tamiflu suspension and Tamiflu 30 mg and 45 mg
capsules.
Children who are able to swallow capsules may receive prevention with Tamiflu capsules (30 mg,
45 mg, 75 mg) once daily for 10 days as an alternative to the recommended dose of Tamiflu
suspension.
¾
For infants below 12 months of age
: The recommended prophylaxis dose for infants less than
12 months during a pandemic influenza outbreak is half of the daily treatment dose. This is based upon
clinical data in children > 1 year of age and adults showing that a prophylaxis dose equivalent to half
the daily treatment dose is clinically efficacious for the prevention of influenza. The following
weight-adjusted dosing prophylaxis regimens are recommended for infants below 1 year of age:
Age Recommended dose for 10 days
> 3 months to 12 months 3 mg/kg once daily
> 1 month to 3 months 2.5 mg/kg once daily
0 to 1 month* 2 mg/kg once daily
* There is no data available regarding the administration of Tamiflu to infants less than one month of
age.
Administration of Tamiflu to infants less than one year of age should be based upon the judgment of
the physician after considering the potential benefit of prophylaxis versus any potential risk to the
infant.
These age-based dosing recommendations are not intended for premature infants, i.e. those with a
postmenstrual age less than 37 weeks. Insufficient data are available for these patients, in whom
different dosing may be required due to the immaturity of physiological functions
Prevention during an influenza epidemic in the community
The recommended dose for prevention of influenza during a community outbreak is 75 mg oseltamivir
once daily for up to 6 weeks.
Extemporaneous formulation
When Tamiflu powder for oral suspension is not available
When commercially manufactured Tamiflu powder for oral suspension is not available, patients who
are unable to swallow capsules may receive appropriate doses of Tamiflu prepared in a pharmacy or
prepared at home.
For infants below 12 months, the pharmacy preparation should be preferred to home preparation.
Detailed information on the home preparation can be found in section 3 of the package leaflet of
Tamiflu capsules.
Pharmacy compounding
¾
Adults and children greater than 1 year who are unable to swallow intact capsules
This procedure describes the preparation of a 15 mg/ml solution that will provide one patient with
enough medication for a 5-day course of treatment or a 10-day course of prophylaxis.














The pharmacist may compound a suspension (15 mg/ml) from Tamiflu 30 mg, 45 mg or 75 mg
capsules using water containing 0.1% w/v sodium benzoate added as a preservative.
First, calculate the Total Volume needed to be compounded and dispensed to provide a 5-day course
of treatment or a 10=day course of prophylaxis for the patient. The Total Volume required is
determined by the weight of the patient according to the recommendation in the table below:
Volume of Compounded Suspension (15 mg/ml) Prepared Based Upon th
e Patient’s Weight
Body Weight
(kg)
Total Volume to Compound per Patient Weight
(ml)
Second, determine the number of capsules and the amount of vehicle (water containing 0.1% w/v
sodium benzoate added as a preservative) that is needed to prepare the Total Volume (calculated from
the table above: 30 ml, 40 ml, 50 ml or 60 ml) of compounded suspension (15 mg/ml) as shown in the
table below:
Number of Capsules and Amount of Vehicle Needed to Prepare the Total Volume of a
Compounded Suspension (15 mg/ml)
Required Number of Tamiflu Capsules
(mg of oseltamivir)
Total Volume
of Compounded
Suspension
to be Prepared
Required Volume
of Vehicle
Please use
alternative capsule
strength*
Please use
alternative capsule
strength*
* No integral number of capsules can be used to achieve the target concentration; therefore, please use
either the 30 mg or 75 mg capsules.
Third, follow the procedure below for compounding the suspension (15 mg/ml) from Tamiflu
capsules:
1.
Carefully separate the capsule body and cap and transfer the contents of the required number of
Tamiflu capsules into a clean mortar.
Add one-third (1/3) of the specified amount of vehicle (water containing 0.1% w/v sodium
benzoate added as a preservative) and triturate the powder until a uniform suspension is
achieved.
Transfer the suspension to an amber glass or amber polyethyleneterephthalate (PET) bottle.
A funnel may be used to eliminate any spillage.
Add another one-third (1/3) of the vehicle to the mortar, rinse the pestle and mortar by a
triturating motion and transfer the vehicle into the bottle.
Repeat the rinsing (Step 5) with the remainder of the vehicle.
Close the bottle using a child-resistant cap.
Triturate the granules to a fine powder.



















8. Shake well to completely dissolve the active drug and to ensure homogeneous distribution of
the dissolved drug in the resulting suspension.
(Note: Undissolved residue may be visible but is comprised of inert ingredients of Tamiflu
capsules, which are insoluble. However, the active drug, oseltamivir phosphate, readily
dissolves in the specified vehicle and therefore forms a uniform solution.)
9. Put an ancillary label on the bottle indicating
“
Shake Gently Before Use
”
.
10. Instruct the parent or caregiver that after the patient has completed the full course of therapy any
remaining solution must be discarded. It is recommended that this information be provided by
affixing an ancillary label to the bottle or adding a statement to the pharmacy label instructions.
11. Place an appropriate expiration date label according to storage condition (see below).
Storage of the pharmacy-compounded suspension (15 mg/ml)
Room temperature storage conditions: Stable for 3 weeks (21 days) when stored at room temperature
“do not store above 25 °C”.
Refrigerated storage conditions: Stable for 6 weeks when stored at 2 °C - 8 °C.
Place a pharmacy label on the bottle that includes the patient’s name, dosing instructions, use by date,
drug name and any other required information to be in compliance with local pharmacy regulations.
Refer to the table below for the proper dosing instructions.
Prophylaxis Dose
(for 10 days)
10 kg to 15 kg 30 mg 2 ml 2 ml twice daily 2 ml once daily
> 15 to 23 kg 45 mg 3 ml 3 ml twice daily 3 ml once daily
> 23 to 40 kg 60 mg 4 ml 4 ml twice daily 4 ml once daily
> 40 kg 75 mg 5 ml 5 ml twice daily 5 ml once daily
Note: This compounding procedure results in a 15 mg/ml suspension, which is different from the
commercially available Tamiflu powder for oral suspension.
Treatment Dose
(for 5 days)
Dispense the suspension with a graduated oral syringe for measuring small amounts of suspension. If
possible, mark or highlight the graduation corresponding to the appropriate dose (2 ml, 3 ml, 4 ml or
5 ml) on the oral syringe for each patient.
The appropriate dose must be mixed by the caregiver with an equal quantity of sweet liquid food, such
as sugar water, chocolate syrup, cherry syrup, dessert toppings (like caramel or fudge sauce) to mask
the bitter taste.
¾
Infants less than 1 year of age
This procedure describes the preparation of a 10 mg/ml solution that will provide one patient with
enough medication for a 5-day course of treatment or a 10-day course of prophylaxis.
The pharmacist may compound a suspension (10 mg/ml) from Tamiflu 30 mg, 45 mg or 75 mg
capsules using water containing 0.1% w/v sodium benzoate added as a preservative.
First, calculate the Total Volume needed to be compounded and dispensed for each patient. The Total
Volume required is determined by the weight of the patient according to the recommendation in the
table below:
Volume of Compounded Suspension (10 mg/ml) Prepared Based Upon th
e Patient’s Weight
Body Weight
(kg)
Total Volume to Compound per Patient Weight
(ml)
Dosing Chart for Pharmacy-Compounded Suspension from Tamiflu Capsules for Children One
Year of Age or Older
Body Weight
(kg)













Second, determine the number of capsules and the amount of vehicle (water containing 0.1% w/v
sodium benzoate added as a preservative) that is needed to prepare the Total Volume (calculated from
the table above: 30 ml, 45 ml) of compounded suspension (10 mg/ml) as shown in the table below:
Number of Capsules and Amount of Vehicle Needed to Prepare the Total Volume of a
Compounded Suspension (10 mg/ml)
Required Number of Tamiflu Capsules
(mg of oseltamivir)
Required Volume
of Vehicle
Please use
alternative capsule
strength*
* No integral number of capsules can be used to achieve the target concentration; therefore, please use
either the 30 mg or 75 mg capsules.
Third, follow the procedure below for compounding the suspension (10 mg/ml) from Tamiflu
capsules:
1. Carefully separate the capsule body and cap and transfer the contents of the required number of
Tamiflu capsules into a clean mortar.
2. Triturate the granules to a fine powder.
3. Add one-third (1/3) of the specified amount of vehicle and triturate the powder until a uniform
suspension is achieved.
4. Transfer the suspension to an amber glass or amber polyethyleneterephthalate (PET) bottle.
A funnel may be used to eliminate any spillage.
5. Add another one-third (1/3) of the vehicle to the mortar, rinse the pestle and mortar by a
triturating motion and transfer the vehicle into the bottle.
6. Repeat the rinsing (Step 5) with the remainder of the vehicle.
7. Close the bottle using a child-resistant cap.
8. Shake well to completely dissolve the active drug and to ensure homogeneous distribution of
the dissolved drug in the resulting suspension.
(Note: Undissolved residue may be visible but is comprised of inert ingredients of Tamiflu
capsules, which are insoluble. However, the active drug, oseltamivir phosphate, readily
dissolves in the specified vehicle and therefore forms a uniform solution.)
9. Put an ancillary label on the bottle indicating “Shake Gently Before Use”.
10. Instruct the parent or caregiver that after the patient has completed the full course of therapy any
remaining solution must be discarded. It is recommended that this information be provided by
affixing an ancillary label to the bottle or adding a statement to the pharmacy label instructions.
11. Place an appropriate expiration date label according to storage condition (see below).
Storage of the pharmacy-compounded suspension (10 mg/ml)
Room temperature storage conditions: Stable for 3 weeks (21 days) when stored at room temperature
“do not store above 25 °C”.
Refrigerated storage conditions: Stable for 6 weeks when stored at 2 °C - 8 °C.
Place a pharmacy label on the bottle that includes the patient’s name, dosing instructions, use by date,
drug name and any other required information to be in compliance with local pharmacy regulations.
Refer to the table below for the proper dosing instructions.
Total Volume
of Compounded
Suspension
to be Prepared










Dosing Chart for Pharmacy-Compounded Suspension (10 mg/ml) from Tamiflu Capsules for
Infants Less Than One Month of Age
Body Weight
(rounded to the
nearest 0.5 kg)
Treatment Dose
(for 5 days)
Prophylaxis Dose
(for 10 days)
Dosing Chart for Pharmacy-Compounded Suspension (10 mg/ml) from Tamiflu Capsules for
Infants One to Twelve Months of Age
Body Weight
(rounded to the
nearest 0.5 kg)
Treatment Dose
(for 5 days)
Prophylaxis Dose
(for 10 days)
Note: This compounding procedure results in a 10 mg/ml suspension, which is different from the
commercially available Tamiflu powder for oral suspension.
Dispense the suspension with a graduated oral syringe for measuring small amounts of suspension. If
possible, mark or highlight the graduation corresponding to the appropriate dose on the oral syringe
for each patient.
The appropriate dose must be mixed by the caregiver with an equal quantity of sweet liquid food, such
as sugar water, chocolate syrup, cherry syrup, dessert toppings (like caramel or fudge sauce) to mask
the bitter taste.
Home preparation
When commercially manufactured Tamiflu oral suspension is not available, a pharmacy preparation
from Tamiflu capsules can be used (detailed instructions above in section 4.2). If the pharmacy
preparation is not available either, Tamiflu doses may be prepared at home. The pharmacy preparation
is the preferred option in infants below 12 months of age.
When appropriate capsule strengths are available, the dose is given by opening the capsule and mixing
its contents with no more than one teaspoon of a suitable sweetened food product. The bitter taste can
be masked by products such as sugar water, chocolate syrup, cherry syrup, dessert toppings (like
caramel or fudge sauce). The mixture should be stirred and given entirely to the patient. The mixture
must be swallowed immediately after its preparation.
When only 75 mg capsules are available, and doses of 30 mg or 45 mg are needed, the preparation
involves additional steps. Detailed instructions can be found in section 3 in the package leaflet of
Tamiflu capsules.
Special populations
Hepatic impairment
No dose adjustment is required either for treatment or for prevention in patients with hepatic
dysfunction. No studies have been carried out in paediatric patients with hepatic disorder.




















Renal impairment
Treatment of influenza
: Dose adjustment is recommended for adults with severe renal impairment.
Recommended doses are detailed in the table below.
Creatinine clearance
Recommended dose for treatment
75 mg once daily,
or 30 mg suspension twice daily,
or 30 mg capsules twice daily
Prevention of influenza
: Dose adjustment is recommended for adults with severe renal impairment as
detailed in the table below.
Creatinine clearance
Recommended dose for prevention
75 mg every second day,
or 30 mg suspension once daily,
or 30 mg capsules once daily
Elderly
No dose adjustment is required, unless there is evidence of severe renal impairment.
Children
There is insufficient clinical data available in children with renal impairment to be able to make any
dosing recommendation.
Immunocompromised patients
Longer duration of seasonal prophylaxis up to 12 weeks has been evaluated in immunocompromised
subjects (see sections 4.4, 4.8 and 5.1).
Hypersensitivity to the active substance or to any of the excipients.
4.4 Special warnings and precautions for use
Oseltamivir is effective only against illness caused by influenza viruses. There is no evidence for
efficacy of oseltamivir in any illness caused by agents other than influenza viruses.
No information is available regarding the safety and efficacy of oseltamivir in patients with any
medical condition sufficiently severe or unstable to be considered at imminent risk of requiring
hospitalisation.
The efficacy of oseltamivir in either treatment or prophylaxis of influenza in immunocompromised
patients has not been firmly established (see section 5.1).
Efficacy of oseltamivir in the treatment of subjects with chronic cardiac disease and/or respiratory
disease has not been established. No difference in the incidence of complications was observed
between the treatment and placebo groups in this population (see section 5.1).
No data allowing a dose recommendation for premature children (< 37 weeks post-menstrual age*) are
currently available.
* Time between first day of last normal menstrual period and day of assessment, gestational age plus
post-natal age.














Tamiflu is not a substitute for influenza vaccination. Use of Tamiflu must not affect the evaluation of
individuals for annual influenza vaccination. The protection against influenza lasts only as long as
Tamiflu is administered. Tamiflu should be used for the treatment and prevention of influenza only
when reliable epidemiological data indicate that influenza virus is circulating in the community.
Susceptibility of circulating influenza virus strains to oseltamivir has been shown to be highly variable
(see section 5.1). Therefore, prescribers should take into account the most recent information available
on oseltamivir susceptibility patterns of the currently circulating viruses when deciding whether to use
Tamiflu.
Severe renal impairment
Dose adjustment is recommended for both treatment and prevention in adults with severe renal
insufficiency. There is insufficient clinical data available in children with renal impairment to be able
to make any dosing recommendation.(see sections 4.2 and 5.2).
Neuropsychiatric events have been reported during administration of Tamiflu in patients with
influenza, especially in children and adolescents. These events are also experienced by patients with
influenza without oseltamivir administration. Patients should be closely monitored for behavioural
changes, and the benefits and risks of continuing treatment should be carefully evaluated for each
patient (see section 4.8).
4.5 Interaction with other medicinal products and other forms of interaction
Pharmacokinetic properties of oseltamivir, such as low protein binding and metabolism independent of
the CYP450 and glucuronidase systems (see section 5.2), suggest that clinically significant drug
interactions via these mechanisms are unlikely.
No dose adjustment is required when co-administering with probenecid in patients with normal renal
function. Co-administration of probenecid, a potent inhibitor of the anionic pathway of renal tubular
secretion, results in an approximate 2-fold increase in exposure to the active metabolite of oseltamivir.
Oseltamivir has no kinetic interaction with amoxicillin, which is eliminated via the same pathway,
suggesting that oseltamivir interaction with this pathway is weak.
Clinically important drug interactions involving competition for renal tubular secretion are unlikely,
due to the known safety margin for most of these substances, the elimination characteristics of the
active metabolite (glomerular filtration and anionic tubular secretion) and the excretion capacity of
these pathways. However, care should be taken when prescribing oseltamivir in subjects when taking
co-excreted agents with a narrow therapeutic margin (e.g., chlorpropamide, methotrexate,
phenylbutazone).
No pharmacokinetic interactions between oseltamivir or its major metabolite have been observed
when co-administering oseltamivir with paracetamol, acetyl-salicylic acid, cimetidine or with antacids
(magnesium and aluminium hydroxides and calcium carbonates).
4.6 Pregnancy and lactation
While no controlled clinical trials have been conducted on the use of oseltamivir in pregnant women,
there is limited data available from post-marketing and retrospective observational surveillance
reports. These data in conjunction with animal studies do not indicate direct or indirect harmful effects
with respect to pregnancy, embryonal/foetal or postnatal development (see section 5.3). Pregnant
women may receive Tamiflu, after considering the available safety information, the pathogenicity of
the circulating influenza virus strain and the underlying condition of the pregnant woman.
In lactating rats, oseltamivir and the active metabolite are excreted in milk. Very limited information is
available on children breast-fed by mothers taking oseltamivir and on excretion of oseltamivir in
breast milk. Limited data demonstrated that oseltamivir and the active metabolite were detected in
breast milk, however the levels were low, which would result in a subtherapeutic dose to the infant.
Considering this information, the pathogenicity of the circulating influenza virus strain and the
underlying condition of the lactating woman, administration of oseltamivir may be considered, where
there are clear potential benefits to lactating mothers.
4.7 Effects on ability to drive and use machines
Tamiflu has no influence on the ability to drive and use machines.
The overall safety profile of Tamiflu is based on data from 2107 adult and 1032 paediatric patients
treated with Tamiflu or placebo for influenza, and on data from 2914 adult and 148 paediatric patients
receiving Tamiflu or placebo for the prophylaxis of influenza in clinical trials. In addition, 475
immunocompromised patients (including 18 children) received Tamiflu or placebo for the prophylaxis
of influenza.
In adults, the most commonly reported adverse drug reactions (ADRs) were vomiting and nausea in
the treatment studies, and nausea and headache in the prevention studies. The majority of these ADRs
were reported on a single occasion on either the first or second treatment day and resolved
spontaneously within 1-2 days. In children, the most commonly reported adverse drug reaction was
vomiting.
The ADRs listed in the tables below fall into the following categories: Very Common (≥
1/10
),
Common (≥
1/100
to <
1/10
), Uncommon (≥
1/1,000
to <
1/100
), Rare (≥
1/10,000
to <
1/1,000
), Very
rare (<
1/10,000
) and not known (cannot be estimated from the available data). ADRs are added to the
appropriate category in the tables according to the pooled analysis from clinical trials. Within each
frequency grouping ADRs are presented in the order of decreasing seriousness.
Treatment and prevention of influenza in adults and adolescents
:
Most Frequent Adverse Drug Reactions (
≥
1 % in the oseltamivir group) in Studies Investigating
Tamiflu for Treatment and Prevention of Influenza in Adults and Adolescents or Through Post-
Marketing Surveillance
System Organ Class (SOC)
Frequency Category
Adverse
Drug
Reaction
Percentage of Patients Experiencing the ADR
Treatment
Oseltamivir
75 mg bid
(n = 1057)
Oseltamivir
75 mg od
(n = 1480)
Infections and infestations
Upper respiratory tract infections
Disorders of the ear and labyrinth
Respiratory, thoracic and
mediastinal disorders












System Organ Class (SOC)
Frequency Category
Adverse
Drug
Reaction
Percentage of Patients Experiencing the ADR
Treatment
Oseltamivir
75 mg bid
(n = 1057)
Oseltamivir
75 mg od
(n = 1480)
Gastrointestinal disorders
Skin and subcutaneous tissue
disorders
Uncommon
:
Dermatitis
a
< 1 % < 1 % 1 % 1 %
Rash
a
< 1 % < 1 % < 1 % < 1 %
Urticaria
a
< 1 % < 1 % < 1 % < 1 %
Eczema
a
< 1 % 0 % < 1 % < 1 %
General disorders
Common
:
Dizziness 2 % 3 % 2 % 2 %
Fatigue 1 % 1 % 8 % 8 %
Pain < 1 % < 1 % 4 % 3 %
a
These are events identified during post-marketing surveillance. They were also reported in the
pooled clinical studies at the incidence presented in the table above.
b
Subjects who experienced nausea alone; excludes subjects who experienced nausea in association
with vomiting.
c
The difference between the placebo and oseltamivir groups was statistically significant.
Treatment and prevention of influenza in children
:
The table below shows the most frequently reported ADRs from paediatric clinical trials.
Most Frequent Adverse Drug Reactions (
≥
1 % in the oseltamivir group in the treatment studies
and
≥
10 % in the oseltamivir group in the prophylaxis study) in Children
Percentage of Patients Experiencing the ADR
Treatment
Oseltamivir
2 mg/kg bid
(n
=
515)
Oseltamivir
30 to 75 mg
b
(n
=
158)
Oseltamivir
30 to 75 mg
b
(n
=
148)
Infections and infestations
Disorders of the blood and
lymphatic system
Respiratory, thoracic and
mediastinal disorders
System Organ Class (SOC)
Frequency Category
Adverse Drug Reaction


















System Organ Class (SOC)
Frequency Category
Adverse Drug Reaction
Percentage of Patients Experiencing the ADR
Treatment
Oseltamivir
2 mg/kg bid
(n
=
515)
Oseltamivir
30 to 75 mg
b
(n
=
158)
Oseltamivir
30 to 75 mg
b
(n
=
148)
Asthma (incl. aggravated)
Gastrointestinal disorder
Disorders of the ear and labyrinth
Tympanic membrane disorder
Skin and subcutaneous tissue
disorders
a
The prevention study did not contain a placebo arm, i.e. was an uncontrolled study.
b
Unit dose = weight-based dosing (see section 4.2).
c
Patients experienced ear ache and ear pain.
In general, the adverse event profile in children with pre-existing bronchial asthma was qualitatively
similar to that of otherwise healthy children.
Further post marketing surveillance data on selected serious adverse drug reactions
:
Immune system disorders
Frequency not known: hypersensitivity reactions, including anaphylactic/anaphylactoid reactions.
Psychiatric disorders and nervous system disorders
Frequency not known: influenza can be associated with a variety of neurologic and behavioural
symptoms which can include events such as hallucinations, delirium, and abnormal behaviour, in some
cases resulting in fatal outcomes. These events may occur in the setting of encephalitis or
encephalopathy but can occur without obvious severe disease.
In patients with influenza who were receiving Tamiflu, there have been postmarketing reports of
convulsions and delirium (including symptoms such as altered level of consciousness, confusion,
abnormal behaviour, delusions, hallucinations, agitation, anxiety, nightmares), in a very few cases
resulting in self-injury or fatal outcomes. These events were reported primarily among pediatric and
adolescent patients and often had an abrupt onset and rapid resolution. The contribution of Tamiflu to
those events is unknown. Such neuropsychiatric events have also been reported in patients with
influenza who were not taking Tamiflu.
Eye disorders
Frequency not known: visual disturbance.
Cardiac disorders
Frequency not known: cardiac arrhythmia.














Gastrointestinal disorders
Frequency not known
:
gastrointestinal bleedings and hemorrhagic colitis.
Hepato-biliary disorders
Frequency not known: hepato-biliary system disorders, including hepatitis and elevated liver enzymes
in patients with influenza-like illness. These cases include fatal fulminant hepatitis/hepatic failure.
Skin and subcutaneous tissue disorders
Frequency not known: severe skin reactions, including Stevens-Johnson syndrome, toxic epidermal
necrolysis, erythema multiforme and angioneurotic oedema.
Additional information on special populations
:
Infants less than one year of age
Safety information available on oseltamivir administered for treatment of influenza in infants less than
one year of age from prospective and retrospective observational trials (comprising together more than
2400 infants of that age class), epidemiological databases research and postmarketing reports suggest
that the safety profile in infants less than one year of age is similar to the established safety profile of
children aged one year and older.
Elderly patients
There were no clinically relevant differences in the safety population of the elderly subjects who
received oseltamivir or placebo compared with the adult population aged up to 65 years.
Patients with chronic cardiac and/or respiratory disease
The adverse event profile in adolescents and patients with chronic cardiac and/or respiratory disease
was qualitatively similar to those of healthy young adults.
Immunocompromised patients
The adverse reactions noted in immunocompromised subjects 13 years of age and older who received
oseltamivir during 12 weeks for the seasonal prophylaxis of influenza were consistent with those
previously observed in Tamiflu clinical trials.
There is no experience with overdose. However, the anticipated manifestations of acute overdose
would be nausea, with or without accompanying vomiting, and dizziness. Patients should discontinue
the treatment in the event of overdose. No specific antidote is known.
PHARMACOLOGICAL PROPERTIES
5.1 Pharmacodynamic properties
Pharmacotherapeutic group: Antiviral ATC code: J05AH02
Oseltamivir phosphate is a pro-drug of the active metabolite (oseltamivir carboxylate). The active
metabolite is a selective inhibitor of influenza virus neuraminidase enzymes, which are glycoproteins
found on the virion surface. Viral neuraminidase enzyme activity is important both for viral entry into
uninfected cells and for the release of recently formed virus particles from infected cells, and for the
further spread of infectious virus in the body.
Oseltamivir carboxylate inhibits influenza A and B neuraminidases
in vitro
. Oseltamivir phosphate
inhibits influenza virus infection and replication
in vitro
. Oseltamivir given orally inhibits influenza A
and B virus replication and pathogenicity
in vivo
in animal models of influenza infection at antiviral
exposures similar to that achieved in man with 75 mg twice daily
.
Antiviral activity of oseltamivir was supported for influenza A and B by experimental challenge
studies in healthy volunteers.
Neuraminidase enzyme IC50 values for oseltamivir for clinically isolated influenza A ranged from
0.1 nM to 1.3 nM, and for influenza B was 2.6 nM. Higher IC50 values for influenza B, up to a
median of 8.5 nM, have been observed in published trials.
Reduced sensitivity of viral neuraminidase
Clinical studies
: The risk of emergence of influenza viruses with reduced susceptibility or frank
resistance to oseltamivir has been examined during Roche-sponsored clinical studies. All patients who
were found to carry oseltamivir-resistant virus did so transiently, cleared the virus normally and
showed no clinical deterioration.
Patients with Resistance Mutations (%)
* Full genotyping was not performed in all studies.
There has been no evidence for emergence of drug resistance associated with the use of Tamiflu in
clinical studies conducted to date in post-exposure (7 days), post-exposure within household groups
(10 days) and seasonal (42 days) prevention of influenza in immunocompetent subjects. There was no
resistance observed during a 12-week prophylaxis study in immunocompromised subjects.
Clinical and surveillance data
: Natural mutations associated with reduced susceptibility to oseltamivir
in vitro
have been detected in influenza A and B viruses isolated from patients without exposure to
oseltamivir. Resistant strains selected during oseltamivir treatment have been isolated from both
immunocompetent and immunocompromised patients. Immunocompromised patients and young
children are at a higher risk of developing oseltamivir-resistant virus during treatment.
Oseltamivir-resistant viruses isolated from oseltamivir-treated patients and oseltamivir-resistant
laboratory strains of influenza viruses have been found to contain mutations in N1 and N2
neuraminidases. Resistance mutations tend to be viral sub-type specific. Since 2007 resistance
associated H275Y mutation in seasonal H1N1 strains has become widespread. The susceptibility to
oseltamivir and the prevalence of such viruses appear to vary seasonally and geographically. In 2008,
H275Y was found in > 99 % of circulating H1N1 influenza isolates in Europe. The 2009 H1N1
influenza (“swine flu”) was almost uniformly susceptible to oseltamivir, with only sporadic reports of
resistance in connection with both therapeutic and prophylactic regimens.
Treatment of influenza infection
Oseltamivir is effective only against illnesses caused by influenza virus. Statistical analyses are
therefore presented only for influenza-infected subjects. In the pooled treatment study population,
which included both influenza-positive and -negative subjects (ITT), primary efficacy was reduced
proportional to the number of influenza-negative individuals. In the overall treatment population,
influenza infection was confirmed in 67 % (range 46 % to 74 %) of the recruited patients. Of the
elderly subjects, 64 % were influenza-positive and of those with chronic cardiac and/or respiratory
disease 62 % were influenza-positive. In all phase III treatment studies, patients were recruited only
during the period in which influenza was circulating in the local community.
Adults and adolescents 13 years of age and older
: Patients were eligible if they reported within
36 hours of onset of symptoms, had fever ≥ 37.8 °C, accompanied by at least one respiratory symptom
(cough, nasal symptoms or sore throat) and at least one systemic symptom (myalgia, chills/sweats,
malaise, fatigue or headache). In a pooled analysis of all influenza-positive adults and adolescents
(N = 2413) enrolled into treatment studies, oseltamivir 75 mg twice daily for 5 days reduced the
median duration of influenza illness by approximately one day from 5.2 days (95 % CI 4.9 – 5.5 days)
in the placebo group to 4.2 days (95 % CI 4.0 – 4.4 days; p
≤
0.0001).








The proportion of subjects who developed specified lower respiratory tract complications (mainly
bronchitis) treated with antibiotics was reduced from 12.7 % (135/1063) in the placebo group to 8.6 %
(116/1350) in the oseltamivir treated population (p = 0.0012).
Treatment of influenza in high risk populations
: The median duration of influenza illness in elderly
subjects (≥ 65 years) and in subjects with chronic cardiac and/or respiratory disease receiving
oseltamivir 75 mg twice daily for 5 days was
not
reduced significantly. The total duration of fever was
reduced by one day in the groups treated with oseltamivir. In the influenza-positive elderly,
oseltamivir significantly reduced the incidence of specified lower respiratory tract complications
(mainly bronchitis) treated with antibiotics from 19 % (52/268) in the placebo group to 12 % (29/250)
in the oseltamivir treated population (p = 0.0156).
In influenza-positive patients with chronic cardiac and/or respiratory disease, the combined incidence
of lower respiratory tract complications (mainly bronchitis) treated with antibiotics was 17 % (22/133)
in the placebo group and 14 % (16/118) in the oseltamivir treated population (p = 0.5976).
Treatment of influenza in children
: In a study of otherwise healthy children (65 % influenza-positive)
aged 1 to 12 years (mean age 5.3 years) who had fever (≥ 37.8 °C) plus either cough or coryza, 67 %
of
influenza-positive patients were infected with influenza A and 33 % with influenza B. Oseltamivir
treatment, started within 48 hours of onset of symptoms, significantly reduced the time to freedom
from illness (defined as the simultaneous return to normal health and activity and alleviation of fever,
cough and coryza) by 1.5 days (95 % CI 0.6 – 2.2 days; p < 0.0001) compared to placebo. Oseltamivir
reduced the incidence of acute otitis media from 26.5 % (53/200) in the placebo group to 16 %
(29/183) in the oseltamivir treated children (p = 0.013).
A second study was completed in 334 asthmatic children aged 6 to 12 years old of which 53.6 % were
influenza-positive. In the oseltamivir treated group, the median duration of illness was
not
reduced
significantly. By day 6 (the last day of treatment) FEV
1
had increased by 10.8 % in the oseltamivir
treated group compared to 4.7 % on placebo (p = 0.0148) in this population.
The European Medicines Agency has deferred the obligation to submit the results of studies with
Tamiflu in one or more subsets of the paediatric population in influenza. See section 4.2 for
information on paediatric use.
Treatment of influenza B infection
:
Overall, 15 % of the influenza-positive population were infected
by influenza B, proportions ranging from 1 to 33 % in individual studies. The median duration of
illness in influenza B infected subjects did not differ significantly between the treatment groups in
individual studies. Data from 504 influenza B infected subjects were pooled across all studies for
analysis. Oseltamivir reduced the time to alleviation of all symptoms by 0.7 days (95 % CI 0.1 – 1.6
days; p = 0.022) and the duration of fever (≥ 37.8 °C), cough and coryza by one day (95 % CI 0.4 –
1.7 days; p < 0.001) compared to placebo.
Prevention of influenza
The efficacy of oseltamivir in preventing naturally occurring influenza illness has been demonstrated
in a post-exposure prevention study in households and two seasonal prevention studies. The primary
efficacy parameter for all of these studies was the incidence of laboratory-confirmed influenza. The
virulence of influenza epidemics is not predictable and varies within a region and from season to
season, therefore the number needed to treat (NNT) in order to prevent one case of influenza illness
varies.
Post-exposure prevention
: In a study in contacts (12.6 % vaccinated against influenza) of an index
case of influenza, oseltamivir 75 mg once daily was started within 2 days of onset of symptoms in the
index case and continued for seven days. Influenza was confirmed in 163 out of 377 index cases.
Oseltamivir significantly reduced the incidence of clinical influenza illness occurring in the contacts of
confirmed influenza cases from 24/200 (12 %) in the placebo group to 2/205 (1 %) in the oseltamivir
group (92 % reduction [95 % CI 6 – 16; p
≤
0.0001]). The number needed to treat (NNT) in contacts
of true influenza cases was 10 (95 % CI 9 – 12) and was 16 (95 % CI 15 – 19) in the whole population
(ITT) regardless of infection status in the index case.
The efficacy of oseltamivir in preventing naturally occurring influenza illness has been demonstrated
in a post-exposure prevention study in households that included adults, adolescents, and children aged
1 to 12 years, both as index cases and as family contacts. The primary efficacy parameter for this study
was the incidence of laboratory-confirmed clinical influenza in the households. Oseltamivir
prophylaxis lasted for 10 days. In the total population, there was a reduction in the incidence of
laboratory-confirmed clinical influenza in households from 20 % (27/136) in the group not receiving
prevention to 7 % (10/135) in the group receiving prevention (62.7 % reduction [95 % CI 26.0 – 81.2;
p = 0.0042]). In households of influenza-infected index cases, there was a reduction in the incidence of
influenza from 26 % (23/89) in the group not receiving prevention to 11 % (9/84) in the group
receiving prevention (58.5 % reduction [95 % CI 15.6 – 79.6; p = 0.0114]).
According to subgroup analysis in children at 1 to 12 years of age, the incidence of laboratory-
confirmed clinical influenza among children was significantly reduced from 19 % (21/111) in the
group not receiving prevention to 7 % (7/104) in the group receiving prevention (64.4 % reduction
[95 % CI 15.8 – 85.0; p = 0.0188]). Among children who were not already shedding virus at baseline,
the incidence of laboratory-confirmed clinical influenza was reduced from 21 % (15/70) in the group
not receiving prevention to 4 % (2/47) in the group receiving prevention (80.1 % reduction [95 % CI
22.0 – 94.9; p = 0.0206]). The NNT for the total paediatric population was 9 (95 % CI 7 – 24) and
8 (95 % CI 6, upper limit not estimable) in the whole population (ITT) and in paediatric contacts of
infected index cases (ITTII), respectively.
Prevention during an influenza epidemic in the community
: In a pooled analysis of two other studies
conducted in unvaccinated otherwise healthy adults, oseltamivir 75 mg once daily given for 6 weeks
significantly reduced the incidence of clinical influenza illness from 25/519 (4.8 %) in the placebo
group to 6/520 (1.2 %) in the oseltamivir group (76 % reduction [95 % CI 1.6 – 5.7; p = 0.0006])
during a community outbreak of influenza. The NNT in this study was 28 (95 % CI 24 – 50).
A study in elderly residents of nursing homes, where 80 % of participants received vaccine in the
season of the study, oseltamivir 75 mg once daily given for 6 weeks significantly reduced the
incidence of clinical influenza illness from 12/272 (4.4 %) in the placebo group to 1/276 (0.4 %) in the
oseltamivir group (92 % reduction [95 % CI 1.5 – 6.6; p = 0.0015]). The NNT in this study was
25 (95 % CI 23 – 62).
Prophylaxis of influenza in immunocompromised patients
:
A double-blind, placebo-controlled,
randomised study was conducted for seasonal prophylaxis of influenza in 475 immunocompromised
subjects (388 subjects with solid organ transplantation [195 placebo; 193 oseltamivir], 87 subjects
with hemopoetic stem cell transplantation [43 placebo; 44 oseltamivir], no subject with other
immunosuppressant conditions), including 18 children 1 to 12 years of age. The primary endpoint in
this study was the incidence of laboratory-confirmed clinical influenza as determined by viral culture
and/or a four-fold rise in HAI antibodies. The incidence of laboratory-confirmed clinical influenza was
2.9 % (7/238) in the placebo group and 2.1 % (5/237) in the oseltamivir group (95 % CI -2.3 % –
4.1 %; p = 0.772).
Specific studies have not been conducted to assess of the reduction in the risk of complications.
5.2 Pharmacokinetic properties
Absorption
Oseltamivir is readily absorbed from the gastrointestinal tract after oral administration of oseltamivir
phosphate (pro-drug) and is extensively converted by predominantly hepatic esterases to the active
metabolite (oseltamivir carboxylate). At least 75 % of an oral dose reaches the systemic circulation as
the active metabolite. Exposure to the pro-drug is less than 5 % relative to the active metabolite.
Plasma concentrations of both pro-drug and active metabolite are proportional to dose and are
unaffected by co-administration with food.
Distribution
The mean volume of distribution at steady state of the oseltamivir carboxylate is approximately
23 litres in humans, a volume equivalent to extracellular body fluid. Since neuraminidase activity is
extracellular, oseltamivir carboxylate distributes to all sites of influenza virus spread.
The binding of the oseltamivir carboxylate to human plasma protein is negligible (approximately 3 %).
Metabolism
Oseltamivir is extensively converted to oseltamivir carboxylate by esterases located predominantly in
the liver.
In vitro
studies demonstrated that neither oseltamivir nor the active metabolite is a substrate
for, or an inhibitor of, the major cytochrome P450 isoforms. No phase 2 conjugates of either
compound have been identified
in vivo
.
Elimination
Absorbed oseltamivir is primarily (> 90 %) eliminated by conversion to oseltamivir carboxylate. It is
not further metabolised and is eliminated in the urine. Peak plasma concentrations of oseltamivir
carboxylate decline with a half-life of 6 to 10 hours in most subjects. The active metabolite is
eliminated entirely by renal excretion. Renal clearance (18.8 l/h) exceeds glomerular filtration rate
(7.5 l/h) indicating that tubular secretion occurs in addition to glomerular filtration. Less than 20 % of
an oral radiolabelled dose is eliminated in faeces.
Renal impairment
Administration of 100 mg oseltamivir phosphate twice daily for 5 days to patients with various
degrees of renal impairment showed that exposure to oseltamivir carboxylate is inversely proportional
to declining renal function. For dosing, see section 4.2.
Hepatic impairment
In vitro
studies have concluded that exposure to oseltamivir is not expected to be increased
significantly nor is exposure to the active metabolite expected to be significantly decreased in patients
with hepatic impairment (see section 4.2).
Elderly
Exposure to the active metabolite at steady state was 25 to 35 % higher in elderly (age 65 to 78 years)
compared to adults less than 65 years of age given comparable doses of oseltamivir. Half-lives
observed in the elderly were similar to those seen in young adults. On the basis of drug exposure and
tolerability, dosage adjustments are not required for elderly patients unless there is evidence of severe
renal impairment (creatinine clearance below 30 ml/min) (see section 4.2).
Children
Children 1 year of age and older
:
The pharmacokinetics of oseltamivir have been evaluated in single-
dose pharmacokinetic studies in children aged 1 to 16 years. Multiple-dose pharmacokinetics were
studied in a small number of children enrolled in a clinical efficacy study. Younger children cleared
both the pro-drug and its active metabolite faster than adults, resulting in a lower exposure for a
given mg/kg dose. Doses of 2 mg/kg give oseltamivir carboxylate exposures comparable to those
achieved in adults receiving a single 75 mg dose (approximately 1 mg/kg). The pharmacokinetics of
oseltamivir in children over 12 years of age are similar to those in adults.
Infants below 12 months of age
:
Limited pharmacokinetic and safety data are available for infants less
than 2 years of age. Pharmacokinetic modeling was undertaken using these data in addition to data
from studies in adults and children older than 1 year of age. The results demonstrate that doses of
3 mg/kg twice daily for infants aged 3 to 12 months and 2.5 mg/kg twice daily for infants aged
between 1 and 3 months provide exposures similar to those shown to be clinically efficacious in adults
and children > 1 year of age (see sections 4.1 and 4.2). There are currently no data available in infants
less than 1 month of age using Tamiflu.
5.3 Preclinical safety data
Preclinical data reveal no special hazard for humans based on conventional studies of safety
pharmacology, repeated-dose toxicity and genotoxicity. Results of the conventional rodent
carcinogenicity studies showed a trend towards a dose-dependent increase in the incidence of some
tumours that are typical for the rodent strains used. Considering the margins of exposure in relation to
the expected exposure in the human use, these findings do not change the benefit-risk of Tamiflu in its
adopted therapeutic indications.
Teratology studies have been conducted in rats and rabbits at doses of up to 1500 mg/kg/day and
500 mg/kg/day, respectively. No effects on foetal development were observed. A rat fertility study up
to a dose of 1500 mg/kg/day demonstrated no adverse effects on either sex. In pre- and post-natal rat
studies, prolonged parturition was noted at 1500 mg/kg/day: the safety margin between human
exposure and the highest no-effect dose (500 mg/kg/day) in rats is 480-fold for oseltamivir and 44-fold
for the active metabolite, respectively. Foetal exposure in the rats and rabbits was approximately
15 to 20 % of that of the mother.
In lactating rats, oseltamivir and the active metabolite are excreted in the milk. Limited data indicate
that oseltamivir and the active metabolite are excreted in human milk. Extrapolation of the animal data
provides estimates of 0.01 mg/day and 0.3 mg/day for the respective compounds.
A potential for skin sensitisation to oseltamivir was observed in a "maximisation" test in guinea pigs.
Approximately 50 % of the animals treated with the unformulated active ingredient showed erythema
after challenging the induced animals. Reversible irritancy of rabbits' eyes was detected.
Whereas very high oral single doses of oseltamivir phosphate salt, up to the highest dose tested
(1310 mg/kg), had no adverse effects in adult rats, such doses resulted in toxicity in juvenile 7-day-old
rat pups, including death. These effects were seen at doses of 657 mg/kg and higher. At 500 mg/kg, no
adverse effects were seen, including upon chronic treatment (500 mg/kg/day administered from 7 to
21 days post partum).
PHARMACEUTICAL PARTICULARS
Capsule core:
Pregelatinized starch (derived from maize starch)
Talc
Povidone
Croscarmellose sodium
Sodium stearyl fumarate
Capsule shell:
Gelatin
Yellow iron oxide (E172)
Red iron oxide (E172)
Black iron oxide (E172)
Titanium dioxide (E171)
Printing ink
:
Shellac
Titanium dioxide (E171)
FD and C Blue 2 (indigo carmine, E132)
6.4 Special precautions for storage
Do not store above 25 °C.
Storage of the pharmacy-compounded suspension
:
Room temperature storage conditions: Stable for 3 weeks (21 days) when stored at room temperature
“do not store above 25 °C”.
Refrigerated storage conditions: Stable for 6 weeks when stored at 2 °C - 8 °C.
6.5 Nature and contents of container
One box contains 10 capsules in a triplex blister pack (PVC/PE/PVDC, sealed with aluminium foil).
6.6 Special precautions for disposal
No special requirements.
Any unused product or waste material should be disposed of in accordance with local requirements.
Special instructions for use, handling and disposal of extemporaneous formulation prepared for
children less than one year of age
Extemporaneous formulation
When commercially manufactured Tamiflu powder for oral suspension is not available, patients who
are unable to swallow capsules may receive appropriate doses of Tamiflu prepared in a pharmacy or
prepared at home.
For infants below 12 months, the pharmacy preparation should be preferred to home preparation.
Detailed information on the pharmacy preparation can be found in section 4.2 and on the home
preparation can be found in section 3 of the package leaflet of Tamiflu capsules.
Syringes of appropriate volume and grading should be provided for administering the pharmacy
compounded suspension as well as for the procedures involved in the home preparation. In both cases,
the correct volumes should preferably be marked on the syringes.
MARKETING AUTHORISATION HOLDER
Roche Registration Limited
6 Falcon Way
Shire Park
Welwyn Garden City
AL7 1TW
United Kingdom
MARKETING AUTHORISATION NUMBER(S)
DATE OF FIRST AUTHORISATION/RENEWAL OF THE AUTHORISATION
Date of first authorisation: 20 June 2002
Date of last renewal: 20 June 2007
10. DATE OF REVISION OF THE TEXT
Detailed information on this medicinal product is available on the website of the European Medicines
Agency (EMEA) http://www.emea.europa.eu/.
NAME OF THE MEDICINAL PRODUCT
Tamiflu 12 mg/ml powder for oral suspension.
QUALITATIVE AND QUANTITATIVE COMPOSITION
1 g of powder for oral suspension contains oseltamivir phosphate equivalent to 30 mg of oseltamivir.
After reconstitution, each ml of suspension contains 12 mg oseltamivir.
One bottle of reconstituted suspension (75 ml) contains 900 mg of active substance (oseltamivir).
A bottle of 30 g Tamiflu powder for oral suspension contains 25.713 g of sorbitol.
30 mg oseltamivir suspension delivers 0.9 g of sorbitol.
45 mg oseltamivir suspension delivers 1.3 g of sorbitol.
60 mg oseltamivir suspension delivers 1.7 g of sorbitol.
75 mg oseltamivir suspension delivers 2.1 g of sorbitol.
For a full list of excipients, see section 6.1.
Powder for oral suspension
The powder is a granulate or clumped granulate with a white to light yellow colour.
4.1 Therapeutic indications
Treatment of influenza
In patients one year of age and older who present with symptoms typical of influenza, when influenza
virus is circulating in the community. Efficacy has been demonstrated when treatment is initiated
within two days of first onset of symptoms. This indication is based on clinical studies of naturally
occurring influenza in which the predominant infection was influenza A (see section 5.1).
Tamiflu is indicated for the treatment of infants below 12 months of age during a pandemic influenza
outbreak (see section 5.2).
Prevention of influenza
-
Post-exposure prevention in individuals one year of age or older following contact with a
clinically diagnosed influenza case when influenza virus is circulating in the community.
The appropriate use of Tamiflu for prevention of influenza should be determined on a case by
case basis by the circumstances and the population requiring protection. In exceptional
situations (e.g., in case of a mismatch between the circulating and vaccine virus strains, and a
pandemic situation) seasonal prevention could be considered in individuals one year of age or
older.
Tamiflu is indicated for post-exposure prevention of influenza in infants below 12 months of
age during a pandemic influenza outbreak (see section 5.2).
Tamiflu is not a substitute for influenza vaccination.
The use of antivirals for the treatment and prevention of influenza should be determined on the basis
of official recommendations. Decisions regarding the use of oseltamivir for treatment and prophylaxis
should take into consideration what is known about the characteristics of the circulating influenza
viruses, available information on influenza drug susceptibility patterns for each season and the impact
of the disease in different geographical areas and patient populations (see section 5.1).
Based on limited pharmacokinetic and safety data, Tamiflu can be used in infants below 12 months of
age for treatment during a pandemic influenza outbreak. The treating physician should take into
account the pathogenicity of the circulating strain and the underlying condition of the patient to ensure
there is a potential benefit to the child.
4.2 Posology and method of administration
Tamiflu suspension and Tamiflu capsules are bioequivalent formulations. 75 mg doses can be
administered as either
-
one 30 mg capsule plus one 45 mg capsule or
by administering one 30 mg dose plus one 45 mg dose of suspension.
Adults, adolescents or children (> 40 kg) who are able to
swallow capsules may receive appropriate
doses of Tamiflu capsules.
For infants below 12 months of age
: This formulation is not suitable for dosing in infants less than
12 months of age. For details, see sections below.
Treatment of influenza
Treatment should be initiated as soon as possible within the first two days of onset of symptoms of
influenza.
¾
For adolescents (13 to 17 years of age) and adults
: The recommended oral dose is 75 mg
oseltamivir twice daily for 5 days.
¾
For infants older than 1 year of age and for children 2 to 12 years of age
: The recommended dose
of Tamiflu oral suspension is indicated in the table below. Tamiflu 30 mg and 45 mg capsules are
available as an alternative to the recommended dose of Tamiflu suspension.
The following weight-adjusted dosing regimens are recommended for children 1 year of
age and older:
Body Weight
Recommended dose for 5 days
For dosing, an oral dispenser with 30 mg, 45 mg and 60 mg graduations is provided in the box. For
accurate dosing, the oral dispenser supplied should be used exclusively (a syringe with ml markings
cannot be used).
Children weighing > 40 kg and who are able to swallow capsules may receive treatment with the adult
dosage of 75 mg capsules twice daily for 5 days as an alternative to the recommended dose of Tamiflu
suspension.
¾
For infants below 1 year of age
: This formulation(Tamiflu 12 mg/ml powder for oral suspension) is
unsuitable since the syringe provided in the pack (with mg markings) does not allow for appropriate
dose adjustments and the use of syringes with ml markings may lead to unacceptable dosing
inaccuracies. In the absence of a suitable formulation, the pharmacy compounded preparation should
preferentially be used. Please refer to the SmPC for the 30 mg. 45 mg and 75 mg capsules (section
4.2).















Post-exposure prevention
¾
For adolescents (13 to 17 years of age) and adults
: The recommended dose for prevention of
influenza following close contact with an infected individual is 75 mg oseltamivir once daily for
10 days. Therapy should begin as soon as possible within two days of exposure to an infected
individual.
¾
For infants older than 1 year of age and for children 2 to 12 years of age
: Tamiflu 30 mg and 45 mg
capsules are available as an alternative to the recommended dose of Tamiflu suspension.
The recommended post-exposure prevention dose of Tamiflu is:
Body Weight
Recommended dose for 10 days
For dosing, an oral dispenser with 30 mg, 45 mg and 60 mg graduations is provided in the box. For
accurate dosing, the oral dispenser supplied should be used exclusively (a syringe with ml markings
cannot be used).
It is recommended that Tamiflu powder for oral suspension be constituted by a pharmacist prior to
dispensing to the patient (see section 6.6).
Children weighing > 40 kg and who are able to swallow capsules may receive prophylaxis with a
75 mg capsule once daily for 10 days as an alternative to the recommended dose of Tamiflu
suspension.
¾
For infants below below 1 year of age
: This formulation (Tamiflu 12 mg/ml powder for oral
suspension) is unsuitable since the syringe provided in the pack (with mg markings) does not allow for
appropriate dose adjustments and the use of syringes with ml markings may lead to unacceptable
dosing inaccuracies. In the absence of a suitable formulation, the pharmacy compounded preparation
should preferentially be used. Please refer to the SmPC for the 30 mg. 45 mg and 75 mg capsules
(section 4.2).
Prevention during an influenza epidemic in the community
The recommended dose for prevention of influenza during a community outbreak is 75 mg oseltamivir
once daily for up to 6 weeks.
Special populations
Hepatic impairment
No dose adjustment is required either for treatment or for prevention in patients with hepatic
dysfunction. No studies have been carried out in paediatric patients with hepatic disorder.
Renal impairment
Treatment of influenza
: Dose adjustment is recommended for adults with severe renal impairment.
Recommended doses are detailed in the table below.
Creatinine clearance
Recommended dose for treatment
75 mg once daily,
or 30 mg suspension twice daily,
or 30 mg capsules twice daily
Prevention of influenza
: Dose adjustment is recommended for adults with severe renal impairment as
detailed in the table below.

















Recommended dose for prevention
75 mg every second day,
or 30 mg suspension once daily,
or 30 mg capsules once daily
Elderly
No dose adjustment is required, unless there is evidence of severe renal impairment.
Children
There is insufficient clinical data available in children with renal impairment to be able to make any
dosing recommendation.
Immunocompromised patients
Longer duration of seasonal prophylaxis up to 12 weeks has been evaluated in immunocompromised
subjects (see sections 4.4, 4.8 and 5.1).
Hypersensitivity to the active substance or to any of the excipients.
4.4 Special warnings and precautions for use
Oseltamivir is effective only against illness caused by influenza viruses. There is no evidence for
efficacy of oseltamivir in any illness caused by agents other than influenza viruses.
No information is available regarding the safety and efficacy of oseltamivir in patients with any
medical condition sufficiently severe or unstable to be considered at imminent risk of requiring
hospitalisation.
The efficacy of oseltamivir in either treatment or prophylaxis of influenza in immunocompromised
patients has not been firmly established (see section 5.1).
Efficacy of oseltamivir in the treatment of subjects with chronic cardiac disease and/or respiratory
disease has not been established. No difference in the incidence of complications was observed
between the treatment and placebo groups in this population (see section 5.1).
No data allowing a dose recommendation for premature children (< 37 weeks post-menstrual age*) are
currently available.
* Time between first day of last normal menstrual period and day of assessment, gestational age plus
post-natal age.
Tamiflu is not a substitute for influenza vaccination
. Use of Tamiflu must not affect the evaluation of
individuals for annual influenza vaccination. The protection against influenza lasts only as long as
Tamiflu is administered. Tamiflu should be used for the treatment and prevention of influenza only
when reliable epidemiological data indicate that influenza virus is circulating in the community.
Susceptibility of circulating influenza virus strains to oseltamivir has been shown to be highly variable
(see section 5.1). Therefore, prescribers should take into account the most recent information available
on oseltamivir susceptibility patterns of the currently circulating viruses when deciding whether to use
Tamiflu.
Severe renal impairment
Dose adjustment is recommended for both treatment and prevention in adults with severe renal
insufficiency. There is insufficient clinical data available in children with renal impairment to be able
to make any dosing recommendation (see sections 4.2 and 5.2).








Neuropsychiatric events have been reported during administration of Tamiflu in patients with
influenza, especially in children and adolescents. These events are also experienced by patients with
influenza without oseltamivir administration. Patients should be closely monitored for behavioural
changes, and the benefits and risks of continuing treatment should be carefully evaluated for each
patient (see section 4.8).
This medicinal product contains sorbitol. Patients with rare hereditary problems of fructose intolerance
should not take this medicine.
Sorbitol can have a mild laxative effect.
4.5 Interaction with other medicinal products and other forms of interaction
Pharmacokinetic properties of oseltamivir, such as low protein binding and metabolism independent of
the CYP450 and glucuronidase systems (see section 5.2), suggest that clinically significant drug
interactions via these mechanisms are unlikely.
No dose adjustment is required when co-administering with probenecid in patients with normal renal
function. Co-administration of probenecid, a potent inhibitor of the anionic pathway of renal tubular
secretion, results in an approximate 2-fold increase in exposure to the active metabolite of oseltamivir.
Oseltamivir has no kinetic interaction with amoxicillin, which is eliminated via the same pathway,
suggesting that oseltamivir interaction with this pathway is weak.
Clinically important drug interactions involving competition for renal tubular secretion are unlikely,
due to the known safety margin for most of these substances, the elimination characteristics of the
active metabolite (glomerular filtration and anionic tubular secretion) and the excretion capacity of
these pathways. However, care should be taken when prescribing oseltamivir in subjects when taking
co-excreted agents with a narrow therapeutic margin (e.g., chlorpropamide, methotrexate,
phenylbutazone).
No pharmacokinetic interactions between oseltamivir or its major metabolite have been observed
when co-administering oseltamivir with paracetamol, acetyl-salicylic acid, cimetidine or with antacids
(magnesium and aluminium hydroxides and calcium carbonates).
4.6 Pregnancy and lactation
While no controlled clinical trials have been conducted on the use of oseltamivir in pregnant women,
there is limited data available from post-marketing and retrospective observational surveillance
reports. These data in conjunction with animal studies do not indicate direct or indirect harmful effects
with respect to pregnancy, embryonal/foetal or postnatal development (see section 5.3). Pregnant
women may receive Tamiflu, after considering the available safety information, the pathogenicity of
the circulating influenza virus strain and the underlying condition of the pregnant woman.
In lactating rats, oseltamivir and the active metabolite are excreted in milk. Very limited information is
available on children breast-fed by mothers taking oseltamivir and on excretion of oseltamivir in
breast milk. Limited data demonstrated that oseltamivir and the active metabolite were detected in
breast milk, however the levels were low, which would result in a subtherapeutic dose to the infant.
Considering this information, the pathogenicity of the circulating influenza virus strain and the
underlying condition of the lactating woman, administration of oseltamivir may be considered, where
there are clear potential benefits to lactating mothers.
4.7 Effects on ability to drive and use machines
Tamiflu has no influence on the ability to drive and use machines.
The overall safety profile of Tamiflu is based on data from 2107 adult and 1032 paediatric patients
treated with Tamiflu or placebo for influenza, and on data from 2914 adult and 148 paediatric patients
receiving Tamiflu or placebo for the prophylaxis of influenza in clinical trials. In addition, 475
immunocompromised patients (including 18 children) received Tamiflu or placebo for the prophylaxis
of influenza.
In adults, the most commonly reported adverse drug reactions (ADRs) were vomiting and nausea in
the treatment studies, and nausea and headache in the prevention studies. The majority of these ADRs
were reported on a single occasion on either the first or second treatment day and resolved
spontaneously within 1-2 days. In children, the most commonly reported adverse drug reaction was
vomiting.
The ADRs listed in the tables below fall into the following categories: Very Common (≥
1/10
),
Common (≥
1/100
to <
1/10
), Uncommon (≥
1/1,000
to <
1/100
), Rare (≥
1/10,000
to <
1/1,000
), Very
rare (<
1/10,000
) and not known (cannot be estimated from the available data). ADRs are added to the
appropriate category in the tables according to the pooled analysis from clinical trials. Within each
frequency grouping ADRs are presented in the order of decreasing seriousness.
Treatment and prevention of influenza in adults and adolescents
:
Most Frequent Adverse Drug Reactions (
≥
1 % in the oseltamivir group) in Studies Investigating
Tamiflu for Treatment and Prevention of Influenza in Adults and Adolescents or Through Post-
Marketing Surveillance
System Organ Class (SOC)
Frequency Category
Adverse
Drug
Reaction
Percentage of Patients Experiencing the ADR
Treatment
Oseltamivir
75 mg bid
(n = 1057)
Oseltamivir
75 mg od
(n = 1480)
Infections and infestations
Upper respiratory tract infections
Disorders of the ear and labyrinth
Respiratory, thoracic and
mediastinal disorders
Gastrointestinal disorders
Skin and subcutaneous tissue













System Organ Class (SOC)
Frequency Category
Adverse
Drug
Reaction
Percentage of Patients Experiencing the ADR
Treatment
Oseltamivir
75 mg bid
(n = 1057)
Oseltamivir
75 mg od
(n = 1480)
disorders
Uncommon
:
Dermatitis
a
< 1 % < 1 % 1 % 1 %
Rash
a
< 1 % < 1 % < 1 % < 1 %
Urticaria
a
< 1 % < 1 % < 1 % < 1 %
Eczema
a
< 1 % 0 % < 1 % < 1 %
General disorders
Common
:
Dizziness 2 % 3 % 2 % 2 %
Fatigue 1 % 1 % 8 % 8 %
Pain < 1 % < 1 % 4 % 3 %
a
These are events identified during post-marketing surveillance. They were also reported in the
pooled clinical studies the incidence presented in the table above.
b
.Subjects who experienced nausea alone; excludes subjects who experienced nausea in association
with vomiting.
c
The difference between the placebo and oseltamivir groups was statistically significant.
Treatment and prevention of influenza in children
:
The table below shows the most frequently reported ADRs from paediatric clinical trials.
Most Frequent Adverse Drug Reactions (
≥
1 % in the oseltamivir group in the treatment studies
and
≥
10 % in the oseltamivir group in the prophylaxis study) in Children
Percentage of Patients Experiencing the ADR
Treatment
Oseltamivir
2 mg/kg bid
(n
=
515)
Oseltamivir
30 to 75 mg
b
(n
=
158)
Oseltamivir
30 to 75 mg
b
(n
=
148)
Infections and infestations
Disorders of the blood and
lymphatic system
Respiratory, thoracic and
mediastinal disorders
Asthma (incl. aggravated)
Gastrointestinal disorder
System Organ Class (SOC)
Frequency Category
Adverse Drug Reaction





















System Organ Class (SOC)
Frequency Category
Adverse Drug Reaction
Percentage of Patients Experiencing the ADR
Treatment
Oseltamivir
2 mg/kg bid
(n
=
515)
Oseltamivir
30 to 75 mg
b
(n
=
158)
Oseltamivir
30 to 75 mg
b
(n
=
148)
Disorders of the ear and labyrinth
Tympanic membrane disorder
Skin and subcutaneous tissue
disorders
a
The prevention study did not contain a placebo arm, i.e. was an uncontrolled study.
b
Unit dose = weight-based dosing (see section 4.2).
c
Patients experienced ear ache and ear pain.
In general, the adverse event profile in children with pre-existing bronchial asthma was qualitatively
similar to that of otherwise healthy children.
Further post marketing surveillance data on selected serious adverse drug reactions
:
Immune system disorders
Frequency not known: hypersensitivity reactions, including anaphylactic/anaphylactoid reactions.
Psychiatric disorders and nervous system disorders
Frequency not known: influenza can be associated with a variety of neurologic and behavioural
symptoms which can include events such as hallucinations, delirium, and abnormal behaviour, in some
cases resulting in fatal outcomes. These events may occur in the setting of encephalitis or
encephalopathy but can occur without obvious severe disease.
In patients with influenza who were receiving Tamiflu, there have been postmarketing reports of
convulsions and delirium (including symptoms such as altered level of consciousness, confusion,
abnormal behaviour, delusions, hallucinations, agitation, anxiety, nightmares), in a very few cases
resulting in self-injury or fatal outcomes. These events were reported primarily among pediatric and
adolescent patients and often had an abrupt onset and rapid resolution. The contribution of Tamiflu to
those events is unknown. Such neuropsychiatric events have also been reported in patients with
influenza who were not taking Tamiflu.
Eye disorders
Frequency not known: visual disturbance.
Cardiac disorders
Frequency not known: cardiac arrhythmia.
Gastrointestinal disorders
Frequency not known
:
gastrointestinal bleedings and hemorrhagic colitis.
Hepato-biliary disorders
Frequency not known: hepato-biliary system disorders, including hepatitis and elevated liver enzymes
in patients with influenza-like illness. These cases include fatal fulminant hepatitis/hepatic failure.
Skin and subcutaneous tissue disorders













Frequency not known: severe skin reactions, including Stevens-Johnson syndrome, toxic epidermal
necrolysis, erythema multiforme and angioneurotic oedema.
Additional information on special populations
:
Infants less than one year of age
Safety information available on oseltamivir administered for treatment of influenza in infants less than
one year of age from prospective and retrospective observational trials (comprising together more than
2400 infants of that age class), epidemiological databases research and postmarketing reports suggest
that the safety profile in infants less than one year of age is similar to the established safety profile of
children aged one year and older.
Elderly patients
There were no clinically relevant differences in the safety population of the elderly subjects who
received oseltamivir or placebo compared with the adult population aged up to 65 years.
Patients with chronic cardiac and/or respiratory disease
The adverse event profile in adolescents and patients with chronic cardiac and/or respiratory disease
was qualitatively similar to those of healthy young adults.
Immunocompromised patients
The adverse reactions noted in immunocompromised subjects 13 years of age and older who received
oseltamivir during 12 weeks for the seasonal prophylaxis of influenza were consistent with those
previously observed in Tamiflu clinical trials.
There is no experience with overdose. However, the anticipated manifestations of acute overdose
would be nausea, with or without accompanying vomiting, and dizziness. Patients should discontinue
the treatment in the event of overdose. No specific antidote is known.
PHARMACOLOGICAL PROPERTIES
5.1 Pharmacodynamic properties
Pharmacotherapeutic group: Antiviral ATC code: J05AH02
Oseltamivir phosphate is a pro-drug of the active metabolite (oseltamivir carboxylate). The active
metabolite is a selective inhibitor of influenza virus neuraminidase enzymes, which are glycoproteins
found on the virion surface. Viral neuraminidase enzyme activity is important both for viral entry into
uninfected cells and for the release of recently formed virus particles from infected cells, and for the
further spread of infectious virus in the body.
Oseltamivir carboxylate inhibits influenza A and B neuraminidases
in vitro
. Oseltamivir phosphate
inhibits influenza virus infection and replication
in vitro
. Oseltamivir given orally inhibits influenza A
and B virus replication and pathogenicity
in vivo
in animal models of influenza infection at antiviral
exposures similar to that achieved in man with 75 mg twice daily
.
Antiviral activity of oseltamivir was supported for influenza A and B by experimental challenge
studies in healthy volunteers.
Neuraminidase enzyme IC50 values for oseltamivir for clinically isolated influenza A ranged from
0.1 nM to 1.3 nM, and for influenza B was 2.6 nM. Higher IC50 values for influenza B, up to a
median of 8.5 nM, have been observed in published trials.
Reduced sensitivity of viral neuraminidase
Clinical studies
: The risk of emergence of influenza viruses with reduced susceptibility or frank
resistance to oseltamivir has been examined during Roche-sponsored clinical studies. All patients who
were found to carry oseltamivir-resistant virus did so transiently, cleared the virus normally and
showed no clinical deterioration.
Patients with Resistance Mutations (%)
* Full genotyping was not performed in all studies.
There has been no evidence for emergence of drug resistance associated with the use of Tamiflu in
clinical studies conducted to date in post-exposure (7 days), post-exposure within household groups
(10 days) and seasonal (42 days) prevention of influenza in immunocompetent subjects. There was no
resistance observed during a 12-week prophylaxis study in immunocompromised subjects.
Clinical and surveillance data
: Natural mutations associated with reduced susceptibility to oseltamivir
in vitro
have been detected in influenza A and B viruses isolated from patients without exposure to
oseltamivir. Resistant strains selected during oseltamivir treatment have been isolated from both
immunocompetent and immunocompromised patients. Immunocompromised patients and young
children are at a higher risk of developing oseltamivir-resistant virus during treatment.
Oseltamivir-resistant viruses isolated from oseltamivir-treated patients and oseltamivir-resistant
laboratory strains of influenza viruses have been found to contain mutations in N1 and N2
neuraminidases. Resistance mutations tend to be viral sub-type specific. Since 2007 resistance
associated H275Y mutation in seasonal H1N1 strains has become widespread. The susceptibility to
oseltamivir and the prevalence of such viruses appear to vary seasonally and geographically. In 2008,
H275Y was found in > 99 % of circulating H1N1 influenza isolates in Europe. The 2009 H1N1
influenza (“swine flu”) was almost uniformly susceptible to oseltamivir, with only sporadic reports of
resistance in connection with both therapeutic and prophylactic regimens.
Treatment of influenza infection
Oseltamivir is effective only against illnesses caused by influenza virus. Statistical analyses are
therefore presented only for influenza-infected subjects. In the pooled treatment study population,
which included both influenza-positive and -negative subjects (ITT), primary efficacy was reduced
proportional to the number of influenza-negative individuals. In the overall treatment population,
influenza infection was confirmed in 67 % (range 46 % to 74 %) of the recruited patients. Of the
elderly subjects, 64 % were influenza-positive and of those with chronic cardiac and/or respiratory
disease 62 % were influenza-positive. In all phase III treatment studies, patients were recruited only
during the period in which influenza was circulating in the local community.
Adults and adolescents 13 years of age and older
: Patients were eligible if they reported within
36 hours of onset of symptoms, had fever ≥ 37.8 °C, accompanied by at least one respiratory symptom
(cough, nasal symptoms or sore throat) and at least one systemic symptom (myalgia, chills/sweats,
malaise, fatigue or headache). In a pooled analysis of all influenza-positive adults and adolescents
(N = 2413) enrolled into treatment studies, oseltamivir 75 mg twice daily for 5 days reduced the
median duration of influenza illness by approximately one day from 5.2 days (95 % CI 4.9 – 5.5 days)
in the placebo group to 4.2 days (95 % CI 4.0 – 4.4 days; p ≤ 0.0001).
The proportion of subjects who developed specified lower respiratory tract complications (mainly
bronchitis) treated with antibiotics was reduced from 12.7 % (135/1063) in the placebo group to 8.6 %
(116/1350) in the oseltamivir treated population (p = 0.0012).
Treatment of influenza in high risk populations
: The median duration of influenza illness in elderly
subjects (≥ 65 years) and in subjects with chronic cardiac and/or respiratory disease receiving
oseltamivir 75 mg twice daily for 5 days was
not
reduced significantly. The total duration of fever was
reduced by one day in the groups treated with oseltamivir. In the influenza-positive elderly,








oseltamivir significantly reduced the incidence of specified lower respiratory tract complications
(mainly bronchitis) treated with antibiotics from 19 % (52/268) in the placebo group to 12 % (29/250)
in the oseltamivir treated population (p = 0.0156).
In influenza-positive patients with chronic cardiac and/or respiratory disease, the combined incidence
of lower respiratory tract complications (mainly bronchitis) treated with antibiotics was 17 % (22/133)
in the placebo group and 14 % (16/118) in the oseltamivir treated population (p = 0.5976).
Treatment of influenza in children
: In a study of otherwise healthy children (65 % influenza-positive)
aged 1 to 12 years (mean age 5.3 years) who had fever (≥ 37.8 °C) plus either cough or coryza, 67 %
of
influenza-positive patients were infected with influenza A and 33 % with influenza B. Oseltamivir
treatment, started within 48 hours of onset of symptoms, significantly reduced the duration of time to
freedom from illness (defined as the simultaneous return to normal health and activity and alleviation
of fever, cough and coryza) by 1.5 days (95 % CI 0.6 – 2.2 days; p < 0.0001) compared to placebo.
Oseltamivir reduced the incidence of acute otitis media from 26.5 % (53/200) in the placebo group to
16 % (29/183) in the oseltamivir treated children (p = 0.013).
A second study was completed in 334 asthmatic children aged 6 to 12 years old of which 53.6 % were
influenza-positive. In the oseltamivir treated group, the median duration of illness was
not
reduced
significantly. By day 6 (the last day of treatment) FEV
1
had increased by 10.8 % in the oseltamivir
treated group compared to 4.7 % on placebo (p = 0.0148) in this population.
The European Medicines Agency has deferred the obligation to submit the results of studies with
Tamiflu in one or more subsets of the paediatric population in influenza. See section 4.2 for
information on paediatric use.
Treatment of influenza B infection
: Overall, 15 % of the influenza-positive population were infected by
influenza B, proportions ranging from 1 to 33 % in individual studies. The median duration of illness
in influenza B infected subjects did not differ significantly between the treatment groups in individual
studies. Data from 504 influenza B infected subjects were pooled across all studies for analysis.
Oseltamivir reduced the time to alleviation of all symptoms by 0.7 days (95 % CI 0.1 – 1.6 days;
p = 0.022) and the duration of fever (≥ 37.8 °C), cough and coryza by one day (95 % CI 0.4 –
1.7 days; p < 0.001) compared to placebo.
Prevention of influenza
The efficacy of oseltamivir in preventing naturally occurring influenza illness has been demonstrated
in a post-exposure prevention study in households and two seasonal prevention studies. The primary
efficacy parameter for all of these studies was the incidence of laboratory-confirmed influenza. The
virulence of influenza epidemics is not predictable and varies within a region and from season to
season, therefore the number needed to treat (NNT) in order to prevent one case of influenza illness
varies.
Post-exposure prevention
: In a study in contacts (12.6 % vaccinated against influenza) of an index
case of influenza, oseltamivir 75 mg once daily was started within 2 days of onset of symptoms in the
index case and continued for seven days. Influenza was confirmed in 163 out of 377 index cases.
Oseltamivir significantly reduced the incidence of clinical influenza illness occurring in the contacts of
confirmed influenza cases from 24/200 (12 %) in the placebo group to 2/205 (1 %) in the oseltamivir
group (92 % reduction [95 % CI 6 – 16; p ≤ 0.0001]). The number needed to treat (NNT) in contacts
of true influenza cases was 10 (95 % CI 9 – 12) and was 16 (95 % CI 15 – 19) in the whole population
(ITT) regardless of infection status in the index case.
The efficacy of oseltamivir in preventing naturally occurring influenza illness has been demonstrated
in a post-exposure prevention study in households that included adults, adolescents, and children aged
1 to 12 years, both as index cases and as family contacts. The primary efficacy parameter for this study
was the incidence of laboratory-confirmed clinical influenza in the households. Oseltamivir
prophylaxis lasted for 10 days. In the total population, there was a reduction in the incidence of
laboratory-confirmed clinical influenza in households from 20 % (27/136) in the group not receiving
prevention to 7 % (10/135) in the group receiving prevention (62.7 % reduction [95 % CI 26.0 – 81.2;
p = 0.0042]). In households of influenza-infected index cases, there was a reduction in the incidence of
influenza from 26 % (23/89) in the group not receiving prevention to 11 % (9/84) in the group
receiving prevention (58.5 % reduction [95 % CI 15.6 – 79.6; p = 0.0114]).
According to subgroup analysis in children at 1 to 12 years of age, the incidence of laboratory-
confirmed clinical influenza among children was significantly reduced from 19 % (21/111) in the
group not receiving prevention to 7 % (7/104) in the group receiving prevention (64.4 % reduction
[95 % CI 15.8 – 85.0; p = 0.0188]). Among children who were not already shedding virus at baseline,
the incidence of laboratory-confirmed clinical influenza was reduced from 21 % (15/70) in the group
not receiving prevention to 4 % (2/47) in the group receiving prevention (80.1 % reduction [95 % CI
22.0 – 94.9; p = 0.0206]). The NNT for the total paediatric population was 9 (95 % CI 7 – 24) and
8 (95 % CI 6, upper limit not estimable) in the whole population (ITT) and in paediatric contacts of
infected index cases (ITTII), respectively.
Prevention during an influenza epidemic in the community
: In a pooled analysis of two other studies
conducted in unvaccinated otherwise healthy adults, oseltamivir 75 mg once daily given for 6 weeks
significantly reduced the incidence of clinical influenza illness from 25/519 (4.8 %) in the placebo
group to 6/520 (1.2 %) in the oseltamivir group (76 % reduction [95 % CI 1.6 – 5.7; p = 0.0006])
during a community outbreak of influenza. The NNT in this study was 28 (95 % CI 24 – 50).
A study in elderly residents of nursing homes, where 80 % of participants received vaccine in the
season of the study, oseltamivir 75 mg once daily given for 6 weeks significantly reduced the
incidence of clinical influenza illness from 12/272 (4.4 %) in the placebo group to 1/276 (0.4 %) in the
oseltamivir group (92 % reduction [95 % CI 1.5 – 6.6; p = 0.0015]). The NNT in this study was
25 (95 % CI 23 – 62).
Prophylaxis of influenza in immunocompromised patients
:
A double-blind, placebo-controlled,
randomised study was conducted for seasonal prophylaxis of influenza in 475 immunocompromised
subjects (388 subjects with solid organ transplantation [195 placebo; 193 oseltamivir], 87 subjects
with hemopoetic stem cell transplantation [43 placebo; 44 oseltamivir], no subject with other
immunosuppressant conditions), including 18 children 1 to 12 years of age. The primary endpoint in
this study was the incidence of laboratory-confirmed clinical influenza as determined by viral culture
and/or a four-fold rise in HAI antibodies. The incidence of laboratory-confirmed clinical influenza was
2.9 % (7/238) in the placebo group and 2.1 % (5/237) in the oseltamivir group (95 % CI -2.3 % –
4.1 %; p = 0.772).
Specific studies have not been conducted to assess of the reduction in the risk of complications.
5.2 Pharmacokinetic properties
Absorption
Oseltamivir is readily absorbed from the gastrointestinal tract after oral administration of oseltamivir
phosphate (pro-drug) and is extensively converted by predominantly hepatic esterases to the active
metabolite (oseltamivir carboxylate). At least 75 % of an oral dose reaches the systemic circulation as
the active metabolite. Exposure to the pro-drug is less than 5 % relative to the active metabolite.
Plasma concentrations of both pro-drug and active metabolite are proportional to dose and are
unaffected by co-administration with food.
Distribution
The mean volume of distribution at steady state of the oseltamivir carboxylate is approximately
23 litres in humans, a volume equivalent to extracellular body fluid. Since neuraminidase activity is
extracellular, oseltamivir carboxylate distributes to all sites of influenza virus spread.
The binding of the oseltamivir carboxylate to human plasma protein is negligible (approximately 3 %).
Metabolism
Oseltamivir is extensively converted to oseltamivir carboxylate by esterases located predominantly in
the liver.
In vitro
studies demonstrated that neither oseltamivir nor the active metabolite is a substrate
for, or an inhibitor of, the major cytochrome P450 isoforms. No phase 2 conjugates of either
compound have been identified
in vivo
.
Elimination
Absorbed oseltamivir is primarily (> 90 %) eliminated by conversion to oseltamivir carboxylate. It is
not further metabolised and is eliminated in the urine. Peak plasma concentrations of oseltamivir
carboxylate decline with a half-life of 6 to 10 hours in most subjects. The active metabolite is
eliminated entirely by renal excretion. Renal clearance (18.8 l/h) exceeds glomerular filtration rate
(7.5 l/h) indicating that tubular secretion occurs in addition to glomerular filtration. Less than 20 % of
an oral radiolabelled dose is eliminated in faeces.
Renal impairment
Administration of 100 mg oseltamivir phosphate twice daily for 5 days to patients with various
degrees of renal impairment showed that exposure to oseltamivir carboxylate is inversely proportional
to declining renal function. For dosing, see section 4.2.
Hepatic impairment
In vitro
studies have concluded that exposure to oseltamivir is not expected to be increased
significantly nor is exposure to the active metabolite expected to be significantly decreased in patients
with hepatic impairment (see section 4.2).
Elderly
Exposure to the active metabolite at steady state was 25 to 35 % higher in elderly (age 65 to 78 years)
compared to adults less than 65 years of age given comparable doses of oseltamivir. Half-lives
observed in the elderly were similar to those seen in young adults. On the basis of drug exposure and
tolerability, dosage adjustments are not required for elderly patients unless there is evidence of severe
renal impairment (creatinine clearance below 30 ml/min) (see section 4.2).
Children
Children 1 year of age and older
: The pharmacokinetics of oseltamivir have been evaluated in single-
dose pharmacokinetic studies in children aged 1 to 16 years. Multiple-dose pharmacokinetics were
studied in a small number of children enrolled in a clinical efficacy study. Younger children cleared
both the pro-drug and its active metabolite faster than adults, resulting in a lower exposure for a
given mg/kg dose. Doses of 2 mg/kg give oseltamivir carboxylate exposures comparable to those
achieved in adults receiving a single 75 mg dose (approximately 1 mg/kg). The pharmacokinetics of
oseltamivir in children over 12 years of age are similar to those in adults.
Infants below 12 months of age
:
Limited pharmacokinetic and safety data are available for infants less
than 2 years of age. Pharmacokinetic modeling was undertaken using these data in addition to data
from studies in adults and children older than 1 year of age. The results demonstrate that doses of
3 mg/kg twice daily for infants aged 3 to 12 months and 2.5 mg/kg twice daily for infants aged
between 1 and 3 months provide exposures similar to those shown to be clinically efficacious in adults
and children > 1 year of age (see sections 4.1 and 4.2). There are currently no data available in infants
less than 1 month of age using Tamiflu.
5.3 Preclinical safety data
Preclinical data reveal no special hazard for humans based on conventional studies of safety
pharmacology, repeated-dose toxicity and genotoxicity. Results of the conventional rodent
carcinogenicity studies showed a trend towards a dose-dependent increase in the incidence of some
tumours that are typical for the rodent strains used. Considering the margins of exposure in relation to
the expected exposure in the human use, these findings do not change the benefit-risk of Tamiflu in its
adopted therapeutic indications.
Teratology studies have been conducted in rats and rabbits at doses of up to 1500 mg/kg/day and
500 mg/kg/day, respectively. No effects on foetal development were observed. A rat fertility study up
to a dose of 1500 mg/kg/day demonstrated no adverse effects on either sex. In pre- and post-natal rat
studies, prolonged parturition was noted at 1500 mg/kg/day: the safety margin between human
exposure and the highest no-effect dose (500 mg/kg/day) in rats is 480-fold for oseltamivir and 44-fold
for the active metabolite, respectively. Foetal exposure in the rats and rabbits was approximately 15 to
20 % of that of the mother.
In lactating rats, oseltamivir and the active metabolite are excreted in the milk. Limited data indicate
that oseltamivir and the active metabolite are excreted in human milk. Extrapolation of the animal data
provides estimates of 0.01 mg/day and 0.3 mg/day for the respective compounds.
A potential for skin sensitisation to oseltamivir was observed in a "maximisation" test in guinea pigs.
Approximately 50 % of the animals treated with the unformulated active ingredient showed erythema
after challenging the induced animals. Reversible irritancy of rabbits' eyes was detected.
Whereas very high oral single doses of oseltamivir phosphate salt, up to the highest dose tested
(1310 mg/kg), had no adverse effects in adult rats, such doses resulted in toxicity in juvenile 7-day-old
rat pups, including death. These effects were seen at doses of 657 mg/kg and higher. At 500 mg/kg, no
adverse effects were seen, including upon chronic treatment (500 mg/kg/day administered from 7 to
21 days post partum).
PHARMACEUTICAL PARTICULARS
Powder for oral suspension:
Sorbitol (E420),
Sodium dihydrogen citrate (E331[a])
Xanthan gum (E415)
Sodium benzoate (E211)
Saccharin sodium (E954)
Titanium dioxide (E171)
Tutti frutti flavour (including maltodextrins [maize], propylene glycol, arabic gum E414 and natural
identical flavouring substances [mainly consisting of banana, pineapple and peach flavour]).
2 years for powder for oral suspension.
6.4 Special precautions for storage
Powder: Do not store above 30°C.
After reconstitution, the suspension can be stored either at room temperature (not above 25°C) for 10
days or in a refrigerator (2°C - 8°C ) for 17 days.
6.5 Nature and contents of container
Carton containing a 100 ml amber glass bottle (with child-resistant plastic screw cap) with 30 g of
powder for oral suspension, a plastic adapter, a plastic oral dispenser and a plastic measuring cup.
After reconstitution with 52 ml of water, the usable volume of oral suspension allows for the retrieval
of a total of 10 doses of 75 mg oseltamivir.
6.6 Special precautions for disposal and other handling
It is recommended that Tamiflu oral suspension should be reconstituted by the pharmacist prior to its
dispensing to the patient.
Only the syringe included in the package with doses indicated in mg should be used. It cannot be
replaced by a syringe with ml markings.
Preparation of Oral Suspension
1.
Measure 52 ml of water by filling the measuring cup to the indicated level (measuring cup
included in the box).
Add all 52 ml of water into the bottle, recap the bottle and shake the closed bottle well for 15
seconds.
Remove the cap and push the bottle adapter into the neck of the bottle.
Close the bottle tightly with the cap (on the top of the bottle adapter). This will make sure that
the bottle adapter fits in the bottle in the right position.
Tamiflu powder for suspension will appear as an opaque and white to light yellow suspension after
reconstitution.
Any unused product or waste material should be disposed of in accordance with local requirements.
MARKETING AUTHORISATION HOLDER
Roche Registration Limited
6 Falcon Way
Shire Park
Welwyn Garden City
AL7 1TW
United Kingdom
MARKETING AUTHORISATION NUMBER(S)
DATE OF FIRST AUTHORISATION/RENEWAL OF THE AUTHORISATION
Date of first authorisation: 20 June 2002
Date of last renewal: 20 June 2007
10. DATE OF REVISION OF THE TEXT
Detailed information on this medicinal product is available on the website of the European Medicines
Agency (EMEA) http://www.emea.europa.eu/.
Tap the closed bottle gently several times to loosen the powder.
A. MANUFACTURING AUTHORISATION HOLDER RESPONSIBLE FOR
BATCH RELEASE
B. CONDITIONS OF THE MARKETING AUTHORISATION
MANUFACTURING AUTHORISATION HOLDER(S) RESPONSIBLE FOR BATCH
RELEASE
Name and address of the manufacturer responsible for batch release
Roche Pharma AG
Emil-Barell-Str. 1
D- 79639 Grenzach –Wyhlen
Germany
CONDITIONS OF THE MARKETING AUTHORISATION
CONDITIONS OR RESTRICTIONS REGARDING SUPPLY AND USE IMPOSED ON
THE MARKETING AUTHORISATION HOLDER
Medicinal product subject to medical prescription
CONDITIONS OR RESTRICTIONS WITH REGARD TO THE SAFE AND
EFFECTIVE USE OF THE MEDICINAL PRODUCT
The MAH must ensure that the system of pharmacovigilance, as described in version 4.1 presented in
Module 1.8.1. of the Marketing Authorisation, is in place and functioning before and whilst the
product is on the market.
The MAH commits to performing the studies and additional pharmacovigilance activities detailed in
the Pharmacovigilance Plan, as agreed in version 6.0 of the Risk management Plan (RMP) presented
in Module 1.8.2. of the Marketing Authorisation and any subsequent updates of the RMP agreed by
the CHMP.
As per the CHMP Guideline on Risk Management Systems for medicinal products for human use, any
updated RMP should be submitted at the same time as the following Periodic Safety Update Report
(PSUR).
In addition, an updated RMP should be submitted
•
When new information is received that may impact on the current Safety Specification,
Pharmacovigilance Plan or risk minimisation activities.
Within 60 days of an important (pharmacovigilance or risk minimisation) milestone being
reached.
At the request of the EMEA.
The MAH will continue to submit yearly PSURs (Data Lock Point 21 September) unless otherwise
specified by the CHMP.
ANNEX III
LABELLING AND PACKAGE LEAFLET
PARTICULARS TO APPEAR ON THE OUTER PACKAGING
NAME OF THE MEDICINAL PRODUCT
Tamiflu 30 mg hard capsules
Oseltamivir
STATEMENT OF ACTIVE SUBSTANCE(S)
Each hard capsule contains oseltamivir phosphate equivalent to 30 mg of oseltamivir.
PHARMACEUTICAL FORM AND CONTENTS
METHOD AND ROUTE(S) OF ADMINISTRATION
Oral use
Read package leaflet before use
SPECIAL WARNING THAT THE MEDICINAL PRODUCT MUST BE STORED OUT
OF THE REACH AND SIGHT OF CHILDREN
Keep out of the reach and sight of children
OTHER SPECIAL WARNING(S), IF NECESSARY
SPECIAL STORAGE CONDITIONS
10. SPECIAL PRECAUTIONS FOR DISPOSAL OF UNUSED MEDICINAL PRODUCTS
OR WASTE MATERIALS DERIVED FROM SUCH MEDICINAL PRODUCTS, IF
APPROPRIATE

























11. NAME AND ADDRESS OF THE MARKETING AUTHORISATION HOLDER
Roche Registration Limited
6 Falcon Way
Shire Park
Welwyn Garden City
AL7 1TW
United Kingdom
12. MARKETING AUTHORISATION NUMBER(S)
14. GENERAL CLASSIFICATION FOR SUPPLY
Medicinal product subject to medical prescription
16. INFORMATION IN BRAILLE















PARTICULARS TO APPEAR ON THE OUTER PACKAGING
NAME OF THE MEDICINAL PRODUCT
Tamiflu 45 mg hard capsules
Oseltamivir
STATEMENT OF ACTIVE SUBSTANCE(S)
Each hard capsule contains oseltamivir phosphate equivalent to 45 mg of oseltamivir.
PHARMACEUTICAL FORM AND CONTENTS
METHOD AND ROUTE(S) OF ADMINISTRATION
Oral use
Read package leaflet before use
SPECIAL WARNING THAT THE MEDICINAL PRODUCT MUST BE STORED OUT
OF THE REACH AND SIGHT OF CHILDREN
Keep out of the reach and sight of children
OTHER SPECIAL WARNING(S), IF NECESSARY
SPECIAL STORAGE CONDITIONS
10. SPECIAL PRECAUTIONS FOR DISPOSAL OF UNUSED MEDICINAL PRODUCTS
OR WASTE MATERIALS DERIVED FROM SUCH MEDICINAL PRODUCTS, IF
APPROPRIATE

























11. NAME AND ADDRESS OF THE MARKETING AUTHORISATION HOLDER
Roche Registration Limited
6 Falcon Way
Shire Park
Welwyn Garden City
AL7 1TW
United Kingdom
12. MARKETING AUTHORISATION NUMBER(S)
14. GENERAL CLASSIFICATION FOR SUPPLY
Medicinal product subject to medical prescription
16. INFORMATION IN BRAILLE















PARTICULARS TO APPEAR ON THE OUTER PACKAGING
NAME OF THE MEDICINAL PRODUCT
Tamiflu 75 mg hard capsules
Oseltamivir
STATEMENT OF ACTIVE SUBSTANCE(S)
Each hard capsule contains oseltamivir phosphate equivalent to 75 mg of oseltamivir.
PHARMACEUTICAL FORM AND CONTENTS
METHOD AND ROUTE(S) OF ADMINISTRATION
Oral use
Read package leaflet before use
SPECIAL WARNING THAT THE MEDICINAL PRODUCT MUST BE STORED OUT
OF THE REACH AND SIGHT OF CHILDREN
Keep out of the reach and sight of children
OTHER SPECIAL WARNING(S), IF NECESSARY
SPECIAL STORAGE CONDITIONS
10. SPECIAL PRECAUTIONS FOR DISPOSAL OF UNUSED MEDICINAL PRODUCTS
OR WASTE MATERIALS DERIVED FROM SUCH MEDICINAL PRODUCTS, IF
APPROPRIATE

























11. NAME AND ADDRESS OF THE MARKETING AUTHORISATION HOLDER
Roche Registration Limited
6 Falcon Way
Shire Park
Welwyn Garden City
AL7 1TW
United Kingdom
12. MARKETING AUTHORISATION NUMBER(S)
14. GENERAL CLASSIFICATION FOR SUPPLY
Medicinal product subject to medical prescription
16. INFORMATION IN BRAILLE















PARTICULARS TO APPEAR ON THE OUTER PACKAGING
NAME OF THE MEDICINAL PRODUCT
Tamiflu 12 mg/ml powder for oral suspension
Oseltamivir
STATEMENT OF ACTIVE SUBSTANCE(S)
1 bottle with 30 g powder for oral suspension contains 900 mg of oseltamivir. Final volume of the
bottle after reconstitution with 52 ml of water is 75 ml. Each ml of suspension contains 12 mg
oseltamivir.
Also contains sorbitol and sodium benzoate.
PHARMACEUTICAL FORM AND CONTENTS
1 bottle
Also contains 1 plastic bottle adapter, 1 plastic measuring cup (52 ml) and 1 plastic oral dispenser
METHOD AND ROUTE(S) OF ADMINISTRATION
For oral administration after constitution
Shake bottle well before use
Read the enclosed leaflet before use and also for the method of preparation
SPECIAL WARNING THAT THE MEDICINAL PRODUCT MUST BE STORED OUT
OF THE REACH AND SIGHT OF CHILDREN
Keep out of the reach and sight of children
OTHER SPECIAL WARNING(S), IF NECESSARY
SPECIAL STORAGE CONDITIONS
Powder: Do not store above 30°C
After reconstitution, the suspension can be stored either at room temperature (not above 25°C) for 10
days or in a refrigerator (2°C - 8°C ) for 17 days



























10. SPECIAL PRECAUTIONS FOR DISPOSAL OF UNUSED MEDICINAL PRODUCTS
OR WASTE MATERIALS DERIVED FROM SUCH MEDICINAL PRODUCTS, IF
APPROPRIATE
11. NAME AND ADDRESS OF THE MARKETING AUTHORISATION HOLDER
Roche Registration Limited
6 Falcon Way
Shire Park
Welwyn Garden City
AL7 1TW
United Kingdom
12. MARKETING AUTHORISATION NUMBER(S)
14. GENERAL CLASSIFICATION FOR SUPPLY
Medicinal product subject to medical prescription
16. INFORMATION IN BRAILLE

















MINIMUM PARTICULARS TO APPEAR ON SMALL IMMEDIATE PACKAGING UNITS
NAME OF THE MEDICINAL PRODUCT
Tamiflu 12 mg/ml powder for oral suspension
Oseltamivir
For oral administration after constitution
Shake bottle well before use
Read the enclosed leaflet before use and also for the method of preparation
CONTENTS BY WEIGHT, BY VOLUME OR BY UNIT
Also contains sorbitol and sodium benzoate.
SPECIAL WARNING THAT THE MEDICINAL PRODUCT MUST BE STORED OUT
OF THE REACH AND SIGHT OF CHILDREN
Keep out of the reach and sight of children
SPECIAL STORAGE CONDITIONS
Powder: Do not store above 30°C
After reconstitution, the suspension can be stored either at room temperature (not above 25°C) for 10
days or in a refrigerator (2°C - 8°C ) for 17 days























NAME AND ADDRESS OF THE MARKETING AUTHORISATION HOLDER
Roche Registration Limited
6 Falcon Way
Shire Park
Welwyn Garden City
AL7 1TW
United Kingdom



PACKAGE LEAFLET: INFORMATION FOR THE USER
Tamiflu 30 mg hard capsules
oseltamivir
Read all of this leaflet carefully before you start taking this medicine.
-
If you have further questions, ask your doctor or pharmacist.
This medicine has been prescribed for you. Do not pass it on to others. It may harm them, even
if their symptoms are the same as yours.
If any of the side effects gets serious, or if you notice any side effects not listed in this leaflet,
please tell your doctor or pharmacist.
What Tamiflu is and what it is used for
WHAT TAMIFLU IS AND WHAT IT IS USED FOR
Tamiflu belongs to a group of medicines named “neuraminidase inhibitors”. These medicines
prevent the influenza virus from spreading inside the body and so help to ease or prevent the
symptoms arising from the influenza virus infection.
Influenza is an infection caused by the influenza virus. The signs (symptoms) of influenza
include the sudden onset of fever (more than 37.8 °C), cough, runny or stuffy nose, headaches,
muscle aches and often extreme fatigue. These symptoms may also be caused by infections
other than influenza. True influenza infection only occurs during annual outbreaks (epidemics)
at times when influenza viruses are spreading in the local community. Outside of the epidemic
period, these symptoms will mainly be caused by a different type of infection or illness.
if you are allergic (hypersensitive) to oseltamivir or any of the other ingredients of Tamiflu.
Take special care with Tamiflu
Before you take Tamiflu, make sure your prescribing doctor knows if you
-
have problems with your kidneys.
Taking other medicines
Please tell your doctor or pharmacist if you are taking or have recently taken any other medicines,
including medicines obtained without a prescription.
Tamiflu can be taken with appropriate medicines to treat fever (high temperature). Tamiflu is not
expected to alter the effect of any other medicines.
Keep this leaflet. You may need to read it again.
Tamiflu is prescribed to you for treating or preventing influenza.
are allergic to other medicines
Are there any interactions with an influenza vaccination?
Tamiflu is not a substitute for influenza vaccination
. Tamiflu will not change the effectiveness of
the influenza vaccine. Even if a vaccination against influenza has been given to you Tamiflu may be
prescribed by your doctor.
Taking Tamiflu with food and drink
Swallow Tamiflu with water. Tamiflu can be taken with or without food, although it is recommended
to take Tamiflu with food to reduce the chance of feeling or being sick (nausea or vomiting).
Pregnancy and breast-feeding
Ask your doctor or pharmacist for advice before taking any medicine.
You must tell your doctor if you are pregnant, if you think you are pregnant or if you are trying to get
pregnant so that your doctor can decide if Tamiflu is right for you.
The effects on nursing infants are unknown. You must tell your doctor if you are breast-feeding so that
your doctor can decide if Tamiflu is right for you.
Driving and using machines
Tamiflu has no effect on your ability to drive or use machines.
Always take Tamiflu exactly as your doctor has told you. You should check with your doctor or
pharmacist if you are not sure.
Take Tamiflu as soon as you get the prescription as this will help to slow the spread of the influenza
virus in the body.
Swallow Tamiflu capsules whole with water. Do not break or chew Tamiflu capsules.
The usual dose is as follows:
Treatment
¾
Adolescents (13 to 17 years of age) and adults
:
30 mg and 45 mg capsules can be used by adults and adolescents as an alternative to 75 mg capsules.
For treatment of influenza take one 30 mg and one 45 mg capsule as soon as you get the prescription
and then take one 30 mg and one 45 mg capsule twice a day (usually it is convenient to take one
30 mg and one 45 mg in the morning and one 30 mg and one 45 mg in the evening for five days). It is
important to complete the whole 5 day course, even if you start to feel better quickly.
¾
Infants 1 year of age and older, and children 2 to 12 years of age:
Tamiflu oral suspension can be
used instead of capsules.
Children weighing more than 40 kg and who can swallow capsules may take a 75 mg dose of Tamiflu
capsules twice daily for 5 days.
You must give the amount of capsules to your child as prescribed by your doctor.
The usual dose for treatment of influenza in children depends on the body weight of the child (see
table below).



Children 1 year of age and older
:
Body Weight
Recommended dose for 5 days (Treatment)
Less than or equal to 15 kg
More than 15 kg and up to 23 kg
More than 23 kg and up to 40 kg
75 mg twice daily
(75 mg is made up of a 30 mg dose plus a 45 mg dose)
¾
Infants below 12 months of age
:
Limited pharmacokinetic and safety data are available for infants
less than 2 years of age. Pharmacokinetic modeling was undertaken using these data in addition to data
from studies in adults and children older than 1 year of age. The results demonstrate that doses of
3 mg/kg twice daily for infants aged 3 to 12 months and 2.5 mg/kg twice daily for infants aged
between 1 and 3 months provide exposures similar to those shown to be clinically efficacious in adults
and children older than 1 year of age (see table below for weight-based dosing recommendations).
There are currently no data available in patients less than 1 month of age using Tamiflu.
Recommended dose for 5 days (Treatment)
Administration of Tamiflu to infants less than one year of age should be based upon the judgment of
the physician after considering the potential benefit of treatment versus any potential risk to the infant.
Prevention
Tamiflu can also be used for prevention of influenza following exposure to an infected individual,
such as family members.
¾
Adolescents (13 to 17 years of age) and adults
:
30 mg and 45 mg capsules can be used by adults and adolescents as an alternative to 75 mg capsules.
Tamiflu should be taken once daily for 10 days. It is best to take this dose in the mornings with
breakfast.
¾
Infants 1 year of age and older, and children 2 to 12 years of age:
Tamiflu oral suspension can be
used instead of capsules.
The usual dose for prevention of influenza in children depends on the body weight of the child (see
table below).
Children 1 year of age and older
:
Body Weight
Recommended dose for 10 days (Prevention)
Less than or equal to 15 kg
More than 15 kg and up to 23 kg
More than 23 kg and up to 40 kg
75 mg once daily
(75 mg is made up of a 30 mg dose plus a 45 mg dose)
Your doctor will recommend the length of time to continue taking Tamiflu, if it is prescribed to
prevent influenza.
¾
Infants below 12 months of age
: The recommended prophylaxis dose for infants less than
12 months of age during a pandemic influenza outbreak is half of the daily treatment dose. This is
based upon clinical data in children older than 1 year of age and adults showing that a prophylaxis
dose equivalent to half the daily treatment dose is clinically efficacious for the prevention of influenza.
The following weight-adjusted dosing prophylaxis regimens are recommended for infants below
1 year of age:


























Recommended dose for 10 days (Prevention)
Administration of Tamiflu to infants less than one year of age should be based upon the judgment of
the physician after considering the potential benefit of prophylaxis versus any potential risk to the
infant.
When Tamiflu oral suspension is not available
When commercially manufactured Tamiflu oral suspension is not available, a pharmacy preparation
from Tamiflu capsules can be used (see end of package leaflet, information for medical or healthcare
personnel). If the pharmacy preparation is not available either, Tamiflu doses may be prepared at
home. The pharmacy preparation is the preferred option in infants below 12 months of age.
When appropriate capsule strengths are available, the dose is given by opening the capsule and mixing
its contents with no more than one teaspoon of a suitable sweetened food product. The bitter taste can
be masked by products such as sugar water, chocolate syrup, cherry syrup, dessert toppings (like
caramel or fudge sauce). The mixture should be stirred and given entirely to the patient. The mixture
must be swallowed immediately after its preparation.
When only 75 mg capsules are available, and doses of 30 mg or 45 mg are needed, the preparation
involves additional steps.
Preparing Tamiflu doses for infants younger than 1 year — 30 mg capsules
Children who weigh 10 kg or less need a Tamiflu dose of 30 mg or less. To make doses of Tamiflu for
these children, you will need to open a Tamiflu capsule, prepare a powder-water mixture and mix the
correct volume of the mixture with food.
One 30 mg Tamiflu capsule
Dosing chart to determine the correct dosage amount (see Tamiflu dosing chart under step 3 in
the next section below)
Two small bowls per child (use separate pairs of bowls for each child)
One 3 ml (three millilitre) oral syringe that shows measurements of 0.1 ml (one-tenth of one
millilitre)
One suitable oral dosing syringe that shows measurements of 0.1 ml (one-tenth of one
millilitre). Ask the pharmacist for the correct syringe (1 ml, 2 ml, or 3 ml).
One of these foods to hide the bitter taste of the Tamiflu powder:
-
Sugar water
(If you plan to use sugar water, make this mixture now in a separate bowl or
cup. Mix a teaspoon of water with three-fourths [3/4] of a teaspoon of sugar.)
Dessert toppings, like caramel or fudge sauce
Making the Tamiflu 10 mg/ml mixture for infants younger than 1 year — 30 mg capsules
These instructions show you how to make one dose of Tamiflu, to be used immediately.
1.
Carefully open ONE 30 mg Tamiflu capsule over the first small bowl. To open a capsule, hold
the capsule upright and snip off the rounded tip with scissors (see accompanying picture). Pour
all of the powder into the bowl. Handle the powder carefully, because it may be irritating to the
skin and eyes.







Follow the chart below. Use the 3 ml dosing syringe to measure 3.0 ml of water, and add it to
the powder in the bowl.
Stir for about 2 minutes
.
3.0 ml (three millilitres)
Follow the chart below. Find the child’s weight on the left side of the chart, and then look at the
column on the right side of chart to see the amount of the Tamiflu powder-water mixture to
draw up into the dosing syringe. Draw up the correct amount of powder-water mixture from the
first small bowl into the dosing syringe, and then gently expel it from the dosing syringe into the
second bowl.
Tamiflu dosing chart for infants 1 to 12 months of age:
Weight
(rounded to the
nearest 0.5 kg)
Amount of Tamiflu powder-water mixture
to be drawn up into the dosing syringe
Tamiflu dosing chart for infants below 1 month of age:
Weight
(rounded to the
nearest 0.5 kg)
Amount of Tamiflu powder-water mixture
to be drawn up into the dosing syringe
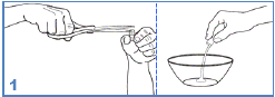

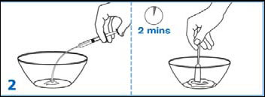


























Add a small amount of one of the sweet foods (use no more than 1 teaspoon) into the second
bowl. This will hide the bitter taste of the powder-water mixture.
Mix the sweet food and powder-water mixture well.
Give the entire contents of the second bowl (sweet food with powder-water mixture) to the
child.
Throw out any unused powder-water mixture left in the first bowl.
(Note: Not all capsule contents may dissolve. Do not worry, as the undissolved materials are inactive
ingredients.)
If you take more Tamiflu, than you should
Contact your doctor or pharmacist immediately.
If you have forgotten to take Tamiflu
Do not take a double dose to make up for a forgotten capsule.
If you stop taking Tamiflu
There are no side effects when Tamiflu is discontinued prior to advice from your doctor. If Tamiflu is
stopped earlier than your doctor told you, the symptoms of influenza may reoccur.
If you have any further questions on the use of this product, ask your doctor or pharmacist.
Like all medicines, Tamiflu can cause side effects, although not everybody gets them.
Common side effects of Tamiflu
The most common side effects of Tamiflu are nausea, vomiting, diarrhoea, stomach ache and
headache. These side effects mostly occur only after the first dose of the medicine and will usually
stop as treatment continues. The frequency of these effects is reduced if the medicinal product is taken
with food.
Give the child something suitable to drink.

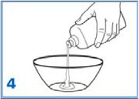

Less common side effects of Tamiflu
During Tamiflu treatment, events like convulsions and delirium (including symptoms such as altered
level of consciousness, confusion, abnormal behaviour, delusions, hallucinations, agitation, anxiety,
nightmares) have been reported, in a very few cases resulting in self-injury, in some instances with
fatal outcome. These events were reported primarily among children and adolescents and often had an
abrupt onset and rapid resolution. Such neuropsychiatric events have also been reported in patients
with influenza who were not taking Tamiflu.
Adults and adolescents (children aged 13 years and older)
Other less common side effects, which may also be caused by influenza, are upper abdominal fullness,
bleeding in the gastrointestinal tract, bronchitis, upper respiratory tract infections, dizziness, tiredness,
sleeping difficulties, skin reactions, mild to severe liver function disorders, visual disturbances and
heart rhythm abnormalities.
Children (aged 1 to 12 years)
Other less common side effects, which may also be caused by influenza, are cough, nasal congestion,
ear inflammation, inflammation of the lungs, sinusitis, bronchitis, aggravation of pre-existing asthma,
nose bleeding, ear disorders, inflammation of the skin, swelling of the lymph nodes, conjunctivitis,
visual disturbances and heart rhythm abnormalities.
Infants (aged 6 to 12 months)
The reported side effects of Tamiflu when used for treatment of influenza in infants 6 to 12 months of
age are similar to the side effects reported for older children (1 year and older). Please read the section
above for your information.
Infants (aged 0 to 6 months)
The reported side effects of Tamiflu when used for treatment of influenza in infants 1 to 6 months of
age are similar to the side effects reported for infants 6 to 12 months of age and older children (1 year
and older). There are no data available for the use of Tamiflu in infants less than 1 month of age.
Please read the section above for your information.
If you or your child are often sick, you should inform your doctor. You should also tell your doctor
if the influenza symptoms get worse or the fever continues.
If any of the side effects get serious, or if you notice any side effects not listed in this leaflet, tell your
doctor or pharmacist.
Keep out of the reach and sight of children.
Do not use Tamiflu after the expiry date which is stated on the carton and blister after EXP. The
expiry date refers to the last day of that month.
Do not store above 25 °C.
Medicines should not be disposed of via wastewater or household waste. Ask your pharmacist how to
dispose of medicines no longer required. These measures will help to protect the environment.



The active substance is oseltamivir phosphate (30 mg oseltamivir in each hard capsule).
The other ingredients are:
capsule contents: pregelatinised starch, talc, povidone, croscarmellose sodium and sodium
stearyl fumarate
capsule shell: gelatin, yellow iron oxide (E172), red iron oxide (E172) and titanium dioxide
(E171)
printing ink: shellac (E904), titanium dioxide (E171) and FD and C Blue 2 (indigocarmine,
E132).
What Tamiflu looks like and contents of the pack
30 mg hard capsule consists of a light yellow opaque body bearing the imprint “ROCHE” and a light
yellow opaque cap bearing the imprint “30 mg”. Imprints are blue.
Tamiflu 30 mg hard capsules are available in blister packs of 10.
Marketing Authorisation Holder and Manufacturer
Roche Registration Limited
6 Falcon Way
Shire Park
Welwyn Garden City
AL7 1TW
United Kingdom
Roche Pharma AG
Emil-Barell-Str. 1,
D-79639 Grenzach-Wyhlen
Germany
For any information about this medicine, please contact the local representative of the Marketing
Authorisation Holder.
België/Belgique/Belgien
N.V. Roche S.A.
Tél/Tel: +32 (0) 2 525 82 11
Luxembourg/Luxemburg
(Voir/siehe Belgique/Belgien)
България
Рош България ЕООД
Тел: +359 2 818 44 44
Magyarország
Roche (Magyarország) Kft.
Tel: +36 - 23 446 800
Česká republika
Roche s. r. o.
Tel: +420 - 2 20382111
Malta
(See United Kingdom)
Danmark
Roche a/s
Tlf: +45 - 36 39 99 99
Nederland
Roche Nederland B.V.
Tel: +31 (0) 348 438050
Deutschland
Roche Pharma AG
Tel: +49 (0) 7624 140
Norge
Roche Norge AS
Tlf: +47 - 22 78 90 00
Eesti
Roche Eesti OÜ
Tel: + 372 - 6 177 380
Österreich
Roche Austria GmbH
Tel: +43 (0) 1 27739
Ελλάδα
Roche (Hellas) A.E.
Τηλ: +30 210 61 66 100
Polska
Roche Polska Sp.z o.o.
Tel: +48 - 22 345 18 88
España
Roche Farma S.A.
Tel: +34 - 91 324 81 00
Portugal
Roche Farmacêutica Química, Lda
Tel: +351 - 21 425 70 00
France
Roche
Tél: +33 (0) 1 46 40 50 00
România
Roche România S.R.L.
Tel: +40 21 206 47 01
Ireland
Roche Products (Ireland) Ltd.
Tel: +353 (0) 1 469 0700
Slovenija
Roche farmacevtska družba d.o.o.
Tel: +386 - 1 360 26 00
Ísland
Roche a/s
c/o Icepharma hf
Sími: +354 540 8000
Slovenská republika
Roche Slovensko, s.r.o.
Tel: +421 - 2 52638201
Italia
Roche S.p.A.
Tel: +39 - 039 2471
Suomi/Finland
Roche Oy
Puh/Tel: +358 (0) 10 554 500
Kύπρος
Γ.Α.Σταμάτης & Σια Λτδ.
Τηλ: +357 - 22 76 62 76
Sverige
Roche AB
Tel: +46 (0) 8 726 1200
Latvija
Roche Latvija SIA
Tel: +371 - 6 7039831
United Kingdom
Roche Products Ltd.
Tel: +44 (0) 1707 366000
Lietuva
UAB “Roche Lietuva”
Tel: +370 5 2546799
This leaflet was last approved in {MM/YYYY}.
Detailed information on this medicine is available on the European Medicines Agency (EMEA) web
site: http://www.emea.europa.eu/.
---------------------------------------------------------------------------------------------------------------------------
The following information is intended for medical or healthcare professionals only:
When commercially manufactured Tamiflu powder for oral suspension is not available, patients who
are unable to swallow capsules may receive appropriate doses of Tamiflu prepared in a pharmacy or
prepared at home.
For infants below 12 months, the pharmacy preparation should be preferred to home preparation.
Detailed information on the pharmacy preparation can be found in the SmPC of Tamiflu capsules
section 4.2.
Syringes of appropriate volume and grading should be provided for administering the pharmacy
compounded suspension as well as for the procedures involved in the home preparation. In both cases,
the correct volumes should preferably be marked on the syringes.
PACKAGE LEAFLET: INFORMATION FOR THE USER
Tamiflu 45 mg hard capsules
oseltamivir
Read all of this leaflet carefully before you start taking this medicine.
-
If you have further questions, ask your doctor or pharmacist.
This medicine has been prescribed for you. Do not pass it on to others. It may harm them, even
if their symptoms are the same as yours.
If any of the side effects gets serious, or if you notice any side effects not listed in this leaflet,
please tell your doctor or pharmacist.
What Tamiflu is and what it is used for
WHAT TAMIFLU IS AND WHAT IT IS USED FOR
Tamiflu belongs to a group of medicines named “neuraminidase inhibitors”. These medicines
prevent the influenza virus from spreading inside the body and so help to ease or prevent the
symptoms arising from the influenza virus infection.
Influenza is an infection caused by the influenza virus. The signs (symptoms) of influenza
include the sudden onset of fever (more than 37.8 °C), cough, runny or stuffy nose, headaches,
muscle aches and often extreme fatigue. These symptoms may also be caused by infections
other than influenza. True influenza infection only occurs during annual outbreaks (epidemics)
at times when influenza viruses are spreading in the local community. Outside of the epidemic
period, these symptoms will mainly be caused by a different type of infection or illness.
if you are allergic (hypersensitive) to oseltamivir or any of the other ingredients of Tamiflu.
Take special care with Tamiflu
Before you take Tamiflu, make sure your prescribing doctor knows if you
-
have problems with your kidneys.
Taking other medicines
Please tell your doctor or pharmacist if you are taking or have recently taken any other medicines,
including medicines obtained without a prescription.
Tamiflu can be taken with appropriate medicines to treat fever (high temperature). Tamiflu is not
expected to alter the effect of any other medicines.
Keep this leaflet. You may need to read it again.
Tamiflu is prescribed to you for treating or preventing influenza.
are allergic to other medicines
Are there any interactions with an influenza vaccination?
Tamiflu is not a substitute for influenza vaccination
. Tamiflu will not change the effectiveness of
the influenza vaccine. Even if a vaccination against influenza has been given to you Tamiflu may be
prescribed by your doctor.
Taking Tamiflu with food and drink
Swallow Tamiflu with water. Tamiflu can be taken with or without food, although it is recommended
to take Tamiflu with food to reduce the chance of feeling or being sick (nausea or vomiting).
Pregnancy and breast-feeding
Ask your doctor or pharmacist for advice before taking any medicine.
You must tell your doctor if you are pregnant, if you think you are pregnant or if you are trying to get
pregnant so that your doctor can decide if Tamiflu is right for you.
The effects on nursing infants are unknown. You must tell your doctor if you are breast-feeding so that
your doctor can decide if Tamiflu is right for you.
Driving and using machines
Tamiflu has no effect on your ability to drive or use machines.
Always take Tamiflu exactly as your doctor has told you. You should check with your doctor or
pharmacist if you are not sure.
Take Tamiflu as soon as you get the prescription as this will help to slow the spread of the influenza
virus in the body.
Swallow Tamiflu capsules whole with water. Do not break or chew Tamiflu capsules.
The usual dose is as follows:
Treatment
¾
Adolescents (13 to 17 years of age) and adults
:
30 mg and 45 mg capsules can be used by adults and adolescents as an alternative to 75 mg capsules.
For treatment of influenza take one 30 mg and one 45 mg capsule as soon as you get the prescription
and then take one 30 mg and one 45 mg capsule twice a day (usually it is convenient to take one
30 mg and one 45 mg in the morning and one 30 mg and one 45 mg in the evening for five days). It is
important to complete the whole 5 day course, even if you start to feel better quickly.
¾
Infants 1 year of age and older, and children 2 to 12 years of age:
Tamiflu oral suspension can be
used instead of capsules.
Children weighing more than 40 kg and who can swallow capsules may take a 75 mg dose of Tamiflu
capsules twice daily for 5 days.
You must give the amount of capsules to your child as prescribed by your doctor.
The usual dose for treatment of influenza in children depends on the body weight of the child (see
table below).



Children 1 year of age and older
:
Body Weight
Recommended dose for 5 days (Treatment)
Less than or equal to 15 kg
More than 15 kg and up to 23 kg
More than 23 kg and up to 40 kg
75 mg twice daily
(75 mg is made up of a 30 mg dose plus a 45 mg dose)
¾
Infants below 12 months of age
:
Limited pharmacokinetic and safety data are available for infants
less than 2 years of age. Pharmacokinetic modeling was undertaken using these data in addition to data
from studies in adults and children older than 1 year of age. The results demonstrate that doses of
3 mg/kg twice daily for infants aged 3 to 12 months and 2.5 mg/kg twice daily for infants aged
between 1 and 3 months provide exposures similar to those shown to be clinically efficacious in adults
and children older than 1 year of age (see table below for weight-based dosing recommendations).
There are currently no data available in infants less than 1 month of age using Tamiflu.
Recommended dose for 5 days (Treatment)
Administration of Tamiflu to infants less than one year of age should be based upon the judgment of
the physician after considering the potential benefit of treatment versus any potential risk to the infant.
Prevention
Tamiflu can also be used for prevention of influenza following exposure to an infected individual,
such as family members.
¾
Adolescents (13 to 17 years of age) and adults
:
30 mg and 45 mg capsules can be used by adults and adolescents as an alternative to 75 mg capsules.
Tamiflu should be taken once daily for 10 days. It is best to take this dose in the mornings with
breakfast.
¾
Infants 1 year of age and older, and children 2 to 12 years of age:
Tamiflu oral suspension can be
used instead of capsules.
The usual dose for prevention of influenza in children depends on the body weight of the child (see
table below).
Children 1 year of age and older
:
Body Weight
Recommended dose for 10 days (Prevention)
Less than or equal to 15 kg
More than 15 kg and up to 23 kg
More than 23 kg and up to 40 kg
75 mg once daily
(75 mg is made up of a 30 mg dose plus a 45 mg dose)
Your doctor will recommend the length of time to continue taking Tamiflu, if it is prescribed to
prevent influenza.
¾
Infants below 12 months of age
: The recommended prophylaxis dose for infants less than
12 months of age during a pandemic influenza outbreak is half of the daily treatment dose. This is
based upon clinical data in children older than 1 year of age and adults showing that a prophylaxis
dose equivalent to half the daily treatment dose is clinically efficacious for the prevention of influenza.
The following weight-adjusted dosing prophylaxis regimens are recommended for infants below
1 year of age:



























Recommended dose for 10 days (Prevention)
Administration of Tamiflu to infants less than one year of age should be based upon the judgment of
the physician after considering the potential benefit of prophylaxis versus any potential risk to the
infant.
When Tamiflu oral suspension is not available
When commercially manufactured Tamiflu oral suspension is not available, a pharmacy preparation
from Tamiflu capsules can be used (see end of package leaflet, information for medical or healthcare
personnel). If the pharmacy preparation is not available either, Tamiflu doses may be prepared at
home. The pharmacy preparation is the preferred option in infants below 12 months of age.
When appropriate capsule strengths are available, the dose is given by opening the capsule and mixing
its contents with no more than one teaspoon of a suitable sweetened food product. The bitter taste can
be masked by products such as sugar water, chocolate syrup, cherry syrup, dessert toppings (like
caramel or fudge sauce). The mixture should be stirred and given entirely to the patient. The mixture
must be swallowed immediately after its preparation.
When only 75 mg capsules are available, and doses of 30 mg or 45 mg are needed, the preparation
involves additional steps.
Preparing Tamiflu doses for infants younger than 1 year — 45 mg capsules
Infants who weigh 10 kg or less need a Tamiflu dose of 30 mg or less. To make doses of Tamiflu for
these infants, you will need to open a Tamiflu capsule, prepare a powder-water mixture and mix the
correct volume of the mixture with food.
One 45 mg Tamiflu capsule
Dosing chart to determine the correct dosage amount (see Tamiflu dosing chart under step 3 in
the next section below)
Two small bowls per child (use separate pairs of bowls for each child)
One 3 ml (three millilitre) oral syringe that shows measurements of 0.1 ml (one-tenth of one
millilitre)
One suitable oral dosing syringe that shows measurements of 0.1 ml (one-tenth of one
millilitre). Ask the pharmacist for the correct syringe (1 ml, 2 ml, or 3 ml).
One of these foods to hide the bitter taste of the Tamiflu powder:
-
Sugar water
(If you plan to use sugar water, make this mixture now in a separate bowl or
cup. Mix a teaspoon of water with three-fourths [3/4] of a teaspoon of sugar.)
Dessert toppings, like caramel or fudge sauce
Making the Tamiflu 10 mg/ml mixture for infants younger than 1 year — 45 mg capsules
These instructions show you how to make one dose of Tamiflu, to be used immediately.
1.
Carefully open ONE 45 mg Tamiflu capsule over the first small bowl. To open a capsule, hold
the capsule upright and snip off the rounded tip with scissors (see accompanying picture). Pour
all of the powder into the bowl. Handle the powder carefully, because it may be irritating to the
skin and eyes







Follow the chart below. Use the 3 ml dosing syringe to measure first 3.0 ml of water, and add it
to the powder in the bowl. Then measure 1.5 ml of water, and add it to the powder in the bowl.
Stir for about 2 minutes
.
4.5 ml (four and a half millilitres)
Follow the chart below. Find the child’s weight on the left side of the chart, and then look at the
column on the right side of chart to see the amount of the Tamiflu powder-water mixture to
draw up into the dosing syringe. Draw up the correct amount of powder-water mixture from the
first small bowl into the dosing syringe, and then gently expel it from the dosing syringe into the
second bowl.
Tamiflu dosing chart for infants 1 to 12 months of age:
Weight
(rounded to the
nearest 0.5 kg)
Amount of Tamiflu powder-water mixture
to be drawn up into the dosing syringe
Tamiflu dosing chart for infants below 1 month of age:
Weight
(rounded to the
nearest 0.5 kg)
Amount of Tamiflu powder-water mixture
to be drawn up into the dosing syringe





























Add a small amount of one of the sweet foods (use no more than 1 teaspoon) into the second
bowl. This will hide the bitter taste of the powder-water mixture.
Mix the sweet food and powder-water mixture well.
Give the entire contents of the second bowl (sweet food with powder-water mixture) to the
child.
Throw out any unused powder-water mixture left in the first bowl.
(Note: Not all capsule contents may dissolve. Do not worry, as the undissolved materials are inactive
ingredients.)
If you take more Tamiflu, than you should
Contact your doctor or pharmacist immediately.
If you have forgotten to take Tamiflu
Do not take a double dose to make up for a forgotten capsule.
If you stop taking Tamiflu
There are no side effects when Tamiflu is discontinued prior to advice from your doctor. If Tamiflu is
stopped earlier than your doctor told you, the symptoms of influenza may reoccur.
If you have any further questions on the use of this product, ask your doctor or pharmacist.
Like all medicines, Tamiflu can cause side effects, although not everybody gets them.
Common side effects of Tamiflu
The most common side effects of Tamiflu are nausea, vomiting, diarrhoea, stomach ache and
headache. These side effects mostly occur only after the first dose of the medicine and will usually
stop as treatment continues. The frequency of these effects is reduced if the medicinal product is taken
with food.
Give the child something suitable to drink.


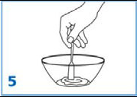
Less common side effects of Tamiflu
During Tamiflu treatment, events like convulsions and delirium (including symptoms such as altered
level of consciousness, confusion, abnormal behaviour, delusions, hallucinations, agitation, anxiety,
nightmares) have been reported, in a very few cases resulting in self-injury, in some instances with
fatal outcome. These events were reported primarily among children and adolescents and often had an
abrupt onset and rapid resolution. Such neuropsychiatric events have also been reported in patients
with influenza who were not taking Tamiflu.
Adults and adolescents (children aged 13 years and older)
Other less common side effects, which may also be caused by influenza, are upper abdominal fullness,
bleeding in the gastrointestinal tract, bronchitis, upper respiratory tract infections, dizziness, tiredness,
sleeping difficulties, skin reactions, mild to severe liver function disorders, visual disturbances and
heart rhythm abnormalities.
Children (aged 1 to 12 years)
Other less common side effects, which may also be caused by influenza, are cough, nasal congestion,
ear inflammation, inflammation of the lungs, sinusitis, bronchitis, aggravation of pre-existing asthma,
nose bleeding, ear disorders, inflammation of the skin, swelling of the lymph nodes, conjunctivitis,
visual disturbances and heart rhythm abnormalities.
Infants (aged 6 to 12 months)
The reported side effects of Tamiflu when used for treatment of influenza in infants 6 to 12 months of
age are similar to the side effects reported for older children (1 year and older). Please read the section
above for your information.
Infants (aged 0 to 6 months)
The reported side effects of Tamiflu when used for treatment of influenza in infants 1 to 6 months of
age are similar to the side effects reported for infants 6 to 12 months of age and older children (1 year
and older). There are no data available for the use of Tamiflu in infants less than 1 month of age.
Please read the section above for your information.
If you or your child are often sick, you should inform your doctor. You should also tell your doctor
if the influenza symptoms get worse or the fever continues.
If any of the side effects get serious, or if you notice any side effects not listed in this leaflet, tell your
doctor or pharmacist.
Keep out of the reach and sight of children.
Do not use Tamiflu after the expiry date which is stated on the carton and blister after EXP. The
expiry date refers to the last day of that month.
Do not store above 25 °C.
Medicines should not be disposed of via wastewater or household waste. Ask your pharmacist how to
dispose of medicines no longer required. These measures will help to protect the environment.



The active substance is oseltamivir phosphate (45 mg oseltamivir in each hard capsule).
The other ingredients are:
capsule contents: pregelatinised starch, talc, povidone, croscarmellose sodium and sodium
stearyl fumarate
capsule shell: gelatin, black iron oxide (E172) and titanium dioxide (E171)
printing ink: shellac (E904), titanium dioxide (E171) and FD and C Blue 2 (indigocarmine,
E132).
What Tamiflu looks like and contents of the pack
45 mg hard capsule consists of a grey opaque body bearing the imprint “ROCHE” and a grey opaque
cap bearing the imprint “45 mg”. Imprints are blue.
Tamiflu 45 mg hard capsules are available in blister packs of 10.
Marketing Authorisation Holder and Manufacturer
Roche Registration Limited
6 Falcon Way
Shire Park
Welwyn Garden City
AL7 1TW
United Kingdom
Roche Pharma AG
Emil-Barell-Str. 1,
D-79639 Grenzach-Wyhlen
Germany
For any information about this medicine, please contact the local representative of the Marketing
Authorisation Holder.
België/Belgique/Belgien
N.V. Roche S.A.
Tél/Tel: +32 (0) 2 525 82 11
Luxembourg/Luxemburg
(Voir/siehe Belgique/Belgien)
България
Рош България ЕООД
Тел: +359 2 818 44 44
Magyarország
Roche (Magyarország) Kft.
Tel: +36 - 23 446 800
Česká republika
Roche s. r. o.
Tel: +420 - 2 20382111
Malta
(See United Kingdom)
Danmark
Roche a/s
Tlf: +45 - 36 39 99 99
Nederland
Roche Nederland B.V.
Tel: +31 (0) 348 438050
Deutschland
Roche Pharma AG
Tel: +49 (0) 7624 140
Norge
Roche Norge AS
Tlf: +47 - 22 78 90 00
Eesti
Roche Eesti OÜ
Tel: + 372 - 6 177 380
Österreich
Roche Austria GmbH
Tel: +43 (0) 1 27739
Ελλάδα
Roche (Hellas) A.E.
Τηλ: +30 210 61 66 100
Polska
Roche Polska Sp.z o.o.
Tel: +48 - 22 345 18 88
España
Roche Farma S.A.
Tel: +34 - 91 324 81 00
Portugal
Roche Farmacêutica Química, Lda
Tel: +351 - 21 425 70 00
France
Roche
Tél: +33 (0) 1 46 40 50 00
România
Roche România S.R.L.
Tel: +40 21 206 47 01
Ireland
Roche Products (Ireland) Ltd.
Tel: +353 (0) 1 469 0700
Slovenija
Roche farmacevtska družba d.o.o.
Tel: +386 - 1 360 26 00
Ísland
Roche a/s
c/o Icepharma hf
Sími: +354 540 8000
Slovenská republika
Roche Slovensko, s.r.o.
Tel: +421 - 2 52638201
Italia
Roche S.p.A.
Tel: +39 - 039 2471
Suomi/Finland
Roche Oy
Puh/Tel: +358 (0) 10 554 500
Kύπρος
Γ.Α.Σταμάτης & Σια Λτδ.
Τηλ: +357 - 22 76 62 76
Sverige
Roche AB
Tel: +46 (0) 8 726 1200
Latvija
Roche Latvija SIA
Tel: +371 - 6 7039831
United Kingdom
Roche Products Ltd.
Tel: +44 (0) 1707 366000
Lietuva
UAB “Roche Lietuva”
Tel: +370 5 2546799
This leaflet was last approved in {MM/YYYY}.
Detailed information on this medicine is available on the European Medicines Agency (EMEA) web
site: http://www.emea.europa.eu/
.
---------------------------------------------------------------------------------------------------------------------------
The following information is intended for medical or healthcare professionals only:
When commercially manufactured Tamiflu powder for oral suspension is not available, patients who
are unable to swallow capsules may receive appropriate doses of Tamiflu prepared in a pharmacy or
prepared at home.
For infants below 12 months, the pharmacy preparation should be preferred to home preparation.
Detailed information on the pharmacy preparation can be found in the SmPC of Tamiflu capsules
section 4.2.
Syringes of appropriate volume and grading should be provided for administering the pharmacy
compounded suspension as well as for the procedures involved in the home preparation. In both cases,
the correct volumes should preferably be marked on the syringes.
PACKAGE LEAFLET: INFORMATION FOR THE USER
Tamiflu 75 mg hard capsules
oseltamivir
Read all of this leaflet carefully before you start taking this medicine.
-
If you have further questions, ask your doctor or pharmacist.
This medicine has been prescribed for you. Do not pass it on to others. It may harm them, even
if their symptoms are the same as yours.
If any of the side effects gets serious, or if you notice any side effects not listed in this leaflet,
please tell your doctor or pharmacist.
What Tamiflu is and what it is used for
WHAT TAMIFLU IS AND WHAT IT IS USED FOR
Tamiflu belongs to a group of medicines named “neuraminidase inhibitors”. These medicines
prevent the influenza virus from spreading inside the body and so help to ease or prevent the
symptoms arising from the influenza virus infection.
Influenza is an infection caused by the influenza virus. The signs (symptoms) of influenza
include the sudden onset of fever (more than 37.8 °C), cough, runny or stuffy nose, headaches,
muscle aches and often extreme fatigue. These symptoms may also be caused by infections
other than influenza. True influenza infection only occurs during annual outbreaks (epidemics)
at times when influenza viruses are spreading in the local community. Outside of the epidemic
period, these symptoms will mainly be caused by a different type of infection or illness.
if you are allergic (hypersensitive) to oseltamivir or any of the other ingredients of Tamiflu.
Take special care with Tamiflu
Before you take Tamiflu, make sure your prescribing doctor knows if you
-
have problems with your kidneys.
Taking other medicines
Please tell your doctor or pharmacist if you are taking or have recently taken any other medicines,
including medicines obtained without a prescription.
Tamiflu can be taken with appropriate medicines to treat fever (high temperature). Tamiflu is not
expected to alter the effect of any other medicines.
Keep this leaflet. You may need to read it again.
Tamiflu is prescribed to you for treating or preventing influenza.
are allergic to other medicines
Are there any interactions with an influenza vaccination?
Tamiflu is not a substitute for influenza vaccination
. Tamiflu will not change the effectiveness of
the influenza vaccine. Even if a vaccination against influenza has been given to you Tamiflu may be
prescribed by your doctor.
Taking Tamiflu with food and drink
Swallow Tamiflu with water. Tamiflu can be taken with or without food, although it is recommended
to take Tamiflu with food to reduce the chance of feeling or being sick (nausea or vomiting).
Pregnancy and breast-feeding
Ask your doctor or pharmacist for advice before taking any medicine.
You must tell your doctor if you are pregnant, if you think you are pregnant or if you are trying to get
pregnant so that your doctor can decide if Tamiflu is right for you.
The effects on nursing infants are unknown. You must tell your doctor if you are breast-feeding so that
your doctor can decide if Tamiflu is right for you.
Driving and using machines
Tamiflu has no effect on your ability to drive or use machines.
Always take Tamiflu exactly as your doctor has told you. You should check with your doctor or
pharmacist if you are not sure.
Take Tamiflu as soon as you get the prescription as this will help to slow the spread of the influenza
virus in the body.
Swallow Tamiflu capsules whole with water. Do not break or chew Tamiflu capsules.
The usual dose is as follows:
Treatment
¾
Adolescents (13 to 17 years of age) and adults
: For treatment of influenza take one capsule as soon
as you get the prescription and then take one capsule twice a day (usually it is convenient to take one
in the morning and one in the evening for five days). It is important to complete the whole 5 day
course, even if you start to feel better quickly.
30 mg and 45 mg capsules can be used by adults and adolescents as an alternative to 75 mg capsules.
¾
Infants 1 year of age and older, and children 2 to 12 years of age:
Tamiflu oral suspension or 30 mg
and 45 mg capsules can be used instead.
Children weighing more than 40 kg and who can swallow capsules may take Tamiflu 75 mg capsules
twice daily for 5 days.
¾
Infants below 12 months of age
:
Limited pharmacokinetic and safety data are available for children
less than 2 years of age. Pharmacokinetic modeling was undertaken using these data in addition to data
from studies in adults and children older than 1 year of age. The results demonstrate that doses of
3 mg/kg twice daily for infants aged 3 to 12 months and 2.5 mg/kg twice daily for infants aged
between 1 and 3 months provide exposures similar to those shown to be clinically efficacious in adults
and children older than 1 year of age (see table below for weight-based dosing recommendations).
There are currently no data available in infants less than 1 month of age using Tamiflu.



Recommended dose for 5 days (Treatment)
Administration of Tamiflu to infants less than one year of age should be based upon the judgment of
the physician after considering the potential benefit of treatment versus any potential risk to the infant.
Prevention
Tamiflu can also be used for prevention of influenza following exposure to an infected individual,
such as family members.
¾
Adolescents (13 to 17 years of age) and adults
: When used to prevent influenza following exposure
to an infected individual, such as family members, Tamiflu should be taken once daily for 10 days. It
is best to take this dose in the mornings with breakfast.
¾
Infants 1 year of age and older and children 2 to 12 years of age
: Tamiflu oral suspension or 30 mg
and 45 mg capsules can be used instead.
Children weighing more than 40 kg and can swallow capsules may take Tamiflu 75 mg capsules once
daily for 10 days.
Your doctor will recommend the length of time to continue taking Tamiflu, if it is prescribed to
prevent influenza.
¾
Infants below 12 months of age
: The recommended prophylaxis dose for infants less than
12 months of age during a pandemic influenza outbreak is half of the daily treatment dose. This is
based upon clinical data in children older than 1 year of age and adults showing that a prophylaxis
dose equivalent to half the daily treatment dose is clinically efficacious for the prevention of influenza.
The following weight-adjusted dosing prophylaxis regimens are recommended for infants below
1 year of age:
Recommended dose for 10 days (Prevention)
Administration of Tamiflu to infants less than one year of age should be based upon the judgment of
the physician after considering the potential benefit of prophylaxis versus any potential risk to the
infant.
When Tamiflu oral suspension is not available
When commercially manufactured Tamiflu oral suspension is not available, a pharmacy preparation
from Tamiflu capsules can be used (see end of package leaflet, information for medical or healthcare
personnel). If the pharmacy preparation is not available either, Tamiflu doses may be prepared at
home. The pharmacy preparation is the preferred option in infants below 12 months of age.
When appropriate capsule strengths are available, the dose is given by opening the capsule and mixing
its contents with no more than one teaspoon of a suitable sweetened food product. The bitter taste can
be masked by products such as sugar water, chocolate syrup, cherry syrup, dessert toppings (like
caramel or fudge sauce). The mixture should be stirred and given entirely to the patient. The mixture
must be swallowed immediately after its preparation.
When only 75 mg capsules are available, and doses of 30 mg or 45 mg are needed, the preparation
involves additional steps.



















Preparing Tamiflu doses for infants younger than 1 year — 75 mg capsules
Infants who weigh 10 kg or less need a Tamiflu dose of 30 mg or less. To make doses of Tamiflu for
these infants, you will need to open a Tamiflu capsule and mix the powder inside the capsule with
food.
Dosing chart to determine the correct dosage amount (see Tamiflu dosing chart under step 3 in
the next section below)
Two small bowls per child (use separate pairs of bowls for each child)
One 5 ml (five millilitre) oral syringe that shows measurements of 0.2 ml (two-tenths of one
millilitre)
One suitable oral dosing syringe that shows measurements of 0.1 ml (one-tenth of one
millilitre). Ask the pharmacist for the correct syringe (1 ml, 2 ml, or 3 ml).
One of these foods to hide the bitter taste of the Tamiflu powder:
-
Sugar water
(If you plan to use sugar water, make this mixture now in a separate bowl or
cup. Mix a teaspoon of water with three-fourths [3/4] of a teaspoon of sugar.)
Dessert toppings, like caramel or fudge sauce
Making the Tamiflu 10 mg/ml mixture for infants younger than 1 year — 75 mg capsules
These instructions show you how to make one dose of Tamiflu, to be used immediately.
1.
Carefully open ONE 75 mg Tamiflu capsule over the first small bowl. To open a capsule, hold
the capsule upright and snip off the rounded tip with scissors (see accompanying picture). Pour
all of the powder into the bowl. Handle the powder carefully, because it may be irritating to the
skin and eyes.
Follow the chart below. Use the 5 ml dosing syringe to measure first 5.0 ml of water, and add it
to the powder in the bowl. Then measure 2.5 ml of water, and add it to the powder in the bowl.
Stir for about 2 minutes
.
7.5 ml (seven and a half
millilitres)
Follow the chart below. Find the child’s weight on the left side of the chart, and then look at the
column on the right side of chart to see the amount of the Tamiflu powder-water mixture to
draw up into the dosing syringe. Draw up the correct amount of powder-water mixture from the
first small bowl into the dosing syringe, and then gently expel it from the dosing syringe into the
second bowl.
One 75 mg Tamiflu capsule








Tamiflu dosing chart for infants 1 to 12 months of age:
Weight
(rounded to the
nearest 0.5 kg)
Amount of Tamiflu powder-water mixture
to be drawn up into the dosing syringe
Tamiflu dosing chart for infants below 1 month of age:
Weight
(rounded to the
nearest 0.5 kg)
Amount of Tamiflu powder-water mixture
to be drawn up into the dosing syringe
Add a small amount of one of the sweet foods (use no more than 1 teaspoon) into the second
bowl. This will hide the bitter taste of the powder-water mixture.
Mix the sweet food and powder-water mixture well.
Give the entire contents of the second bowl (sweet food with powder-water mixture) to the
child.
Give the child something suitable to drink.
Throw out any unused powder-water mixture left in the first bowl.
(Note: Not all capsule contents may dissolve. Do not worry, as the undissolved materials are inactive
ingredients.)


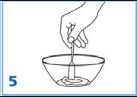






















Preparing Tamiflu doses for children 1 year of age or older — if you have only 75 mg capsules
available
Children who weigh less than 40 kg need a Tamiflu dose of less than 75 mg. To make doses of
Tamiflu for these children, you will need to open a 75 mg capsule, prepare a powder-water mixture
and mix the correct volume of the mixture with food.
You will need
1.
Dosing chart to determine the correct dosage amount (see charts under step 3 in the next section
below)
Two small bowls per child (use separate pairs of bowls for each child)
One 5 ml (five millilitre) oral syringe that shows measurements of 0.2 ml (two-tenths of one
millilitre)
One suitable oral dosing syringe that shows measurements of 0.1 ml (one-tenth of one
millilitre). Ask the pharmacist for the correct syringe (1 ml, 2 ml, or 3 ml).
One of these foods to hide the bitter taste of the Tamiflu powder:
-
Sugar water
(If you plan to use sugar water, make this mixture now in a separate bowl or
cup. Mix a teaspoon of water with three-fourths [3/4] of a teaspoon of sugar.)
Dessert toppings, like caramel or fudge sauce
Making the Tamiflu mixture for children 1 year of age or older — if you have only 75 mg
capsules available
These instructions show you how to make one dose of Tamiflu, to be used immediately.
1.
Carefully open ONE 75 mg Tamflu capsule over the first small bowl. To open a capsule, hold
the capsule upright and snip off the rounded tip with scissors (see accompanying picture). Pour
all of the powder into the bowl. Handle the powder carefully, because it may be irritating to the
skin and eyes.
Use the 5 ml syringe to add 5 ml of water to the powder in the bowl.
Stir for about 2 minutes
.
(Note: Not all capsule contents may dissolve. Do not worry, as the undissolved materials are
inactive ingredients.)
The next step will be to draw up the correct amount of powder-water mixture from the first
small bowl, and to gently expel it into the second small bowl.
You must find what the correct amount of powder-water mixture to draw up into the dosing
syringe is. If you know the child’s weight, you can do this by finding the child’s weight on the
left side of the first table below. Then, look at the middle column of the table to see the correct
amount of the powder-water mixture to draw up into the dosing syringe.
One 75 mg Tamiflu capsule

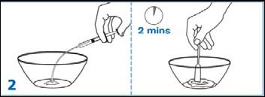
If you do not know the child’s weight, find the child’s age on the left side of the second table
below. Then, look at the middle column of the table to see the correct amount of the powder-
water mixture to draw up into the dosing syringe
.
Amount of Tamiflu Mixture
Amount of Tamiflu Mixture
Once you have drawn up the correct amount of powder-water mixture into the dosing syringe,
gently expel it from the dosing syringe into the second bowl.
Add a small amount of one of the sweet foods into the second bowl to hide the bitter taste of the
powder-water mixture.
Mix the sweet food and powder-water mixture well.
Give the entire contents of the second bowl (sweet food with powder-water mixture) to the
child.
Throw out any unused powder-water mixture left in the first bowl.
Repeat this procedure every time you need to take the medicine.
If you take more Tamiflu, than you should
Contact your doctor or pharmacist immediately.
If you have forgotten to take Tamiflu
Do not take a double dose to make up for a forgotten capsule.
















If you stop taking Tamiflu
There are no side effects when Tamiflu is discontinued prior to advice from your doctor. If Tamiflu is
stopped earlier than your doctor told you, the symptoms of influenza may reoccur.
If you have any further questions on the use of this product, ask your doctor or pharmacist.
Like all medicines, Tamiflu can cause side effects, although not everybody gets them.
Common side effects of Tamiflu
The most common side effects of Tamiflu are nausea, vomiting, diarrhoea, stomach ache and
headache. These side effects mostly occur only after the first dose of the medicine and will usually
stop as treatment continues. The frequency of these effects is reduced if the medicinal product is taken
with food.
Less common side effects of Tamiflu
During Tamiflu treatment, events like convulsions and delirium (including symptoms such as altered
level of consciousness, confusion, abnormal behaviour, delusions, hallucinations, agitation, anxiety,
nightmares) have been reported, in a very few cases resulting in self-injury, in some instances with
fatal outcome. These events were reported primarily among children and adolescents and often had an
abrupt onset and rapid resolution. Such neuropsychiatric events have also been reported in patients
with influenza who were not taking Tamiflu.
Adults and adolescents (children aged 13 years and older)
Other less common side effects, which may also be caused by influenza, are upper abdominal fullness,
bleeding in the gastrointestinal tract, bronchitis, upper respiratory tract infections, dizziness, tiredness,
sleeping difficulties, skin reactions, mild to severe liver function disorders, visual disturbances and
heart rhythm abnormalities.
Children (aged 1 to 12 years)
Other less common side effects, which may also be caused by influenza, are cough, nasal congestion,
ear inflammation, inflammation of the lungs, sinusitis, bronchitis, aggravation of pre-existing asthma,
nose bleeding, ear disorders, inflammation of the skin, swelling of the lymph nodes, conjunctivitis,
visual disturbances and heart rhythm abnormalities.
Infants (aged 6 to 12 months)
The reported side effects of Tamiflu when used for treatment of influenza in infants 6 to 12 months of
age are similar to the side effects reported for older children (1 year and older). Please read the section
above for your information.
Infants (aged 0 to 6 months)
The reported side effects of Tamiflu when used for treatment of influenza in infants 1 to 6 months of
age are similar to the side effects reported for infants 6 to 12 months of age and older children (1 year
and older). There are no data available for the use of Tamiflu in infants less than 1 month of age.
Please read the section above for your information.
If you or your child are often sick, you should inform your doctor. You should also tell your doctor
if the influenza symptoms get worse or the fever continues.
If any of the side effects get serious, or if you notice any side effects not listed in this leaflet, tell your
doctor or pharmacist.



Keep out of the reach and sight of children.
Do not use Tamiflu after the expiry date which is stated on the carton and blister after EXP. The
expiry date refers to the last day of that month.
Do not store above 25 °C.
Medicines should not be disposed of via wastewater or household waste. Ask your pharmacist how to
dispose of medicines no longer required. These measures will help to protect the environment.
The active substance is oseltamivir phosphate (75 mg oseltamivir in each hard capsule).
The other ingredients are:
capsule contents: pregelatinised starch, talc, povidone, croscarmellose sodium and sodium
stearyl fumarate
capsule shell: gelatin, yellow iron oxide (E172), red iron oxide (E172), black iron oxide (E172)
and titanium dioxide (E171)
printing ink: shellac (E904), titanium dioxide (E171) and indigocarmine (E132).
What Tamiflu looks like and contents of the pack
The hard capsule consists of a grey opaque body bearing the imprint “ROCHE” and a light yellow
opaque cap bearing the imprint “75 mg”. Imprints are blue.
Tamiflu 75 mg hard capsules are available in blister packs of 10.
Marketing Authorisation Holder and Manufacturer
Roche Registration Limited
6 Falcon Way
Shire Park
Welwyn Garden City
AL7 1TW
United Kingdom
Roche Pharma AG
Emil-Barell-Str. 1,
D-79639 Grenzach-Wyhlen
Germany
For any information about this medicine, please contact the local representative of the Marketing
Authorisation Holder.
België/Belgique/Belgien
N.V. Roche S.A.
Tél/Tel: +32 (0) 2 525 82 11
Luxembourg/Luxemburg
(Voir/siehe Belgique/Belgien)
България
Рош България ЕООД
Тел: +359 2 818 44 44
Magyarország
Roche (Magyarország) Kft.
Tel: +36 - 23 446 800
Česká republika
Roche s. r. o.
Tel: +420 - 2 20382111
Malta
(See United Kingdom)
Danmark
Roche a/s
Tlf: +45 - 36 39 99 99
Nederland
Roche Nederland B.V.
Tel: +31 (0) 348 438050
Deutschland
Roche Pharma AG
Tel: +49 (0) 7624 140
Norge
Roche Norge AS
Tlf: +47 - 22 78 90 00
Eesti
Roche Eesti OÜ
Tel: + 372 - 6 177 380
Österreich
Roche Austria GmbH
Tel: +43 (0) 1 27739
Ελλάδα
Roche (Hellas) A.E.
Τηλ: +30 210 61 66 100
Polska
Roche Polska Sp.z o.o.
Tel: +48 - 22 345 18 88
España
Roche Farma S.A.
Tel: +34 - 91 324 81 00
Portugal
Roche Farmacêutica Química, Lda
Tel: +351 - 21 425 70 00
France
Roche
Tél: +33 (0) 1 46 40 50 00
România
Roche România S.R.L.
Tel: +40 21 206 47 01
Ireland
Roche Products (Ireland) Ltd.
Tel: +353 (0) 1 469 0700
Slovenija
Roche farmacevtska družba d.o.o.
Tel: +386 - 1 360 26 00
Ísland
Roche a/s
c/o Icepharma hf
Sími: +354 540 8000
Slovenská republika
Roche Slovensko, s.r.o.
Tel: +421 - 2 52638201
Italia
Roche S.p.A.
Tel: +39 - 039 2471
Suomi/Finland
Roche Oy
Puh/Tel: +358 (0) 10 554 500
Kύπρος
Γ.Α.Σταμάτης & Σια Λτδ.
Τηλ: +357 - 22 76 62 76
Sverige
Roche AB
Tel: +46 (0) 8 726 1200
Latvija
Roche Latvija SIA
Tel: +371 - 6 7039831
United Kingdom
Roche Products Ltd.
Tel: +44 (0) 1707 366000
Lietuva
UAB “Roche Lietuva”
Tel: +370 5 2546799
This leaflet was last approved in {MM/YYYY}.
Detailed information on this medicine is available on the European Medicines Agency (EMEA) web
site: http://www.emea.europa.eu/.
---------------------------------------------------------------------------------------------------------------------------
The following information is intended for medical or healthcare professionals only:
When commercially manufactured Tamiflu powder for oral suspension is not available, patients who
are unable to swallow capsules may receive appropriate doses of Tamiflu prepared in a pharmacy or
prepared at home.
For infants below 12 months, the pharmacy preparation should be preferred to home preparation.
Detailed information on the pharmacy preparation can be found in the SmPC of Tamiflu capsules
section 4.2.
Syringes of appropriate volume and grading should be provided for administering the pharmacy
compounded suspension as well as for the procedures involved in the home preparation. In both cases,
the correct volumes should preferably be marked on the syringes.
PACKAGE LEAFLET: INFORMATION FOR THE USER
Tamiflu 12 mg/ml powder for oral suspension
oseltamivir
Read all of this leaflet carefully before you start taking this medicine.
-
If you have further questions, ask your doctor or pharmacist.
This medicine has been prescribed for you. Do not pass it on to others. It may harm them, even
if their symptoms are the same as yours.
If any of the side effects gets serious, or if you notice any side effects not listed in this leaflet,
please tell your doctor or pharmacist.
What Tamiflu is and what it is used for
WHAT TAMIFLU IS AND WHAT IT IS USED FOR
Tamiflu belongs to a group of medicines named “neuraminidase inhibitors”. These medicines
prevent the influenza virus from spreading inside the body and so help to ease or prevent the
symptoms arising from the influenza virus infection.
Influenza is an infection caused by the influenza virus. The signs (symptoms) of influenza
include the sudden onset of fever (more than 37.8 °C), cough, runny or stuffy nose, headaches,
muscle aches and often extreme fatigue. These symptoms may also be caused by infections
other than influenza. True influenza infection only occurs during annual outbreaks (epidemics)
at times when influenza viruses are spreading in the local community. Outside of the epidemic
period, these symptoms will mainly be caused by a different type of infection or illness.
if you are allergic (hypersensitive) to oseltamivir or any of the other ingredients of Tamiflu
.
Take special care with Tamiflu
Before you take Tamiflu, make sure your prescribing doctor knows if you
-
have problems with your kidneys
If you have been told by your doctor that you have an intolerance to some sugars, contact your doctor
before taking this medicinal product.
Taking other medicines
Please tell your doctor or pharmacist if you are taking or have recently taken any other medicines,
including medicines obtained without a prescription.
Tamiflu can be taken with appropriate medicines to treat fever (high temperature). Tamiflu is not
expected to alter the effect of any other medicines.
Keep this leaflet. You may need to read it again.
Tamiflu is prescribed to you for treating or preventing influenza.
are allergic to other medicines
Are there any interactions with an influenza vaccination?
Tamiflu is not a substitute for influenza vaccination
.
Tamiflu will not change the effectiveness of
the influenza vaccine. Even if a vaccination against influenza has been given to you Tamiflu may be
prescribed by your doctor.
Taking Tamiflu with food and drink
Tamiflu oral suspension can be taken with or without food, although it is recommended to take
Tamiflu with food to reduce the chance of feeling or being sick (nausea or vomiting). It can also be
taken with a drink of water.
Pregnancy and breast-feeding
Ask your doctor or pharmacist for advice before taking any medicine.
You must tell your doctor if you are pregnant, if you think you are pregnant or if you are trying to get
pregnant so that your doctor can decide if Tamiflu is right for you.
The effects on nursing infants are unknown. You must tell your doctor if you are breast-feeding so that
your doctor can decide if Tamiflu is right for you.
Driving and using machines
Tamiflu has no effect on your ability to drive or use machines.
Important information about some of the ingredients of Tamiflu
Before you take Tamiflu, make sure your prescribing doctor knows if you have hereditary fructose
intolerance.
This medicine contains sorbitol which is a form of fructose.
Sorbitol can have a mild laxative effect.
Always take Tamiflu exactly as your doctor has told you. You should check with your doctor or
pharmacist if you are not sure.
Always use the syringe that is provided in the box and has markings indicating the dose in mg.
Take Tamiflu as soon as you get the prescription as this will help to slow the spread of the influenza
virus in the body.
¾
Infants below 12 months of age
: This formulation is not suitable for dosing infants less than
12 months of age.
The usual dose is as follows:
How much Tamiflu should be given to infants 1 year of age and older and children 2 to 12 years
of age)
Treatment
You must give the amount of oral suspension to your child as prescribed by your doctor.
30 mg and 45 mg capsules can be used as an alternative to suspension.



The usual dose for treatment of influenza in children depends on the body weight of the child (see
table below):
Body Weight
Recommended dose for 5 days (Treatment)
Less than or equal to 15 kg
More than 15 kg and up to 23 kg
More than 23 kg and up to 40 kg
75 mg twice daily
(75 mg is made up of a 30 mg dose plus a 45 mg dose)
¾
Infants below 12 months of age
: This formulation is not suitable for dosing infants less than
12 months of age.
Prevention
Tamiflu can also be used to prevent influenza following exposure to an infected individual, such as
family members.
30 mg and 45 mg capsules can be used as an alternative to suspension.
Tamiflu should be taken once daily for 10 days. It is best to take this dose in the mornings with
breakfast.
The usual dose for prevention of influenza in children depends on the body weight of the child (see
table below):
Body Weight
Recommended dose for 10 days (Prevention)
Less than or equal to 15 kg
More than 15 kg and up to 23 kg
More than 23 kg and up to 40 kg
75 mg once daily
(75 mg is made up of a 30 mg dose plus a 45 mg dose)
Children weighing more than 40 kg and can swallow capsules may take a 75 mg dose of Tamiflu
capsules once daily for 10 days.
Your doctor will recommend the length of time to continue taking Tamiflu, if it is prescribed to
prevent influenza.
¾
Infants below 12 months of age
: This formulation is not suitable for dosing infants less than
12 months of age.
How Much Tamiflu should be given to Adolescents (13 to 17 years of age) and Adults
The suspension can be used by adults and adolescents instead of the capsule.
30 mg and 45 mg capsules can be used as an alternative to suspension.
Treatment
Usually, your doctor will prescribe a 75 mg dose of Tamiflu twice daily for 5 days.
Prevention
Usually, your doctor will prescribe a 75 mg dose of Tamiflu once daily for 10 days. Your doctor will
recommend the length of time you will need to take Tamiflu.
A single 75 mg dose of Tamiflu requires two doses of the suspension: one 30 mg dose and one 45 mg
dose.
























9. Push the suspension directly into the mouth by pushing down the plunger of the dispenser.
Swallow the medicine. You may drink and eat something after taking the medicine.
10. Immediately after administration, take the dispenser apart and rinse both parts of the dispenser
under running tap water.
If you take more Tamiflu, than you should
Contact your doctor or pharmacist immediately.
If you have forgotten to take Tamiflu
Do not take a double dose to make up for a forgotten dose.
If you stop taking Tamiflu
There are no side effects when Tamiflu is discontinued prior to advice from your doctor. If Tamiflu is
stopped earlier than your doctor told you, the symptoms of influenza may reoccur.
If you have any further questions on the use of this product, ask your doctor or pharmacist.
Like all medicines, Tamiflu can cause side effects, although not everybody gets them.
Common side effects of Tamiflu
The most common side effects of Tamiflu are nausea, vomiting, diarrhoea, stomach ache and
headache. These side effects mostly occur only after the first dose of the medicine and will usually
stop as treatment continues. The frequency of these effects is reduced if the medicinal product is taken
with food.
Less common side effects of Tamiflu
During Tamiflu treatment, events like convulsions and delirium (including symptoms such as altered
level of consciousness, confusion, abnormal behaviour, delusions, hallucinations, agitation, anxiety,
nightmares) have been reported, in a very few cases resulting in self-injury, in some instances with
fatal outcome. These events were reported primarily among children and adolescents and often had an
abrupt onset and rapid resolution. Such neuropsychiatric events have also been reported in patients
with influenza who were not taking Tamiflu.
Adults and adolescents (children aged 13 years and older)
Other less common side effects, which may also be caused by influenza, are upper abdominal fullness,
bleeding in the gastrointestinal tract, bronchitis, upper respiratory tract infections, dizziness, tiredness,
sleeping difficulties, skin reactions, mild to severe liver function disorders, visual disturbances and
heart rhythm abnormalities.
Children (aged 1 to 12 years)
Other less common side effects, which may also be caused by influenza, are cough, nasal congestion,
ear inflammation, inflammation of the lungs, sinusitis, bronchitis, aggravation of pre-existing asthma,
nose bleeding, ear disorders, inflammation of the skin, swelling of the lymph nodes, conjunctivitis,
visual disturbances and heart rhythm abnormalities.
Infants (aged 6 to 12 months)
The reported side effects of Tamiflu when used for treatment of influenza in infants 6 to 12 months of
age are similar to the side effects reported for older children (1 year and older). Please read the section
above for your information.
Infants (aged 0 to 6 months)
The reported side effects of Tamiflu when used for treatment of influenza in infants 1 to 6 months of
age are similar to the side effects reported for infants 6 to 12 months of age and older children (1 year
and older). There are no data available for the use of Tamiflu in infants less than 1 month of age.
Please read the section above for your information.
If you or your child are often sick, you should inform your doctor. You should also tell your doctor
if the influenza symptoms get worse or the fever continues.
If any of the side effects gets serious, or if you notice any side effects not listed in this leaflet, please
tell your doctor or pharmacist.
Keep out of the reach and sight of children.
Do not use Tamiflu powder for oral suspension after the expiry date which is stated on the carton and
bottle after EXP. The expiry date refers to the last day of that month.
Powder: Do not store above 30°C.
After reconstitution, the suspension can be stored either at room temperature (not above 25°C) for
10 days or in a refrigerator (2°C - 8°C) for 17 days.
Medicines should not be disposed of via wastewater or household waste. Ask your pharmacist how to
dispose of medicines no longer required. These measures will help to protect the environment.
The other ingredients are sorbitol (E420), sodium dihydrogen citrate (E331[a]), xanthan gum
(E415), sodium benzoate (E211), saccharin sodium (E954), titanium dioxide (E171) and
flavour.
What Tamiflu looks like and contents of the pack
The powder is a granulate or clumped granulate with a white to light yellow colour.
Tamiflu 12 mg/ml powder for oral suspension is available in a bottle containing 30 g powder for
mixing with 52 ml of water.
The box also contains 1 plastic measuring cup (52 ml), 1 plastic bottle adapter (to help get the drug
into the dispenser) and 1 plastic oral dispenser (to give the correct amount of medicine via the mouth).
Shown on the oral dispenser are marks for 30 mg, 45 mg and 60 mg of the medicine (see figures 1 and
2 above).
For details on how to prepare the oral suspension and how to measure and take the medicine, read
section 3, “How to take Tamiflu”.
The active substance is oseltamivir phosphate (12 mg/ml oseltamivir after reconstitution).



Marketing Authorisation Holder and Manufacturer
Roche Registration Limited
6 Falcon Way
Shire Park
Welwyn Garden City
AL7 1TW
United Kingdom
Roche Pharma AG
Emil-Barell-Str. 1,
D-79639 Grenzach-Wyhlen
Germany
For any information about this medicine, please contact the local representative of the Marketing
Authorisation Holder.
België/Belgique/Belgien
N.V. Roche S.A.
Tél/Tel: +32 (0) 2 525 82 11
Luxembourg/Luxemburg
(Voir/siehe Belgique/Belgien)
България
Рош България ЕООД
Тел: +359 2 818 44 44
Magyarország
Roche (Magyarország) Kft.
Tel: +36 - 23 446 800
Česká republika
Roche s. r. o.
Tel: +420 - 2 20382111
Malta
(See United Kingdom)
Danmark
Roche a/s
Tlf: +45 - 36 39 99 99
Nederland
Roche Nederland B.V.
Tel: +31 (0) 348 438050
Deutschland
Roche Pharma AG
Tel: +49 (0) 7624 140
Norge
Roche Norge AS
Tlf: +47 - 22 78 90 00
Eesti
Roche Eesti OÜ
Tel: + 372 - 6 177 380
Österreich
Roche Austria GmbH
Tel: +43 (0) 1 27739
Ελλάδα
Roche (Hellas) A.E.
Τηλ: +30 210 61 66 100
Polska
Roche Polska Sp.z o.o.
Tel: +48 - 22 345 18 88
España
Roche Farma S.A.
Tel: +34 - 91 324 81 00
Portugal
Roche Farmacêutica Química, Lda
Tel: +351 - 21 425 70 00
France
Roche
Tél: +33 (0) 1 46 40 50 00
România
Roche România S.R.L.
Tel: +40 21 206 47 01
Ireland
Roche Products (Ireland) Ltd.
Tel: +353 (0) 1 469 0700
Slovenija
Roche farmacevtska družba d.o.o.
Tel: +386 - 1 360 26 00
Ísland
Roche a/s
c/o Icepharma hf
Sími: +354 540 8000
Slovenská republika
Roche Slovensko, s.r.o.
Tel: +421 - 2 52638201
Italia
Roche S.p.A.
Tel: +39 - 039 2471
Suomi/Finland
Roche Oy
Puh/Tel: +358 (0) 10 554 500
Kύπρος
Γ.Α.Σταμάτης & Σια Λτδ.
Τηλ: +357 - 22 76 62 76
Sverige
Roche AB
Tel: +46 (0) 8 726 1200
Latvija
Roche Latvija SIA
Tel: +371 - 6 7039831
United Kingdom
Roche Products Ltd.
Tel: +44 (0) 1707 366000
Lietuva
UAB “Roche Lietuva”
Tel: +370 5 2546799
This leaflet was last approved in {MM/YYYY}.
Detailed information on this medicine is available on the European Medicines Agency (EMEA) web
site: http://www.emea.europa.eu/.
Source: European Medicines Agency
 - Please bookmark this page (add it to your favorites).
- Please bookmark this page (add it to your favorites).
- If you wish to link to this page, you can do so by referring to the URL address below this line.
https://theodora.com/drugs/eu/tamiflu.html
Copyright © 1995-2021 ITA all rights reserved.
|



















































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































































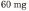

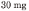


























 - Please bookmark this page (add it to your favorites).
- Please bookmark this page (add it to your favorites).










































